Abstract
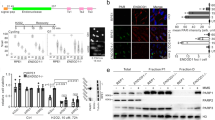
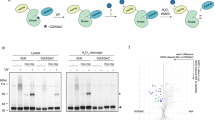
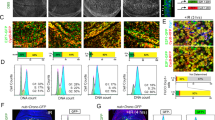
The programmed cell death gene 4 (Pdcd4) gene has been implicated as a new tumor suppressor gene in the development of several types of human cancer. Pdcd4 interacts with the translation initiation factor, eIF4A, and is thought to act as a translation inhibitor. Here, we have used the chicken B-cell line DT40 to disrupt the Pdcd4 gene by homologous recombination. Our study shows that cells lacking a functional Pdcd4 gene are viable and have no obvious defects when cultivated under normal growth conditions. However, Pdcd4 knockout cells show an increased sensitivity to agents that cause DNA damage, such as UV light, etoposide or ethyl-methanesulfonate. In summary, our findings show that Pdcd4 has an important function in the cellular response to DNA damage. Low Pdcd4 expression, which is frequently observed in tumor cells, might therefore contribute to tumorigenesis by disturbing the cellular DNA-damage response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Asangani IA, Rasheed SA, Nikolova DA, Leupold JH, Colburn NH, Post S et al. (2008). MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 27: 2128–2136.
Bezzubova O, Silbergleit A, Yamaguchi-Iwai Y, Takeda S, Buerstedde JM . (1997). Reduced X-ray resistance and homologous recombination frequencies in a RAD54−/− mutant of the chicken DT40 cell line. Cell 89: 185–193.
Bitomsky N, Böhm M, Klempnauer K-H . (2004). Transformation suppressor protein Pdcd4 interferes with JNK-mediated phosphorylation of c-Jun and recruitment of the coactivator p300 by c-Jun. Oncogene 23: 7484–7493.
Bitomsky N, Wethkamp N, Marikkannu R, Klempnauer K-H . (2008). siRNA-mediated knock-down of Pdcd4 expression causes up-regulation of p21(Waf1/Cip1) expression. Oncogene 27: 4820–4829.
Böhm M, Sawicka K, Siebrasse JP, Brehmer-Fastnacht A, Peters R, Klempnauer K-H . (2003). The transformation suppressor protein Pdcd4 shuttles between nucleus and cytoplasm and binds RNA. Oncogene 22: 4905–4910.
Buerstedde JM, Takeda S . (1991). Increased ratio of targeted to random integration after transfection of chicken B cell lines. Cell 67: 179–188.
Chang JH, Cho YH, Sohn SY, Choi JM, Kim A, Kim YC et al. (2009). Cyrstal structure of the eIF4A–PDCD4 complex. Proc Natl Acad Sci USA 106: 3148–3153.
Chen Y, Knosel T, Kristiansen G, Pietas A, Garber ME, Matsuhashi S et al. (2003). Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. J Pathol 200: 640–646.
Cmarik JL, Min H, Hegamyer G, Zhan S, Kulesz-Martin M, Yoshinaga H et al. (1999). Differentially expressed protein Pdcd4 inhibits tumor promoter-induced neoplastic transformation. Proc Natl Acad Sci USA 96: 14037–14042.
Dorello NV, Peschiaroli A, Guardavaccaro D, Colburn NH, Sherman NE, Pagano M . (2006). S6K1- and betaTRCP-mediated degradation of PDCD4 promotes protein translation and cell growth. Science 314: 467–471.
Frankel LB, Christoffersen NR, Jacobsen A, Lindow M, Krogh A, Lund AH . (2008). Programmed cell death 4 (PDCD4) is an important functional target of the microRNA miR-21 in breast cancer cells. J Biol Chem 283: 1026–1033.
Göke A, Göke R, Knolle A, Trusheim H, Schmidt H, Wilmen A et al. (2002). DUG is a novel homologue of translation initiation factor 4G that binds eIF4A. Biochem Biophys Res Commun 297: 78–82.
Gorospe M, Cirielli C, Wang X, Seth P, Capogrossi MC, Holbrook NJ . (1997). p21(Waf1/Cip1) protects against p53-mediated apoptosis of human melanoma cells. Oncogene 14: 929–935.
Hershey JWB, Merrick WC . (2000). Translational control of gene expression: pathway and mechanism of initiation of protein synthesis. In: Sonenberg N, Hershey JWB, Mathews MB (eds). Cold Spring Harbor Laboratory Press, Cold Spring Harbor: New York. pp 33–88.
Jansen AP, Camalier CE, Colburn NH . (2005). Epidermal expression of the translation inhibitor programmed cell death 4 suppresses tumorigenesis. Cancer Res 65: 6034–6041.
LaRonde-Blanc N, Santhanam AN, Baker AR, Wlodawer A, Colburn NH . (2007). Structural basis for inhibition of translation by the tumor suppressor Pdcd4. Mol Cell Biol 27: 147–156.
Leupold JH, Yang HS, Colburn NH, Asangani I, Post S, Allgayer H . (2007). Tumor suppressor Pdcd4 inhibits invasion/intravasation and regulates urokinase receptor (u-PAR) gene expression via Sp-transcription factors. Oncogene 26: 4550–4562.
Loh PG, Yang HS, Walsh MA, Wang Q, Wang X, Cheng Z et al. (2009). Structural basis for translational inhibition by the tumour suppressor Pdcd4. EMBO J 28: 274–285.
