Abstract
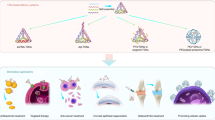
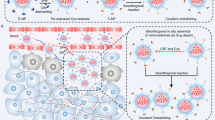
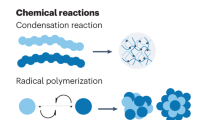
The assembly of nanomaterials using DNA can produce complex nanostructures, but the biological applications of these structures remain unexplored. Here, we describe the use of DNA to control the biological delivery and elimination of inorganic nanoparticles by organizing them into colloidal superstructures. The individual nanoparticles serve as building blocks, whose size, surface chemistry and assembly architecture dictate the overall superstructure design. These superstructures interact with cells and tissues as a function of their design, but subsequently degrade into building blocks that can escape biological sequestration. We demonstrate that this strategy reduces nanoparticle retention by macrophages and improves their in vivo tumour accumulation and whole-body elimination. Superstructures can be further functionalized to carry and protect imaging or therapeutic agents against enzymatic degradation. These results suggest a different strategy to engineer nanostructure interactions with biological systems and highlight new directions in the design of biodegradable and multifunctional nanomedicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kim, J., Piao, Y. & Hyeon, T. Multifunctional nanostructured materials for multimodal imaging, and simultaneous imaging and therapy. Chem. Soc. Rev. 38, 372–390 (2009).
Giljohann, D. A. et al. Gold nanoparticles for biology and medicine. Angew. Chem. Int. Ed. 49, 3280–3294 (2010).
Gao, J., Gu, H. & Xu, B. Multifunctional magnetic nanoparticles: design, synthesis, and biomedical applications. Acc. Chem. Res. 42, 1097–1107 (2009).
Smith, A. M., Duan, H., Mohs, A. M. & Nie, S. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv. Drug Deliv. Rev. 60, 1226–1240 (2008).
Jiang, W., Kim, B. Y. S., Rutka, J. T. & Chan, W. C. W. Nanoparticle-mediated cellular response is size-dependent. Nature Nanotech. 3, 145–150 (2008).
Albanese, A., Tang, P. S. & Chan, W. C. W. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu. Rev. Biomed. Eng. 14, 1–16 (2012).
Petros, R. A. & DeSimone, J. M. Strategies in the design of nanoparticles for therapeutic applications. Nature Rev. Drug Discov. 9, 615–627 (2010).
Davis, M. E., Chen, Z. & Shin, D. M. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nature Rev. Drug Discov. 7, 771–782 (2008).
Perrault, S. D., Walkey, C., Jennings, T., Fischer, H. C. & Chan, W. C. W. Mediating tumor targeting efficiency of nanoparticles through design. Nano Lett. 9, 1909–1915 (2009).
Robinson, J. T. et al. In vivo fluorescence imaging in the second near-infrared window with long circulating carbon nanotubes capable of ultrahigh tumor uptake. J. Am. Chem. Soc. 134, 10664–10669 (2012).
Park, J. H. et al. Cooperative nanomaterial system to sensitize, target, and treat tumors. Proc. Natl Acad. Sci. USA 107, 981–986 (2010).
Choi, H. S. et al. Renal clearance of quantum dots. Nature Biotechnol. 25, 1165–1170 (2007).
Zhou, C., Long, M., Qin, Y., Sun, X. & Zheng, J. Luminescent gold nanoparticles with efficient renal clearance. Angew. Chem. Int. Ed. 50, 3168–3172 (2011).
Ballou, B. et al. Sentinel lymph node imaging using quantum dots in mouse tumor models. Bioconjug. Chem. 18, 389–396 (2007).
Albanese, A. & Chan, W. C. W. Effect of gold nanoparticle aggregation on cell uptake and toxicity. ACS Nano 5, 5478–5489 (2011).
Díaz, B. et al. Assessing methods for blood cell cytotoxic responses to inorganic nanoparticles and nanoparticle aggregates. Small 4, 2025–2034 (2008).
Lovrić, J., Cho, S. J., Winnik, F. M. & Maysinger, D. Unmodified cadmium telluride quantum dots induce reactive oxygen species formation leading to multiple organelle damage and cell death. Chem. Biol. 12, 1227–1234 (2005).
Derfus, A. M., Chan, W. C. W. & Bhatia, S. N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 4, 11–18 (2004).
Yang, R. S. H. et al. Persistent tissue kinetics and redistribution of nanoparticles, quantum dot 705, in mice: ICP-MS quantitative assessment. Environ. Health Perspect. 115, 1339–1343 (2007).
