Key Points
-
CD8+ T-cell homeostasis is altered in the peripheral blood of patients with autoimmune arthritis
-
The close association of MHC class I polymorphisms with disease risk and the correlation of CD8+ T-cell number with disease outcome support the idea that this cell population has a role in autoimmune arthritis
-
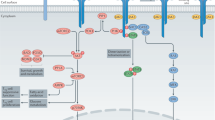
At sites of chronic inflammation, the phenotype of CD8+ T cells is heterogeneous and includes proinflammatory and anti-inflammatory features
-
In inflammatory environments, CD8+ T cells lose susceptibility to regulation, and this loss is sustained by autocrine release of proinflammatory cytokines
-
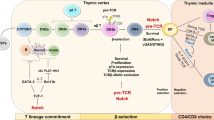
Subsets of memory CD8+ T cells upregulate negative costimulatory markers and either develop an exhausted phenotype or display a tissue-instructed differentiation, as in tissue-resident memory T cells
Abstract
CD8+ T cells are key players in the body's defence against viral infections and cancer. To date, data on the role of CD8+ T cells in autoimmune diseases have been scarce, especially when compared with the wealth of research on CD4+ T cells. However, growing evidence suggests that CD8+ T-cell homeostasis is impaired in human autoimmune diseases. The contribution of CD8+ T cells to autoimmune arthritis is indicated by the close association of MHC class I polymorphisms with disease risk, as well as the correlation between CD8+ T-cell phenotype and disease outcome. The heterogeneous phenotype, resistance to regulation and impaired regulatory function of CD8+ T cells — especially at the target organ — might contribute to the persistence of autoimmune inflammation. Moreover, newly identified populations of tissue-resident CD8+ T cells and their interaction with antigen-presenting cells might have a key role in disease pathology. In this Review, we assess the link between CD8+ T cells, autoimmune arthritis and the basis of their homeostatic changes under inflammatory conditions. Improved insight into CD8+ T cell-specific pathogenicity will be essential for a better understanding of autoimmune arthritis and the identification of new therapeutic targets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Palmer, M. T. & Weaver, C. T. Autoimmunity: increasing suspects in the CD4+ T cell lineup. Nat. Immunol. 11, 36–40 (2010).
Gravano, D. M. & Hoyer, K. K. Promotion and prevention of autoimmune disease by CD8+ T cells. J. Autoimmun. 45, 68–79 (2013).
Brewerton, D. A. et al. Ankylosing spondylitis and HLA 27. Lancet 1, 904–907 (1973).
Brown, M. A., Kenna, T. & Wordsworth, B. P. Genetics of ankylosing spondylitis-insights into pathogenesis. Nat. Rev. Rheumatol. 12, 81–91 (2016).
Evans, D. M. et al. Interaction between ERAP1 and HLAB27 in ankylosing spondylitis implicates peptide handling in the mechanism for HLAB27 in disease susceptibility. Nat. Genet. 43, 761–767 (2011).
Cortes, A. et al. Major histocompatibility complex associations of ankylosing spondylitis are complex and involve further epistasis with ERAP1. Nat. Commun. 6, 7146 (2015).
Bowes, J. et al. Dense genotyping of immune-related susceptibility loci reveals new insights into the genetics of psoriatic arthritis. Nat. Commun. 6, 6046 (2015).
Raychaudhuri, S. et al. Five amino acids in three HLA proteins explain most of the association between MHC and seropositive rheumatoid arthritis. Nat. Genet. 44, 291–296 (2012).
Hollenbach, J. A. et al. Juvenile idiopathic arthritis and HLA class I and class II interactions and ageatonset effects. Arthritis Rheum. 62, 1781–1791 (2010).
Hinks, A. et al. Dense genotyping of immune-related disease regions identifies 14 new susceptibility loci for juvenile idiopathic arthritis. Nat. Genet. 45, 664–669 (2013).
Ombrello, M. J. et al. HLADRB1*11 and variants of the MHC class II locus are strong risk factors for systemic juvenile idiopathic arthritis. Proc. Natl Acad. Sci. USA 115, 15970–15975 (2015).
