Key Points
-
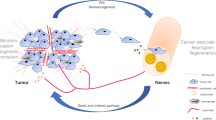
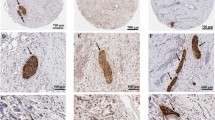
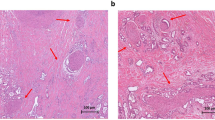
Perineural invasion (PNI) is the process through which cancer cells invade the perineural spaces of surrounding nerves and is not simply the movement of cancer cells along a path of low resistance, as was previously thought. PNI is a directed process that involves many signalling molecules from various signalling pathways; these signalling molecules are produced by both the cancer cells and the nerves. Once the cancer cells have invaded the nerves, they are able to thrive within the neuronal spaces. This constitutes a means for the cancer cells to spread to distant locations.
-
The incidence of PNI is particularly high in pancreatic cancer. Although the exact cause for this increased affinity is as yet unclear, the strong neurotropic effects of pancreatic cancer cells are thought to contribute to this phenomenon. Additionally, reciprocal signalling between the pancreatic cancer cells and the surrounding nerves leads to neurogenesis, as well as the increased growth of pancreatic cancer cells.
-
PNI also contributes to the generation of the pain that is experienced by pancreatic cancer patients, and many of the signalling molecules that are involved in PNI are also known to be involved in pain signalling. Thus, we hypothesize that agents targeting these signalling pathways may have the potential to prevent PNI and may help to alleviate pain in patients with pancreatic cancer.
Abstract
Perineural invasion (PNI) is a prominent characteristic of pancreatic cancer. PNI is a process whereby cancer cells invade the surrounding nerves, thus providing an alternative route for metastatic spread and pain generation. PNI is thought to be an indicator of aggressive tumour behaviour and has been shown to correlate with poor prognosis of patients with pancreatic cancer. Recent studies demonstrated that some signalling molecules and pathways that are involved in PNI are also involved in pain generation. Targeting these signalling pathways has shown some promise in alleviating pain and reducing PNI, which could potentially improve treatment outcomes for patients with pancreatic cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lesnik, D. J. & Boey, H. P. Perineural invasion of the facial nerve by a cutaneous squamous cell cancer: a case report. Ear Nose Throat J. 83, 826–827 (2004).
Liebig, C., Ayala, G., Wilks, J. A., Berger, D. H. & Albo, D. Perineural invasion in cancer: a review of the literature. Cancer 115, 3379–3391 (2009).
Demir, I. E. et al. Neural invasion in pancreatic cancer: the past, present and future. Cancers 2, 1513–1527 (2010).
Batsakis, J. G. Nerves and neurotropic carcinomas. Ann. Otol. Rhinol. Laryngol. 94, 426–427 (1985).
Bockman, D. E., Buchler, M. & Beger, H. G. Interaction of pancreatic ductal carcinoma with nerves leads to nerve damage. Gastroenterology 107, 219–230 (1994).
Ceyhan, G. O. et al. Neural invasion in pancreatic cancer: a mutual tropism between neurons and cancer cells. Biochem. Biophys. Res. Commun. 374, 442–447 (2008).
Ceyhan, G. O. et al. The neurotrophic factor artemin promotes pancreatic cancer invasion. Ann. Surg. 244, 274–281 (2006).
Rodin, A. E., Larson, D. L. & Roberts, D. K. Nature of the perineural space invaded by prostatic carcinoma. Cancer 20, 1772–1779 (1967).
Marchesi, F., Piemonti, L., Mantovani, A. & Allavena, P. Molecular mechanisms of perineural invasion, a forgotten pathway of dissemination and metastasis. Cytokine Growth Factor Rev. 21, 77–82 (2010).
Ceyhan, G. O., Michalski, C. W., Demir, I. E., Muller, M. W. & Friess, H. Pancreatic pain. Best Pract. Res. Clin. Gastroenterol. 22, 31–44 (2008).
Hirai, I. et al. Perineural invasion in pancreatic cancer. Pancreas 24, 15–25 (2002).
Liu, B. & Lu, K. Y. Neural invasion in pancreatic carcinoma. Hepatobiliary Pancreat. Dis. Int. 1, 469–476 (2002).
Pour, P. M., Bell, R. H. & Batra, S. K. Neural invasion in the staging of pancreatic cancer. Pancreas 26, 322–325 (2003).
Ayala, G. E. et al. Growth and survival mechanisms associated with perineural invasion in prostate cancer. Cancer Res. 64, 6082–6090 (2004).