Lu Z, Liu M, Stribinskis V, Klinge CM, Ramos KS, Colburn NH . (2008). MicroRNA-21 promotes cell transformation by targeting the programmed cell death 4 gene. Oncogene 27: 4373–4379.
Mudduluru G, Medved F, Grobholz R, Jost C, Gruber A, Leupold JH et al. (2007). Loss of programmed cell death 4 expression marks adenoma-carcinoma transition, correlates inversely with phosphorylated protein kinase B, and is an independent prognostic factor in resected colorectal cancer. Cancer 110: 1697–1707.
Palamarchuk A, Efanov A, Maximov V, Aqeilan RI, Croce CM, Pekarsky Y . (2005). Akt phosphorylates and regulates Pdcd4 tumor suppressor protein. Cancer Res 65: 11282–11286.
Schlichter U, Kattmann D, Appl H, Miethe J, Brehmer-Fastnacht A, Klempnauer K-H . (2001). Identification of the myb-inducible promoter of the chicken Pdcd4 gene. Biochim Biophys Acta 1520: 99–104.
Seoane J, Le HV, Massague J . (2002). Myc suppression of the p21(Cip1) Cdk inhibitor influences the outcome of the p53 response to DNA damage. Nature 419: 729–734.
Shibahara K, Asano M, Ishida Y, Aoki T, Koike T, Honjo T . (1995). Isolation of a novel mouse gene MA-3 that is induced upon programmed cell death. Gene 166: 297–301.
Suzuki C, Garces RG, Edmonds KA, Hiller S, Hyberts SG, Marintchev A et al. (2008). PDCD4 inhibits translation initiation by binding to eIF4A using both its MA3 domains. Proc Natl Acad Sci USA 105: 3274–3279.
Takao N, Kato H, Mori R, Morrison C, Sonada E, Sun X et al. (1999). Disruption of ATM in p53-null cells causes multiple functional abnormalities in cellular response to ionizing radiation. Oncogene 18: 7002–7009.
Wang Q, Sun Z, Yang H-S . (2008). Downregulation of tumour suppressor Pdcd4 promotes invasion and activates both β-catenin/TCF and AP-1-dependent transcription in colon carcinoma cells. Oncogene 27: 1527–1535.
Waters LC, Veverka V, Böhm M, Schmedt T, Choong PT, Muskett FW et al. (2007). Structure of the C-terminal MA-3 domain of the tumour suppressor protein Pdcd4 and characterization of its interaction with eIF4A. Oncogene 26: 4941–4950.
Yang HS, Cho MH, Zacowicz H, Hegamyer G, Sonenberg N, Colburn N . (2004). A novel function of the MA-3 domains in transformation and translation suppressor Pdcd4 is essential for its binding to eukaryotic translation initiation factor 4A. Mol Cell Biol 24: 3894–3906.
Yang HS, Jansen AP, Komar AA, Zheng X, Merrick WC, Costes S et al. (2003a). The transformation suppressor Pdcd4 is a novel eukaryotic translation initiation factor 4A binding protein that inhibits translation. Mol Cell Biol 23: 26–37.
Yang HS, Knies JL, Stark C, Colburn NH . (2003b). Pdcd4 suppresses tumor phenotype in JB6 cells by inhibiting AP-1 transactivation. Oncogene 22: 3712–3720.
Yang HS, Matthews CP, Clair T, Wang Q, Baker AR, Li C-CH et al. (2006). Tumorigenesis suppressor Pdcd4 down-regulates mitogen-activated protein kinase kinase kinase kinase 1 expression to suppress colon carcinoma cell invasion. Mol Cell Biol 26: 1297–1306.
Zhang H, Ozaki I, Mizuta T, Hamajima H, Yasutake T, Eguchi Y et al. (2006). Involvement of programmed cell death 4 in transforming growth factor-beta1-induced apoptosis in human hepatocellular carcinoma. Oncogene 25: 6101–6112.
Acknowledgements
We thank B Berkenfeld for excellent technical assistance. This study was supported by grants from the Deutsche Krebshilfe (10-1716) and the Wilhelm-Sander-Stiftung (2004.088.1). PS and RM were supported by fellowships from the Graduate School of Chemistry (GSC-MS) at the University of Münster.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, P., Marikkannu, R., Bitomsky, N. et al. Disruption of the Pdcd4 tumor suppressor gene in chicken DT40 cells reveals its role in the DNA-damage response. Oncogene 28, 3758–3764 (2009). https://doi.org/10.1038/onc.2009.239
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.239
Keywords
This article is cited by
-
PDCD4 controls the G1/S-phase transition in a telomerase-immortalized epithelial cell line and affects the expression level and translation of multiple mRNAs
Scientific Reports (2020)
-
SKP2 promotes breast cancer tumorigenesis and radiation tolerance through PDCD4 ubiquitination
Journal of Experimental & Clinical Cancer Research (2019)
-
A novel mechanism for the control of translation of specific mRNAs by tumor suppressor protein Pdcd4: inhibition of translation elongation
Oncogene (2015)
-
Tumor suppressor protein Pdcd4 interacts with Daxx and modulates the stability of Daxx and the Hipk2-dependent phosphorylation of p53 at serine 46
Oncogenesis (2013)
-
Pdcd4 directly binds the coding region of c-myb mRNA and suppresses its translation
Oncogene (2011)