Coulthard, L. R. et al. Differential effects of infliximab on absolute circulating blood leucocyte counts of innate immune cells in early and late rheumatoid arthritis patients. Clin. Exp. Immunol. 170, 36–46 (2012).
Carvalheiro, H., Duarte, C., Silva-Cardoso, S., da Silva, J. A. & Souto-Carneiro, M. M. CD8+ T cell profiles in patients with rheumatoid arthritis and their relationship to disease activity. Arthritis Rheumatol. 67, 363–371 (2015).
Li, S. et al. Expression of programmed death1 (PD1) on CD4+ and CD8+ T cells in rheumatoid arthritis. Inflammation 37, 116–121 (2014).
Liu, Y. et al. Increased Tim3 expression on peripheral lymphocytes from patients with rheumatoid arthritis negatively correlates with disease activity. Clin. Immunol. 137, 288–295 (2010).
Costello, P., Bresnihan, B., O'Farrelly, C. & FitzGerald, O. Predominance of CD8+ T lymphocytes in psoriatic arthritis. J. Rheumatol. 26, 1117–1124 (1999).
Hunter, P. J. et al. Biologic predictors of extension of oligoarticular juvenile idiopathic arthritis as determined from synovial fluid cellular composition and gene expression. Arthritis Rheum. 62, 896–907 (2010).
Cho, B. A. et al. Characterization of effector memory CD8+ T cells in the synovial fluid of rheumatoid arthritis. J. Clin. Immunol. 32, 709–720 (2012).
Romio, M. et al. Extracellular purine metabolism and signaling of CD73derived adenosine in murine Treg and Teff cells. Am. J. Physiol. Cell Physiol. 301, C530–C539 (2011).
Botta Gordon-Smith, S., Ursu, S., Eaton, S., Moncrieffe, H. & Wedderburn, L. R. Correlation of low CD73 expression on synovial lymphocytes with reduced adenosine generation and higher disease severity in juvenile idiopathic arthritis. Arthritis Rheumatol. 67, 545–554 (2015).
Kohlmann, W. M., Urban, W., Sterry, W. & Foerster, J. Correlation of psoriasis activity with abundance of CD25+CD8+ T cells: conditions for cloning T cells from psoriatic plaques. Exp. Dermatol. 13, 607–612 (2004).
Menon, B. et al. Interleukin17+CD8+ T cells are enriched in the joints of patients with psoriatic arthritis and correlate with disease activity and joint damage progression. Arthritis Rheumatol. 66, 1272–1281 (2014).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Kaech, S. M. & Cui, W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nat. Rev. Immunol. 12, 749–761 (2012).
Borgato, L. et al. The T cell receptor repertoire in psoriatic synovitis is restricted and T lymphocytes expressing the same TCR are present in joint and skin lesions. J. Rheumatol. 29, 1914–1919 (2002).
Mueller, S. N. & Mackay, L. K. Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol. 16, 79–89 (2016).
Vukmanovic-Stejic, M., Vyas, B., Gorak-Stolinska, P., Noble, A. & Kemeny, D. M. Human Tc1 and Tc2/Tc0 CD8 Tcell clones display distinct cell surface and functional phenotypes. Blood 95, 231–240 (2000).
Yen, H. R. et al. Tc17 CD8 T cells: functional plasticity and subset diversity. J. Immunol. 183, 7161–7168 (2009).
Liu, Y. et al. Interleukin21 induces the differentiation of human Tc22 cells via phosphorylation of signal transducers and activators of transcription. Immunology 132, 540–548 (2011).
Luan, L., Ding, Y., Han, S., Zhang, Z. & Liu, X. An increased proportion of circulating Th22 and Tc22 cells in psoriasis. Cell. Immunol. 290, 196–200 (2014).
Ortega, C. et al. IL17producing CD8+ T lymphocytes from psoriasis skin plaques are cytotoxic effector cells that secrete Th17related cytokines. J. Leukoc. Biol. 86, 435–443 (2009).