Ayala, G. E. et al. In vitro dorsal root ganglia and human prostate cell line interaction: redefining perineural invasion in prostate cancer. Prostate 49, 213–223 (2001). This article describes the co-culture of DRGs and cancer cells as an experimental model for studying PNI. The study demonstrated reciprocal signalling between prostate cancer cells and neurites.
Maru, N., Ohori, M., Kattan, M. W., Scardino, P. T. & Wheeler, T. M. Prognostic significance of the diameter of perineural invasion in radical prostatectomy specimens. Hum. Pathol. 32, 828–833 (2001).
Horn, A., Dahl, O. & Morild, I. Venous and neural invasion as predictors of recurrence in rectal adenocarcinoma. Dis. Colon Rectum 34, 798–804 (1991).
Huh, J. W., Kim, H. R. & Kim, Y. J. Prognostic value of perineural invasion in patients with stage II colorectal cancer. Ann. Surg. Oncol. 17, 2066–2072 (2010).
Pages, F. et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N. Engl. J. Med. 353, 2654–2666 (2005).
Cowan, W. K. et al. The pathological and biological nature of screen-detected breast carcinomas: a morphological and immunohistochemical study. J. Pathol. 182, 29–35 (1997).
Dunn, M. & Morgan, M. B. Perineural invasion progressing to leptomeningeal carcinomatosis: is the absence of peripheral nerves an important sign? J. Am. Acad. Dermatol. 62, 270–276 (2010).
Duraker, N., Sisman, S. & Can, G. The significance of perineural invasion as a prognostic factor in patients with gastric carcinoma. Surg. Today 33, 95–100 (2003).
Haddad, R. I. & Shin, D. M. Recent advances in head and neck cancer. N. Engl. J. Med. 359, 1143–1154 (2008).
Kurtz, K. A., Hoffman, H. T., Zimmerman, M. B. & Robinson, R. A. Perineural and vascular invasion in oral cavity squamous carcinoma: increased incidence on re-review of slides and by using immunohistochemical enhancement. Arch. Pathol. Lab. Med. 129, 354–359 (2005).
Scartozzi, M. et al. Lymphatic, blood vessel and perineural invasion identifies early-stage high-risk radically resected gastric cancer patients. Br. J. Cancer 95, 445–449 (2006).
Shirai, K. et al. Perineural invasion is a prognostic factor in intrahepatic cholangiocarcinoma. World J. Surg. 32, 2395–2402 (2008).
Su, C. H. et al. Factors influencing postoperative morbidity, mortality, and survival after resection for hilar cholangiocarcinoma. Ann. Surg. 223, 384–394 (1996).
Takahashi, T. et al. Perineural invasion by ductal adenocarcinoma of the pancreas. J. Surg. Oncol. 65, 164–170 (1997).
Ceyhan, G. O. et al. Nerve growth factor and artemin are paracrine mediators of pancreatic neuropathy in pancreatic adenocarcinoma. Ann. Surg. 251, 923–931 (2010).
Stolinski, C. Structure and composition of the outer connective tissue sheaths of peripheral nerve. J. Anat. 186, 123–130 (1995).
Badger, S. A. et al. The role of surgery for pancreatic cancer: a 12-year review of patient outcome. Ulster Med. J. 79, 70–75 (2010).
Chen, J. W. et al. Predicting patient survival after pancreaticoduodenectomy for malignancy: histopathological criteria based on perineural infiltration and lymphovascular invasion. HPB (Oxford) 12, 101–108 (2010).
Garcea, G. et al. Survival following curative resection for pancreatic ductal adenocarcinoma. A systematic review of the literature. JOP 9, 99–132 (2008).
Meduri, F. et al. Pancreatic cancer and retroperitoneal neural tissue invasion. Its implication for survival following radical surgery. Zentralbl. Pathol. 140, 277–279 (1994).
Mossner, J. What's new in therapy of pancreatic cancer? Dig. Dis. 28, 679–683 (2010).
Perini, M. V. et al. Clinical and pathologic prognostic factors for curative resection for pancreatic cancer. HPB (Oxford) 10, 356–362 (2008).
Dai, H. et al. Enhanced survival in perineural invasion of pancreatic cancer: an in vitro approach. Hum. Pathol. 38, 299–307 (2007).
Gil, Z. et al. Paracrine regulation of pancreatic cancer cell invasion by peripheral nerves. J. Natl Cancer Inst. 102, 107–118 (2010).