Kim, J. S. et al. Toxicity and tissue distribution of magnetic nanoparticles in mice. Toxicol. Sci. 89, 338–347 (2006).
Ye, L. et al. A pilot study in non-human primates shows no adverse response to intravenous injection of quantum dots. Nature Nanotech. 7, 453–458 (2012).
Choi, H. S. et al. Design considerations for tumour-targeted nanoparticles. Nature Nanotech. 5, 42–47 (2010).
Yoon, J., Lim, J. & Yoon, S. Controlled assembly and plasmonic properties of asymmetric core-satellite nanoassemblies. ACS Nano 6, 7199–7208 (2012).
Xu, X., Rosi, N. L., Wang, Y., Huo, F. & Mirkin, C. A. Asymmetric functionalization of gold nanoparticles with oligonucleotides. J. Am. Chem. Soc. 128, 9286–9287 (2006).
Knop, K., Hoogenboom, R., Fischer, D. & Schubert, U. S. Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew. Chem. Int. Ed. 49, 6288–6308 (2010).
Chaires, J., Herrera, J. & Waring, M. Preferential binding of daunomycin to 5′TACG and 5′TAGC sequences revealed by footprinting titration experiments. Biochemistry 29, 6145–6153 (1990).
Owens, D. E. & Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Khlebtsov, N. & Dykman, L. Biodistribution and toxicity of engineered gold nanoparticles: a review of in vitro and in vivo studies. Chem. Soc. Rev. 40, 1647–1671 (2011).
Clift, M. J. D. et al. The impact of different nanoparticle surface chemistry and size on uptake and toxicity in a murine macrophage cell line. Toxicol. Appl. Pharmacol. 232, 418–427 (2008).
He, C., Hu, Y., Yin, L., Tang, C. & Yin, C. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 31, 3657–3666 (2010).
Patel, P. C. et al. Scavenger receptors mediate cellular uptake of polyvalent oligonucleotide-functionalized gold nanoparticles. Bioconjug. Chem. 21, 2250–2256 (2010).
Claus, V. et al. Lysosomal enzyme trafficking between phagosomes, endosomes, and lysosomes in J774 macrophages. Enrichment of cathepsin H in early endosomes. J. Biol. Chem. 273, 9842–9851 (1998).
Odaka, C. & Mizuochi, T. Role of macrophage lysosomal enzymes in the degradation of nucleosomes of apoptotic cells. J. Immunol. 163, 5346–5352 (1999).
Chou, L. Y. T. & Chan, W. C. W. Fluorescence-tagged gold nanoparticles for rapidly characterizing the size-dependent biodistribution in tumor models. Adv. Healthcare Mater. 1, 714–721 (2012).
Acknowledgements
This research was funded by the Canadian Institute of Health Research (MOP-93532, COP-126588, RMF-111623), the Natural Sciences and Engineering Research Council (NSERC, RGPIN-288231), the Collaborative Health Research Program (CPG-104290, CHRPJ385829), the Canadian Foundation for Innovation and the Ontario Ministry of Research and Innovation. L.Y.T.C. thanks the Canadian Breast Cancer Foundation for a fellowship, and L.Y.T.C. and K.Z. acknowledge NSERC for a fellowship. The authors thank C. Lo and Q. Dai for assistance with animal blood collection, the Advanced Bioimaging Centre at Mt Sinai Hospital, Toronto, Canada, for the use of the TEM, and the ANALEST facility in the Department of Chemistry, University of Toronto, for the use of ICP-AES.
Author information
Authors and Affiliations
Contributions
W.C.W.C., L.Y.T.C. and K.Z. conceived the idea. W.C.W.C and L.Y.T.C. wrote the paper. L.Y.T.C. and K.Z. performed experiments. All authors analysed data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 9056 kb)
Rights and permissions
About this article
Cite this article
Chou, L., Zagorovsky, K. & Chan, W. DNA assembly of nanoparticle superstructures for controlled biological delivery and elimination. Nature Nanotech 9, 148–155 (2014). https://doi.org/10.1038/nnano.2013.309
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2013.309
This article is cited by
-
Preparation, applications, and challenges of functional DNA nanomaterials
Nano Research (2023)
-
Quantification of monodisperse and biocompatible gold nanoparticles by single-particle ICP-MS
Analytical and Bioanalytical Chemistry (2023)
-
Stimuli-responsive DNA-based hydrogels for biosensing applications
Journal of Nanobiotechnology (2022)
-
Comprehensive view of microscopic interactions between DNA-coated colloids
Nature Communications (2022)
-
Core-satellite nanostructures and their biomedical applications
Microchimica Acta (2022)