Kirkham, B. W., Kavanaugh, A. & Reich, K. Interleukin17A: a unique pathway in immune-mediated diseases: psoriasis, psoriatic arthritis and rheumatoid arthritis. Immunology 141, 133–142 (2014).
Prezzemolo, T. et al. Functional signatures of human CD4 and CD8 T cell responses to Mycobacterium tuberculosis. Front. Immunol. 5, 180 (2014).
Li, S. et al. Expression of TIM3 on CD4+ and CD8+ T cells in the peripheral blood and synovial fluid of rheumatoid arthritis. APMIS 122, 899–904 (2014).
Kang, Y. M. et al. CD8 T cells are required for the formation of ectopic germinal centers in rheumatoid synovitis. J. Exp. Med. 195, 1325–1336 (2002).
Harada, S. et al. Production of interleukin7 and interleukin15 by fibroblast-like synoviocytes from patients with rheumatoid arthritis. Arthritis Rheum. 42, 1508–1516 (1999).
McInnes, I. B. et al. The role of interleukin15 in T cell migration and activation in rheumatoid arthritis. Nat. Med. 2, 175–182 (1996).
Fitzgerald, J. E. et al. Analysis of clonal CD8+ T cell expansions in normal individuals and patients with rheumatoid arthritis. J. Immunol. 154, 3538–3547 (1995).
Hingorani, R. et al. Oligoclonality of V beta 3 TCR chains in the CD8+ T cell population of rheumatoid arthritis patients. J. Immunol. 156, 852–858 (1996).
Hall, F. C., Thomson, K., Procter, J., McMichael, A. J. & Wordsworth, B. P. TCRβ spectratyping in RA: evidence of clonal expansions in peripheral blood lymphocytes. Ann. Rheum. Dis. 57, 319–322 (1998).
Fazou, C., Yang, H., McMichael, A. J. & Callan, M. F. Epitope specificity of clonally expanded populations of CD8+ T cells found within the joints of patients with inflammatory arthritis. Arthritis Rheum. 44, 2038–2045 (2001).
Klarenbeek, P. L. et al. Inflamed target tissue provides a specific niche for highly expanded Tcell clones in early human autoimmune disease. Ann. Rheum. Dis. 71, 1088–1093 (2012).
Sathaliyawala, T. et al. Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity 38, 187–197 (2013).
Iijima, N. & Iwasaki, A. Tissue instruction for migration and retention of TRM cells. Trends Immunol. 36, 556–564 (2015).
Thome, J. J. et al. Spatial map of human T cell compartmentalization and maintenance over decades of life. Cell 159, 814–828 (2014).
Farber, D. L., Yudanin, N. A. & Restifo, N. P. Human memory T cells: generation, compartmentalization and homeostasis. Nat. Rev. Immunol. 14, 24–35 (2014).
Mackay, L. K. et al. The developmental pathway for CD103+CD8+ tissue-resident memory T cells of skin. Nat. Immunol. 14, 1294–1301 (2013).
Clark, R. A. Resident memory T cells in human health and disease. Sci. Transl. Med. 7, 269rv261 (2015).
Cheuk, S. et al. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J. Immunol. 192, 3111–3120 (2014).
Henderson, L. A. et al. Novel 3dimensional explant method facilitates the study of lymphocyte populations in the synovium and reveals a large population of resident memory T cells in rheumatoid arthritis. Arthritis Rheum. 66, S209–S209 (2014).
Keir, M. E., Butte, M. J., Freeman, G. J. & Sharpe, A. H. PD1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 26, 677–704 (2008).
Okazaki, T., Chikuma, S., Iwai, Y., Fagarasan, S. & Honjo, T. A rheostat for immune responses: the unique properties of PD1 and their advantages for clinical application. Nat. Immunol. 14, 1212–1218 (2013).
Butte, M. J., Keir, M. E., Phamduy, T. B., Sharpe, A. H. & Freeman, G. J. Programmed death1 ligand 1 interacts specifically with the B71 costimulatory molecule to inhibit T cell responses. Immunity 27, 111–122 (2007).