Ketterer, K. et al. Reverse transcription-PCR analysis of laser-captured cells points to potential paracrine and autocrine actions of neurotrophins in pancreatic cancer. Clin. Cancer Res. 9, 5127–5136 (2003).
Zhu, Z. et al. Nerve growth factor expression correlates with perineural invasion and pain in human pancreatic cancer. J. Clin. Oncol. 17, 2419–2428 (1999). The first study to demonstrate the involvement of the NGF signalling pathway in PNI and pain generation in pancreatic cancer.
Okada, Y., Eibl, G., Duffy, J. P., Reber, H. A. & Hines, O. J. Glial cell-derived neurotrophic factor upregulates the expression and activation of matrix metalloproteinase-9 in human pancreatic cancer. Surgery 134, 293–299 (2003).
Okada, Y. et al. Nerve growth factor stimulates MMP-2 expression and activity and increases invasion by human pancreatic cancer cells. Clin. Exp. Metastasis 21, 285–292 (2004).
Marchesi, F. et al. The chemokine receptor CX3CR1 is involved in the neural tropism and malignant behavior of pancreatic ductal adenocarcinoma. Cancer Res. 68, 9060–9069 (2008). This study showed for the first time that CX3CR1 is involved in PNI in pancreatic cancer.
Singh, P. K. et al. Platelet-derived growth factor receptor β-mediated phosphorylation of MUC1 enhances invasiveness in pancreatic adenocarcinoma cells. Cancer Res. 67, 5201–5210 (2007).
Swanson, B. J. et al. MUC1 is a counter-receptor for myelin-associated glycoprotein (Siglec-4a) and their interaction contributes to adhesion in pancreatic cancer perineural invasion. Cancer Res. 67, 10222–10229 (2007).
Miknyoczki, S. J. et al. Neurotrophins and Trk receptors in human pancreatic ductal adenocarcinoma: expression patterns and effects on in vitro invasive behavior. Int. J. Cancer 81, 417–427 (1999).
Schneider, M. B. et al. Expression of nerve growth factors in pancreatic neural tissue and pancreatic cancer. J. Histochem. Cytochem. 49, 1205–1210 (2001).
Reichardt, L. F. Neurotrophin-regulated signalling pathways. Phil. Trans. R. Soc. B 361, 1545–1564 (2006).
Arevalo, J. C. & Wu, S. H. Neurotrophin signaling: many exciting surprises! Cell. Mol. Life Sci. 63, 1523–1537 (2006).
Schweigreiter, R. The dual nature of neurotrophins. Bioessays 28, 583–594 (2006).
Patapoutian, A. & Reichardt, L. F. Trk receptors: mediators of neurotrophin action. Curr. Opin. Neurobiol. 11, 272–280 (2001).
Barker, P. A. p75NTR: a study in contrasts. Cell Death Differ. 5, 346–56 (1998).
Chao, M. V. The p75 neurotrophin receptor. J. Neurobiol. 25, 1373–1385 (1994).
Hempstead, B. L. The many faces of p75NTR. Curr. Opin. Neurobiol. 12, 260–267 (2002).
Cornell, R. J., Rowley, D., Wheeler, T., Ali, N. & Ayala, G. Neuroepithelial interactions in prostate cancer are enhanced in the presence of prostatic stroma. Urology 61, 870–875 (2003).
Malin, S. A., Davis, B. M. & Molliver, D. C. Production of dissociated sensory neuron cultures and considerations for their use in studying neuronal function and plasticity. Nature Protoc. 2, 152–160 (2007).
Tonge, D. A. et al. Effects of extracellular matrix components on axonal outgrowth from peripheral nerves of adult animals in vitro. Exp. Neurol. 146, 81–90 (1997).
Klesse, L. J., Meyers, K. A., Marshall, C. J. & Parada, L. F. Nerve growth factor induces survival and differentiation through two distinct signaling cascades in PC12 cells. Oncogene 18, 2055–2068 (1999).
Zhu, Z. et al. Nerve growth factor and enhancement of proliferation, invasion, and tumorigenicity of pancreatic cancer cells. Mol. Carcinog. 35, 138–147 (2002).
Ma, J., Jiang, Y., Sun, Y. & Zhao, X. Expression of nerve growth factor and tyrosine kinase receptor A and correlation with perineural invasion in pancreatic cancer. J. Gastroenterol. Hepatol. 23, 1852–1859 (2008).
