Abstract
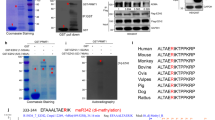
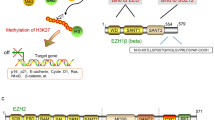
EZH2 overexpression occurs in various malignancies and is associated with a poor outcome. We have so far demonstrated that EZH2 downregulates the important genes such as E-cadherin and RUNX3 by increasing histone H3K27 trimethylation. However, the mechanism of EZH2 overexpression in various cancer cells remains unclear. In this study we carried out a promoter analysis of the EZH2 gene and investigated whether a survival signal that is upregulated in cancer cells is related to overexpression at the transcription level. We also explored the clinical relevance of the signaling pathway that leads to EZH2 overexpression in breast cancer and demonstrated that MEK–ERK1/2–Elk–1 pathway leads to EZH2 overexpression. The triple-negative and ERBB2-overexpressing subtypes of breast cancer are known to contain more rapidly proliferating breast cancer cells. The signaling pathway connected to EZH2 overexpression was associated with both aggressive subtypes of breast cancer. We show the significance that overexpression of histone modifier protein EZH2 in cancer cells and our study could pave the way for EZH2 inhibition to become an efficient treatment for more aggressive breast cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Arai A, Kanda E, Miura O . (2002). Rac is activated by erythropoietin or interleukin-3 and is involved in activation of the Erk signaling pathway. Oncogene 21: 2641–2651.
Bachmann IM, Halvorsen OJ, Collett K, Stefansson IM, Straume O, Haukaas SA et al. (2006). EZH2 expression is associated with high proliferation rate and aggressive tumor subgroups in cutaneous melanoma and cancers of the endometrium, prostate, and breast. J Clin Oncol 24: 268–273.
Cao R, Wang L, Wang H, Xia L, Erdjument-Bromage H, Tempst P et al. (2002). Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298: 1039–1043.
Cleator S, Heller W, Coombes RC . (2007). Triple-negative breast cancer: therapeutic options. Lancet Oncol 8: 235–244.
Crook T, Brooks LA, Crossland S, Osin P, Barker KT, Waller J et al. (1998). p53 mutation with frequent novel condons but not a mutator phenotype in BRCA1- and BRCA2-associated breast tumours. Oncogene 17: 1681–1689.
Foulkes WD, Brunet JS, Stefansson IM, Straume O, Chappuis PO, Bégin LR et al. (2004). The prognostic implication of the basal-like (cyclin E high/p27 low/p53+/glomeruloid-microvascular-proliferation+) phenotype of BRCA1-related breast cancer. Cancer Res 64: 830–835.
Fujii S, Ito K, Ito Y, Ochiai A . (2008). Enhancer of zeste homologue 2 (EZH2) down-regulates RUNX3 by increasing histone H3 methylation. J Biol Chem 283: 17324–17332.
Fujii S, Luo RZ, Yuan J, Kadota M, Oshimura M, Dent SR et al. (2003). Reactivation of the silenced and imprinted alleles of ARHI is associated with increased histone H3 acetylation and decreased histone H3 lysine 9 methylation. Hum Mol Genet 12: 1791–1800.
Fujii S, Ochiai A . (2008). Enhancer of zeste homolog 2 downregulates E-cadherin by mediating histone H3 methylation in gastric cancer cells. Cancer Sci 99: 738–746.
Futreal PA, Liu Q, Shattuck-Eidens D, Cochran C, Harshman K, Tavtigian S et al. (1994). BRCA1 mutations in primary breast and ovarian carcinomas. Science 266: 120–122.
Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ et al. (2010). Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 464: 1071–1076.
Hollestelle A, Nagel JH, Smid M, Lam S, Elstrodt F, Wasielewski M et al. (2010). Distinct gene mutation profiles among luminal-type and basal-type breast cancer cell lines. Breast Cancer Res Treat 121: 53–64.
Ito K, Liu Q, Salto-Tellez M, Yano T, Tada K, Ida H et al. (2005). RUNX3, a novel tumor suppressor, is frequently inactivated in gastric cancer by protein mislocalization. Cancer Res 65: 7743–7750.
Kenny PA, Lee GY, Myers CA, Neve RM, Semeiks JR, Spellman PT et al. (2007). The morphologies of breast cancer cell lines in three-dimensional assays correlate with their profiles of gene expression. Mol Oncol 1: 84–96.