Schneider, H. et al. Reversal of the TCR stop signal by CTLA4. Science 313, 1972–1975 (2006).
Francisco, L. M. et al. PDL1 regulates the development, maintenance, and function of induced regulatory T cells. J. Exp. Med. 206, 3015–3029 (2009).
Oderup, C., Cederbom, L., Makowska, A., Cilio, C. M. & Ivars, F. Cytotoxic T lymphocyte antigen-4dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory Tcell-mediated suppression. Immunology 118, 240–249 (2006).
Sehrawat, S. et al. Galectin9/TIM3 interaction regulates virus-specific primary and memory CD8 T cell response. PLoS Pathog. 6, e1000882 (2010).
Kim, P. S. & Ahmed, R. Features of responding T cells in cancer and chronic infection. Curr. Opin. Immunol. 22, 223–230 (2010).
Speiser, D. E. et al. T cell differentiation in chronic infection and cancer: functional adaptation or exhaustion? Nat. Rev. Immunol. 14, 768–774 (2014).
Wakim, L. M. et al. The molecular signature of tissue resident memory CD8 T cells isolated from the brain. J. Immunol. 189, 3462–3471 (2012).
Raptopoulou, A. P. et al. The programmed death 1/programmed death ligand 1 inhibitory pathway is upregulated in rheumatoid synovium and regulates peripheral T cell responses in human and murine arthritis. Arthritis Rheum. 62, 1870–1880 (2010).
Schneider, A. et al. The effector T cells of diabetic subjects are resistant to regulation via CD4+FOXP3+ regulatory T cells. J. Immunol. 181, 7350–7355 (2008).
Schneider, A. et al. In active relapsing-remitting multiple sclerosis, effector T cell resistance to adaptive Tregs involves IL6mediated signaling. Sci. Transl. Med. 5, 170ra115 (2013).
Venigalla, R. K. et al. Reduced CD4+,CD25− T cell sensitivity to the suppressive function of CD4+,CD25high, CD127−/low regulatory T cells in patients with active systemic lupus erythematosus. Arthritis Rheum. 58, 2120–2130 (2008).
Xiao, H. TRAIL is associated with impaired regulation of CD4+CD25− T cells by regulatory T cells in patients with rheumatoid arthritis. J. Clin. Immunol. 6, 1112–1119 (2011).
Wehrens, E. J. et al. Functional human regulatory T cells fail to control autoimmune inflammation due to PKB/cakt hyperactivation in effector cells. Blood 118, 3538–3548 (2011).
Haufe, S. et al. Impaired suppression of synovial fluid CD4+CD25− T cells from patients with juvenile idiopathic arthritis by CD4+CD25+ Treg cells. Arthritis Rheum. 63, 3153–3162 (2011).
Petrelli, A. et al. Self-sustained resistance to suppression of CD8+ Teff cells at the site of autoimmune inflammation can be reversed by tumor necrosis factor and interferon-γ blockade. Arthritis Rheumatol. 68, 229–236 (2016).
Wehrens, E. J. et al. Anti-tumor necrosis factor α targets protein kinase B/cAkt-induced resistance of effector cells to suppression in juvenile idiopathic arthritis. Arthritis Rheum. 65, 3279–3284 (2013).
Rifa'i, M., Kawamoto, Y., Nakashima, I. & Suzuki, H. Essential roles of CD8+CD122+ regulatory T cells in the maintenance of T cell homeostasis. J. Exp. Med. 200, 1123–1134 (2004).
Moore, J. S. & Calkins, C. E. In vitro regulation of the pathogenic autoantibody response of New Zealand black mice. I. Loss with age of suppressive activity in T cell populations. J. Immunol. 134, 3838–3844 (1985).
Notley, C. A., McCann, F. E., Inglis, J. J. & Williams, R. O. ANTICD3 therapy expands the numbers of CD4+ and CD8+ Treg cells and induces sustained amelioration of collagen-induced arthritis. Arthritis Rheum. 62, 171–178 (2010).