Miknyoczki, S. J. et al. The neurotrophin-trk receptor axes are critical for the growth and progression of human prostatic carcinoma and pancreatic ductal adenocarcinoma xenografts in nude mice. Clin. Cancer Res. 8, 1924–1931 (2002).
Zhu, Z. W. et al. Nerve growth factor exerts differential effects on the growth of human pancreatic cancer cells. Clin. Cancer Res. 7, 105–112 (2001).
Friess, H. et al. Nerve growth factor and its high-affinity receptor in chronic pancreatitis. Ann. Surg. 230, 615–624 (1999).
Dang, C., Zhang, Y., Ma, Q. & Shimahara, Y. Expression of nerve growth factor receptors is correlated with progression and prognosis of human pancreatic cancer. J. Gastroenterol. Hepatol. 21, 850–858 (2006).
Wang, W. et al. Patterns of expression and function of the p75(NGFR) protein in pancreatic cancer cells and tumours. Eur. J. Surg. Oncol. 35, 826–832 (2009).
Sclabas, G. M. et al. Overexpression of tropomysin-related kinase B in metastatic human pancreatic cancer cells. Clin. Cancer Res. 11, 440–449 (2005).
Airaksinen, M. S. & Saarma, M. The GDNF family: signalling, biological functions and therapeutic value. Nature Rev. Neurosci. 3, 383–394 (2002).
Takahashi, M. The GDNF/RET signaling pathway and human diseases. Cytokine Growth Factor Rev. 12, 361–373 (2001).
Ito, Y. et al. Expression of glial cell line-derived neurotrophic factor family members and their receptors in pancreatic cancers. Surgery 138, 788–794 (2005).
Balkwill, F. Cancer and the chemokine network. Nature Rev. Cancer 4, 540–550 (2004).
Hedin, K. E. Chemokines: new, key players in the pathobiology of pancreatic cancer. Int. J. Gastrointest. Cancer 31, 23–29 (2002).
Marchesi, F. et al. Role of CX3CR1/CX3CL1 axis in primary and secondary involvement of the nervous system by cancer. J. Neuroimmunol. 224, 39–44 (2010).
Verge, G. M. et al. Fractalkine (CX3CL1) and fractalkine receptor (CX3CR1) distribution in spinal cord and dorsal root ganglia under basal and neuropathic pain conditions. Eur. J. Neurosci. 20, 1150–1160 (2004).
Muller, M. W. et al. Association of axon guidance factor semaphorin 3A with poor outcome in pancreatic cancer. Int. J. Cancer 121, 2421–2433 (2007).
Bloomston, M., Zervos, E. E. & Rosemurgy, A. S. Matrix metalloproteinases and their role in pancreatic cancer: a review of preclinical studies and clinical trials. Ann. Surg. Oncol. 9, 668–674 (2002).
Takahashi, H. et al. Antiproteases in preventing the invasive potential of pancreatic cancer cells. JOP 8, 501–508 (2007).
Pryczynicz, A., Guzinska-Ustymowicz, K., Dymicka-Piekarska, V., Czyzewska, J. & Kemona, A. Expression of matrix metalloproteinase 9 in pancreatic ductal carcinoma is associated with tumor metastasis formation. Folia Histochem. Cytobiol. 45, 37–40 (2007).
Zhi, Y. H., Song, M. M., Wang, P. L., Zhang, T. & Yin, Z. Y. Suppression of matrix metalloproteinase-2 via RNA interference inhibits pancreatic carcinoma cell invasiveness and adhesion. World J. Gastroenterol. 15, 1072–1078 (2009).
Okada, Y. et al. Experimental implication of celiac ganglionotropic invasion of pancreatic-cancer cells bearing c-ret proto-oncogene with reference to glial-cell-line-derived neurotrophic factor (GDNF). Int. J. Cancer 81, 67–73 (1999).
Koide, N. et al. Establishment of perineural invasion models and analysis of gene expression revealed an invariant chain (CD74) as a possible molecule involved in perineural invasion in pancreatic cancer. Clin. Cancer Res. 12, 2419–2426 (2006).
Hustinx, S. R. et al. Differentially expressed genes in pancreatic ductal adenocarcinomas identified through serial analysis of gene expression. Cancer Biol. Ther. 3, 1254–1261 (2004).
Nagata, S. et al. CD74 is a novel prognostic factor for patients with pancreatic cancer receiving multimodal therapy. Ann. Surg. Oncol. 16, 2531–2538 (2009).