Kleer CG, Cao Q, Varambally S, Shen R, Ota I, Tomlins SA et al. (2003). EZH2 is a marker of aggressive breast cancer and promotes neoplastic transformation of breast epithelial cells. Proc Natl Acad Sci USA 100: 11606–11611.
Konecny G, Pauletti G, Pegram M, Untch M, Dandekar S, Aguilar Z et al. (2003). Quantitative association between HER-2/neu and steroid hormone receptors in hormone receptor-positive primary breast cancer. J Natl Cancer Inst 95: 142–153.
Korsching E, Jeffrey SS, Meinerz W, Decker T, Boecker W, Buerger H . (2008). Basal carcinoma of the breast revisited: an old entity with new interpretations. J Clin Pathol 61: 553–560.
Morris DP, Michelotti GA, Schwinn DA . (2005). Evidence that phosphorylation of the RNA polymerase II carboxyl-terminal repeats is similar in yeast humans. J Biol Chem 280: 31368–31377.
Nielsen TO, Hsu FD, Jensen K, Cheang M, Karaca G, Hu Z et al. (2004). Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 10: 5367–5374.
Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. (2000). Molecular portraits of human breast tumours. Nature 406: 747–752.
Plath K, Fang J, Mlynarczyk-Evans SK, Cao R, Worringer KA, Wang H et al. (2003). Role of histone H3 lysine 27 methylation in X inactivation. Science 300: 131–135.
Puppe J, Drost R, Liu X, Joosse SA, Evers B, Cornelissen-Steijger P et al. (2009). BRCA1-deficient mammary tumor cells are dependent on EZH2 expression and sensitive to Polycomb Repressive Complex 2-inhibitor 3-deazaneplanocin A. Breast Cancer Res 11: R63.
Raaphorst FM, van Kemenade FJ, Blokzijl T, Fieret E, Hamer KM, Satijn DP et al. (2000). Coexpression of BMI-1 and EZH2 polycomb group genes in Reed-Sternberg cells of Hodgkin′s disease. Am J Pathol 157: 709–715.
Schuettengruber B, Chourrout D, Vervoort M, Leblanc B, Cavalli G . (2007). Genome regulation by polycomb and trithorax proteins. Cell 128: 735–745.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE et al. (1989). Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244: 707–712.
Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al. (2001). Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98: 10869–10874.
Varambally S, Cao Q, Mani RS, Shankar S, Wang X, Ateeq B et al. (2008). Genomic loss of microRNA-101 leads to overexpression of histone methyltransferase EZH2 in cancer. Science 322: 1695–1699.
Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG et al. (2002). The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 419: 624–629.
Acknowledgements
We wish to acknowledge the assistance of Ms Hiroko Hashimoto for cell culture support and the assistance of Mr Shinya Yanagi for making paraffin-embedded thin sections for immunohistochemistry. The research described in this report has been funded by a grant from the Ministry of Education, Culture, Sports, Science and Technology, Japan (21590453), to SF. Individual contributions of each author; Study concept and design; SF, AO, Acquisition of data; SF, KT, Analysis and interpretation of data; SF, KT, NW, Drafting the manuscript; SF, Critical revision of the manuscript for important intellectual content; SF, KI, YI, AO. Statistical analysis; SF, KT, NW Obtained funding; SF, Material support; NW, KI, YI, CY, Study supervision; SF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Fujii, S., Tokita, K., Wada, N. et al. MEK–ERK pathway regulates EZH2 overexpression in association with aggressive breast cancer subtypes. Oncogene 30, 4118–4128 (2011). https://doi.org/10.1038/onc.2011.118
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.118
Keywords
This article is cited by
-
The roles of EZH2 in cancer and its inhibitors
Medical Oncology (2023)
-
DNA methylation of miR-138 regulates cell proliferation and EMT in cervical cancer by targeting EZH2
BMC Cancer (2022)
-
B7 homolog 3 induces lung metastasis of breast cancer through Raf/MEK/ERK axis
Breast Cancer Research and Treatment (2022)
-
Increased CD44 Expression and MEK Activity Predict Worse Prognosis in Gastric Adenocarcinoma Patients Undergoing Gastrectomy
Journal of Gastrointestinal Surgery (2021)
-
EZH2 overexpression dampens tumor-suppressive signals via an EGR1 silencer to drive breast tumorigenesis
Oncogene (2020)