Yamaguchi, T., Wing, J. B. & Sakaguchi, S. Two modes of immune suppression by Foxp3+ regulatory T cells under inflammatory or non-inflammatory conditions. Seminars Immunol. 23, 424–430 (2011).
Li, S. et al. A naturally occurring CD8+CD122+ Tcell subset as a memory-like Treg family. Cell. Mol. Immunol. 11, 326–331 (2014).
Cosmi, L. et al. Human CD8+CD25+ thymocytes share phenotypic and functional features with CD4+CD25+ regulatory thymocytes. Blood 102, 4107–4114 (2003).
Ceeraz, S., Hall, C., Choy, E. H., Spencer, J. & Corrigall, V. M. Defective CD8+CD28− regulatory T cell suppressor function in rheumatoid arthritis is restored by tumour necrosis factor inhibitor therapy. Clin. Exp. Immunol. 174, 18–26 (2013).
Ellis, S. D. et al. Induced CD8+FoxP3+ Treg cells in rheumatoid arthritis are modulated by p38 phosphorylation and monocytes expressing membrane tumor necrosis factor α and CD86. Arthritis Rheumatol. 66, 2694–2705 (2014).
Guilliams, M. et al. Dendritic cells, monocytes and macrophages: a unified nomenclature based on ontogeny. Nat. Rev. Immunol. 14, 571–578 (2014).
Boltjes, A. & van Wijk, F. Human dendritic cell functional specialization in steady-state and inflammation. Front. Immunol. 5, 131 (2014).
Ziegler-Heitbrock, L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J. Leukoc. Biol. 81, 584–592 (2007).
Thomas, R. & Quinn, C. Functional differentiation of dendritic cells in rheumatoid arthritis: role of CD86 in the synovium. J. Immunol. 156, 3074–3086 (1996).
Moret, F. M. et al. Intra-articular CD1cexpressing myeloid dendritic cells from rheumatoid arthritis patients express a unique set of T cell-attracting chemokines and spontaneously induce Th1, Th17 and Th2 cell activity. Arthritis Res. Ther. 15, R155 (2013).
Jongbloed, S. L. et al. Enumeration and phenotypical analysis of distinct dendritic cell subsets in psoriatic arthritis and rheumatoid arthritis. Arthritis Res. Ther. 8, R15 (2006).
Kawanaka, N. et al. CD14+,CD16+ blood monocytes and joint inflammation in rheumatoid arthritis. Arthritis Rheum. 46, 2578–2586 (2002).
Yoon, B. R. et al. Functional phenotype of synovial monocytes modulating inflammatory Tcell responses in rheumatoid arthritis (RA). PLoS ONE 9, e109775 (2014).
Jongbloed, S. L. et al. Human CD141+ (BDCA3)+ dendritic cells (DCs) represent a unique myeloid DC subset that cross-presents necrotic cell antigens. J. Exp. Med. 207, 1247–1260 (2010).
Kassianos, A. J., Jongbloed, S. L., Hart, D. N. & Radford, K. J. Isolation of human blood DC subtypes. Methods Mol. Biol. 595, 45–54 (2010).
Kurts, C. et al. Constitutive class Irestricted exogenous presentation of self antigens in vivo. J. Exp. Med. 184, 923–930 (1996).
Schreibelt, G. et al. The Ctype lectin receptor CLEC9A mediates antigen uptake and (cross-)presentation by human blood BDCA3+ myeloid dendritic cells. Blood 119, 2284–2292 (2012).
Haniffa, M. et al. Human tissues contain CD141hi cross-presenting dendritic cells with functional homology to mouse CD103+ nonlymphoid dendritic cells. Immunity 37, 60–73 (2012).
Smolewska, E. et al. Distribution and clinical significance of blood dendritic cells in children with juvenile idiopathic arthritis. Ann. Rheum. Dis. 67, 762–768 (2008).
Prakken, B., Albani, S. & Martini, A. Juvenile idiopathic arthritis. Lancet 377, 2138–2149 (2011).