Abiatari, I. et al. Consensus transcriptome signature of perineural invasion in pancreatic carcinoma. Mol. Cancer Ther. 8, 1494–1504 (2009). This study establishes a new ex vivo co-culture model for PNI that allows the study of the differences between highly nerve-invasive and non-invasive pancreatic cancer cells.
Corson, T. W., Huang, A., Tsao, M. S. & Gallie, B. L. KIF14 is a candidate oncogene in the 1q minimal region of genomic gain in multiple cancers. Oncogene 24, 4741–4753 (2005).
Carleton, M. et al. RNA interference-mediated silencing of mitotic kinesin KIF14 disrupts cell cycle progression and induces cytokinesis failure. Mol. Cell. Biol. 26, 3853–3863 (2006).
Harding, M. A. & Theodorescu, D. RhoGDI signaling provides targets for cancer therapy. Eur. J. Cancer 46, 1252–1259 (2010).
Zhang, B., Zhang, Y., Dagher, M. C. & Shacter, E. Rho GDP dissociation inhibitor protects cancer cells against drug-induced apoptosis. Cancer Res. 65, 6054–6062 (2005).
Zhang, Y. & Zhang, B. D4-GDI, a Rho GTPase regulator, promotes breast cancer cell invasiveness. Cancer Res. 66, 5592–5598 (2006).
Li, Z. et al. Overexpression of synuclein-γ in pancreatic adenocarcinoma. Cancer 101, 58–65 (2004).
Hibi, T. et al. Synuclein-γ is closely involved in perineural invasion and distant metastasis in mouse models and is a novel prognostic factor in pancreatic cancer. Clin. Cancer Res. 15, 2864–2871 (2009).
Ahmad, M., Attoub, S., Singh, M. N., Martin, F. L. & El-Agnaf, O. M. γ-synuclein and the progression of cancer. FASEB J. 21, 3419–3430 (2007).
Trapp, B. D., Andrews, S. B., Cootauco, C. & Quarles, R. The myelin-associated glycoprotein is enriched in multivesicular bodies and periaxonal membranes of actively myelinating oligodendrocytes. J. Cell Biol. 109, 2417–2426 (1989).
Kameda, K. et al. Expression of highly polysialylated neural cell adhesion molecule in pancreatic cancer neural invasive lesion. Cancer Lett. 137, 201–207 (1999).
Schreiber, S. C. et al. Polysialylated NCAM represses E-cadherin-mediated cell-cell adhesion in pancreatic tumor cells. Gastroenterology 134, 1555–1566 (2008).
Tezel, E., Kawase, Y., Takeda, S., Oshima, K. & Nakao, A. Expression of neural cell adhesion molecule in pancreatic cancer. Pancreas 22, 122–125 (2001).
Ben, Q. W. et al. Positive expression of L1-CAM is associated with perineural invasion and poor outcome in pancreatic ductal adenocarcinoma. Ann. Surg. Oncol. 17, 2213–2221 (2010).
Demir, I. E. et al. The microenvironment in chronic pancreatitis and pancreatic cancer induces neuronal plasticity. Neurogastroenterol. Motil. 22, 480–490, e112–e113 (2010).
Lussier, D., Huskey, A. G. & Portenoy, R. K. Adjuvant analgesics in cancer pain management. Oncologist 9, 571–591 (2004).
Mantyh, P. W., Clohisy, D. R., Koltzenburg, M. & Hunt, S. P. Molecular mechanisms of cancer pain. Nature Rev. Cancer 2, 201–209 (2002).
de Leon-Casasola, O. A. Critical evaluation of chemical neurolysis of the sympathetic axis for cancer pain. Cancer Control 7, 142–148 (2000). An overview of the different types of neurolytic blocks available and the pros and cons that are associated with them.
Kaufman, M. et al. Efficacy of endoscopic ultrasound-guided celiac plexus block and celiac plexus neurolysis for managing abdominal pain associated with chronic pancreatitis and pancreatic cancer. J. Clin. Gastroenterol. 44, 127–134 (2010).
Vranken, J. H., Zuurmond, W. W. & de Lange, J. J. Increasing the efficacy of a celiac plexus block in patients with severe pancreatic cancer pain. J. Pain Symptom Manage. 22, 966–977 (2001).
Yang, I. Y. & Oraee, S. A modified approach to transcrural celiac plexus block. Reg. Anesth. Pain Med. 30, 303–307 (2005).
di Mola, F. F. & di Sebastiano, P. Pain and pain generation in pancreatic cancer. Langenbecks Arch. Surg. 393, 919–922 (2008).
Lindsay, T. H. et al. Pancreatic cancer pain and its correlation with changes in tumor vasculature, macrophage infiltration, neuronal innervation, body weight and disease progression. Pain 119, 233–246 (2005).
Zhu, Y. et al. Nerve growth factor modulates TRPV1 expression and function and mediates pain in chronic pancreatitis. Gastroenterology 141, 370–377 (2011).
Nilius, B., Owsianik, G., Voets, T. & Peters, J. A. Transient receptor potential cation channels in disease. Physiol. Rev. 87, 165–217 (2007).
Jara-Oseguera, A., Simon, S. A. & Rosenbaum, T. TRPV1: on the road to pain relief. Curr. Mol. Pharmacol. 1, 255–269 (2008).
Hartel, M. et al. Vanilloids in pancreatic cancer: potential for chemotherapy and pain management. Gut 55, 519–528 (2006). The first paper to report that TRPV1 expression correlates with the severity of pain suffered by patients with pancreatic cancer; patients whose tumours had more TRPV1-positive nerves infiltrated by pancreatic cancer cells had high pain scores.
Pingle, S. C., Matta, J. A. & Ahern, G. P. Capsaicin receptor: TRPV1 a promiscuous TRP channel. Handb. Exp. Pharmacol. 179, 155–171 (2007).
Anand, U. et al. The effect of neurotrophic factors on morphology, TRPV1 expression and capsaicin responses of cultured human DRG sensory neurons. Neurosci. Lett. 399, 51–56 (2006).
Liddle, R. A. The role of transient receptor potential vanilloid 1 (TRPV1) channels in pancreatitis. Biochim. Biophys. Acta 1772, 869–878 (2007).
Amaya, F. et al. NGF and GDNF differentially regulate TRPV1 expression that contributes to development of inflammatory thermal hyperalgesia. Eur. J. Neurosci. 20, 2303–2310 (2004).
Malin, S. A. et al. Glial cell line-derived neurotrophic factor family members sensitize nociceptors in vitro and produce thermal hyperalgesia in vivo. J. Neurosci. 26, 8588–8599 (2006).
Schweizerhof, M. et al. Hematopoietic colony-stimulating factors mediate tumor-nerve interactions and bone cancer pain. Nature Med. 15, 802–807 (2009).
Stosser, S., Schweizerhof, M. & Kuner, R. Hematopoietic colony-stimulating factors: new players in tumor-nerve interactions. J. Mol. Med. 89, 321–329 (2011).
Abdiche, Y. N., Malashock, D. S. & Pons, J. Probing the binding mechanism and affinity of tanezumab, a recombinant humanized anti-NGF monoclonal antibody, using a repertoire of biosensors. Protein Sci. 17, 1326–1335 (2008).
Hefti, F. F. et al. Novel class of pain drugs based on antagonism of NGF. Trends Pharmacol. Sci. 27, 85–91 (2006).
Lane, N. E. et al. Tanezumab for the treatment of pain from osteoarthritis of the knee. N. Engl. J. Med. 363, 1521–1531 (2010).
Watson, J. J., Allen, S. J. & Dawbarn, D. Targeting nerve growth factor in pain: what is the therapeutic potential? BioDrugs 22, 349–359 (2008).
Wood, J. N. Nerve growth factor and pain. N. Engl. J. Med. 363, 1572–1573 (2010).
Cattaneo, A. et al. Functional blockade of tyrosine kinase A in the rat basal forebrain by a novel antagonistic anti-receptor monoclonal antibody. J. Neurosci. 19, 9687–9697 (1999).
Covaceuszach, S., Cattaneo, A. & Lamba, D. Neutralization of NGF-TrkA receptor interaction by the novel antagonistic anti-TrkA monoclonal antibody MNAC13: a structural insight. Proteins 58, 717–727 (2005).
Ugolini, G., Marinelli, S., Covaceuszach, S., Cattaneo, A. & Pavone, F. The function neutralizing anti-TrkA antibody MNAC13 reduces inflammatory and neuropathic pain. Proc. Natl Acad. Sci. USA 104, 2985–2990 (2007).
Wehrman, T. et al. Structural and mechanistic insights into nerve growth factor interactions with the TrkA and p75 receptors. Neuron 53, 25–38 (2007).
Watson, J. J. et al. TrkAd5: a novel therapeutic agent for treatment of inflammatory pain and asthma. J. Pharmacol. Exp. Ther. 316, 1122–1129 (2006).