Sakkas, L. I., Bogdanos, D. P., Katsiari, C. & Platsoucas, C. D. Anti-citrullinated peptides as autoantigens in rheumatoid arthritis-relevance to treatment. Autoimmun. Rev. 13, 1114–1120 (2014).
Bulatovic Calasan, M. et al. Methotrexate treatment affects effector but not regulatory T cells in juvenile idiopathic arthritis. Rheumatology 54, 1724–1734 (2015).
Aravena, O. et al. Anti-TNF therapy in patients with rheumatoid arthritis decreases Th1 and Th17 cell populations and expands IFN-γ-producing NK cell and regulatory T cell subsets. Immunobiology 216, 1256–1263 (2011).
Scarsi, M. et al. Reduction of peripheral blood T cells producing IFN-γ and IL17 after therapy with abatacept for rheumatoid arthritis. Clin. Exp. Rheumatol. 32, 204–210 (2014).
Melet, J. et al. Rituximab-induced T cell depletion in patients with rheumatoid arthritis: association with clinical response. Arthritis Rheum. 65, 2783–2790 (2013).
Nejentsev, S. et al. Localization of type 1 diabetes susceptibility to the MHC class I genes HLAB and HLAA. Nature 450, 887–892 (2007).
Sawcer, S. et al. Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis. Nature 476, 214–219 (2011).
Coppieters, K. T. et al. Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. J. Exp. Med. 209, 51–60 (2012).
Unger, W. W. et al. Islet-specific CTL cloned from a type 1 diabetes patient cause beta-cell destruction after engraftment into HLAA2 transgenic NOD/scid/IL2RG null mice. PLoS ONE 7, e49213 (2012).
Skulina, C. et al. Multiple sclerosis: brain-infiltrating CD8+ T cells persist as clonal expansions in the cerebrospinal fluid and blood. Proc. Natl Acad. Sci. USA 101, 2428–2433 (2004).
Crawford, M. P. et al. High prevalence of autoreactive, neuroantigen-specific CD8+ T cells in multiple sclerosis revealed by novel flow cytometric assay. Blood 103, 4222–4231 (2004).
McKinney, E. F. et al. A CD8+ T cell transcription signature predicts prognosis in autoimmune disease. Nat. Med. 16, 586–591 (2010).
McKinney, E. F., Lee, J. C., Jayne, D. R., Lyons, P. A. & Smith, K. G. Tcell exhaustion, costimulation and clinical outcome in autoimmunity and infection. Nature 523, 612–616 (2015).
Acknowledgements
A.P. is supported by the European Union Seventh Framework Programme (501100004963, 289903). F.V.W. is supported by the VIDI grant from the Netherlands Organization for Scientific Research (NWO, ZonMW) and by the Dutch Arthritis Foundation Reumafonds
Author information
Authors and Affiliations
Contributions
A.P. researched data for the article and wrote the manuscript. F.V.W. reviewed and edited the manuscript before submission. A.P. and F.V.W. contributed equally to discussions of the content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Petrelli, A., van Wijk, F. CD8+ T cells in human autoimmune arthritis: the unusual suspects. Nat Rev Rheumatol 12, 421–428 (2016). https://doi.org/10.1038/nrrheum.2016.74
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2016.74
This article is cited by
-
Virulence factors and mechanisms of paediatric pneumonia caused by Enterococcus faecalis
Gut Pathogens (2023)
-
Transcriptomic profiling of programmed cell death 1 (PD-1) expressing T cells in early rheumatoid arthritis identifies a decreased CD4 + PD-1 + signature post-treatment
Scientific Reports (2023)
-
Rheumatoid nodules: a narrative review of histopathological progression and diagnostic consideration
Clinical Rheumatology (2023)
-
Low positivity rates for HBeAg and HBV DNA in rheumatoid arthritis patients: a case–control study
BMC Infectious Diseases (2022)
-
Navigating the diverse immune landscapes of psoriatic arthritis
Seminars in Immunopathology (2021)