Dawbarn, D. et al. NGF receptor TrkAd5: therapeutic agent and drug design target. Biochem. Soc. Trans. 34, 587–590 (2006).
Sutherland, S. Peptibodies: the new cool technology. Drug Discov. Today 9, 683 (2004).
Wang, T., Yu, D. & Lamb, M. L. Trk kinase inhibitors as new treatments for cancer and pain. Expert Opin. Ther. Pat. 19, 305–319 (2009). An extensive review of small-molecule TRK kinase inhibitors that are being developed by pharmaceutical companies for the treatment of cancer and associated pain.
Wood, E. R. et al. Discovery and in vitro evaluation of potent TrkA kinase inhibitors: oxindole and aza-oxindoles. Bioorg. Med. Chem. Lett. 14, 953–957 (2004).
Ghilardi, J. R. et al. Sustained blockade of neurotrophin receptors TrkA, TrkB and TrkC reduces non-malignant skeletal pain but not the maintenance of sensory and sympathetic nerve fibers. Bone 48, 389–398 (2011).
Tibes, R. et al. Phase I dose escalation study of the oral multi-CDK inhibitor PHA-848125. J. Clin. Oncol. 26 (Suppl.), Abstract 3531 (2008).
Brasca, M. G. et al. Identification of N-1,4,4-tetramethyl-8-{[4-(4-methylpiperazin-1-yl)phenyl]amino}-4,5-dihydr o-1H-pyrazolo[4,3-h]quinazoline-3-carboxamide (PHA-848125), a potent, orally available cyclin dependent kinase inhibitor. J. Med. Chem. 52, 5152–5163 (2009).
Caporali, S. et al. The cyclin-dependent kinase inhibitor PHA-848125 suppresses the in vitro growth of human melanomas sensitive or resistant to temozolomide, and shows synergistic effects in combination with this triazene compound. Pharmacol. Res. 61, 437–448 (2010).
Degrassi, A. et al. Efficacy of PHA-848125, a cyclin-dependent kinase inhibitor, on the K-Ras(G12D)LA2 lung adenocarcinoma transgenic mouse model: evaluation by multimodality imaging. Mol. Cancer Ther. 9, 673–681 (2010).
Albanese, C. et al. Dual targeting of CDK and tropomyosin receptor kinase families by the oral inhibitor PHA-848125, an agent with broad-spectrum antitumor efficacy. Mol. Cancer Ther. 9, 2243–2254 (2010).
Premkumar, L. S. Targeting TRPV1 as an alternative approach to narcotic analgesics to treat chronic pain conditions. AAPS J. 12, 361–370 (2010).
Wong, G. Y. & Gavva, N. R. Therapeutic potential of vanilloid receptor TRPV1 agonists and antagonists as analgesics: recent advances and setbacks. Brain Res. Rev. 60, 267–277 (2009). An extensive review of TRPV1-targeted molecules and their activities and clinical development statuses.
Ghilardi, J. R. et al. Selective blockade of the capsaicin receptor TRPV1 attenuates bone cancer pain. J. Neurosci. 25, 3126–3131 (2005).
Dorgham, K. et al. An engineered CX3CR1 antagonist endowed with anti-inflammatory activity. J. Leukoc. Biol. 86, 903–911 (2009).
Yin, Q., Cheng, W., Cheng, M. Y., Fan, S. Z. & Shen, W. Intrathecal injection of anti-CX3CR1 neutralizing antibody delayed and attenuated pain facilitation in rat tibial bone cancer pain model. Behav. Pharmacol. 21, 595–601 (2010).
Cameron, J. L. et al. Factors influencing survival after pancreaticoduodenectomy for pancreatic cancer. Am. J. Surg. 161, 120–124 (1991).
Jemal, A., Siegel, R., Xu, J. & Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 60, 277–300 (2010).
Hezel, A. F., Kimmelman, A. C., Stanger, B. Z., Bardeesy, N. & Depinho, R. A. Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 20, 1218–1249 (2006).
Kayahara, M. et al. An evaluation of radical resection for pancreatic cancer based on the mode of recurrence as determined by autopsy and diagnostic imaging. Cancer 72, 2118–2123 (1993).
Shimada, K. et al. Intrapancreatic nerve invasion as a predictor for recurrence after pancreaticoduodenectomy in patients with invasive ductal carcinoma of the pancreas. Pancreas 40, 464–468 (2011).
Samkharadze, T. et al. Pigment epithelium-derived factor associates with neuropathy and fibrosis in pancreatic cancer. Am. J. Gastroenterol. 106, 968–980 (2011).
Guerra, C. et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 11, 291–302 (2007).
Hingorani, S. R. et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell 4, 437–450 (2003).
Freelove, R. & Walling, A. D. Pancreatic cancer: diagnosis and management. Am. Fam. Physician 73, 485–492 (2006).
Kang, S. P. & Saif, M. W. Optimal second line treatment options for gemcitabine refractory advanced pancreatic cancer patients. Can we establish standard of care with available data? JOP 9, 83–90 (2008).
Burris, H. A. et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J. Clin. Oncol. 15, 2403–2413 (1997).
Campen, C. J., Dragovich, T. & Baker, A. F. Management strategies in pancreatic cancer. Am. J. Health Syst. Pharm. 68, 573–584 (2011).
Di Marco, M. et al. Metastatic pancreatic cancer: is gemcitabine still the best standard treatment? Oncol. Rep. 23, 1183–1192 (2010).
Fung, M. C. & Sakata, T. What's new in pancreatic cancer treatment? J. Hepatobiliary Pancreat. Surg. 9, 61–75 (2002).
Li, J. & Saif, M. W. Advancements in the management of pancreatic cancer. JOP 10, 109–117 (2009).
Sharma, C. et al. Adjuvant therapy of pancreatic cancer. Highlights from the “2011 ASCO Annual Meeting”. Chicago, IL, USA; June 3–4, 2011. JOP 12, 343–346 (2011).
Helm, J. et al. Histologic characteristics enhance predictive value of American Joint Committee on Cancer staging in resectable pancreas cancer. Cancer 115, 4080–4089 (2009).
Pawlik, T. M. et al. Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery 141, 610–618 (2007).
Washington, K. et al. Protocol for the examination of specimens from patients with carcinoma of the exocrine pancreas: protocol applies to all epithelial tumors of the exocrine pancreas. endocrine tumors and tumors of the ampulla of vater are not included. College of American Pathologists [online], (2009).
Acknowledgements
This work is dedicated to S. Greene and S. Salmon, who taught us just how tough perineural invasion by pancreatic cancer can be. We would also like to thank C. Nulsen for her invaluable and critical insights during the preparation of this manuscript. Research in the authors' laboratories was supported by grants from the US National Institutes of Health National Cancer Institute (grants CA140924 and CA109552), the American Association for Cancer Research Stand Up to Cancer programme and the US National Foundation for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Perineurium
-
The connective tissue sheath that surrounds bundles of nerves known as fascicles.
- Dorsal root ganglia
-
A mass of sensory afferent nerve cell bodies that is located on the dorsal root of the spinal cord, one on each side for each spinal nerve.
- Neurolytic blocks
-
A process in which a neurolytic or analgesic agent is injected into or near nerves that are involved in pain signalling. Neurolytic blocks are used to combat chronic pain states or pain that is caused by cancer.
- Coeliac plexus blocks
-
A process in which the nerves of the coeliac plexus are subjected to neurolysis using neurolytic agents, such as a 50–100% solution of alcohol or a 10% solution of phenol, that are injected into the coeliac plexus guided by ultrasound and computed tomography (CT) imaging.
- Thermal hyperalgesia
-
Increased pain responses following an increase in temperature.
- Tactile allodynia
-
Pain sensations caused by mechanical stimuli such as touch that usually do not invoke pain responses.
Rights and permissions
About this article
Cite this article
Bapat, A., Hostetter, G., Von Hoff, D. et al. Perineural invasion and associated pain in pancreatic cancer. Nat Rev Cancer 11, 695–707 (2011). https://doi.org/10.1038/nrc3131
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3131
This article is cited by
-
The preliminary analysis of lymphatic flow around the connective tissues surrounding SMA and SpA elucidates patients’ oncological condition in borderline-resectable pancreatic cancer
BMC Surgery (2024)
-
Exosomal lncRNA XIST promotes perineural invasion of pancreatic cancer cells via miR-211-5p/GDNF
Oncogene (2024)
-
Predicting response to immunotherapy in gastric cancer via assessing perineural invasion-mediated inflammation in tumor microenvironment
Journal of Experimental & Clinical Cancer Research (2023)
-
Cancer–nerve interplay in cancer progression and cancer-induced bone pain
Journal of Bone and Mineral Metabolism (2023)
-
Sonic Hedgehog Signaling Pathway: A Role in Pain Processing
Neurochemical Research (2023)