Abstract
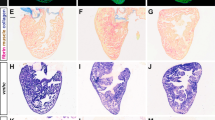
Neurotrophin 3 (Nt3) is one of five neurotrophin growth factors which shape the development of the nervous system by regulating neuronal survival and differentiation. Peripheral neuronal subpopulations expressing the TrkC receptor tyrosine kinase respond to Nt3 with enhanced survival, mitogenesis or cell migration1–3 and these neurons are lost in homozygous Nt3 null (−/−) mutant mice4–7. The unexplained perinatal lethality in the Nt3−/− mice, however, suggests a wider function for this neurotrophin. Here we report that Nt3 is essential for the normal development of atria, ventricles, and cardiac outflow tracts. Histological and echocardiographic image analysis of Nt3−/− animals reveal severe cardiovascular abnormalities including atrial and ventricular septal defects, and tetralogy of Fallot, resembling some of the most common congenital malformations in humans. The observed defects are consistent with abnormalities in the survival and/or migration of cardiac neural crest early in embryogenesis8 and establish an essential role for neurotrophin 3 in regulating the development of the mammalian heart.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Korshing, S. The neurotrophic factor concept: a reexamination. J. Neurosci. 13, 2739–2749 (1993).
Snider, W. Functions of the neurotrophins during nervous system development: what the knockouts are teaching us. Cell 77, 627–638 (1994).
Raffioni, S., Bradshaw, R.A. & Buxer, S.E. The receptors for nerve growth factor and other neurotrophins. Amu. Rev. Biochem. 62, 823–850 (1993).
Emfors, P., Lee, K.-F., Kucera, J. & Jaenisch, R. Lack of neurotrophin-3 leads to deficiencies in the peripheral nervous system and loss of limb proprioceptive afferents. Cell 77, 503–512 (1994).
Farinas, I., Jones, K.R., Backus, C., Wang, X.-Y. & Reichardt, L.F. Severe sensory and sympathetic deficits in mice lacking neurotrophin-3 . Nature 369, 658–661 (1994).
Tessarollo, L., Vogel, K.S., Palko, M.E., Reid, S.W. & Parada, L.F. Targeted mutation in the neurotrophin-3 gene results in loss of muscle sensory neurons. Proc. Natl. Acad. Sci. USA 91, 11844–11848 (1994).
Tojo, H. et al. Targeted disruption of the neurotrophin-3 gene with lacZ induces loss of TrkC-positive neurons in sensory ganglia but not in spinal cord. Brain Res. 669, 163–175 (1995).
Kirby, M.L. & Waldo, M. Role of neural crest in congenital heart disease. Circulation 82, 332–340 (1990).
Tessarolio, L. et al. TrkC, a receptor for neurotrophin-3, is widely expressed in the developing nervous system and in non-neuronal tissues. Development 118, 463–475 (1993).
Tsoulfas, P. et al. The rat TrkC encodes multiple neurogenic receptors that exhibit differential response to Nt3 in PC12 cells. Neuron 10, 975–990 (1993).
Lamballe, F., Klein, R. & Barbacid, M. Developmental expression of TrkC, the neurotrophin-3 receptor in the mammalian nervous system. J. Neurosci. 14, 14–28 (1994).
Donovan, M. et al. Neurotrophins and neurotrophin receptors in vascular smooth muscle cells: regulation of expression in response to injury. Amer. J. Path. 147, 309–324 (1995).
Brill, G., Kahane, N., Carmeli, C., Von Schack, D., Barde, Y.-A. & Kalcheim, C. Epithelial-mesenchymal conversion of dermatome progenitors requires neural tube-derived signals: characterization of the role of Neurotrophin-3 . Development 121, 2583–2594 (1995).
Kahane, N. & Kalcheim, C. Expression of TrkC receptor mRNA during development of the avian nervous system. J. Neurobiol. 25, 571–584 (1994).
Emfors, P., Merlio, J.-R. & Persson, H. Cells expressing mRNA for neurotrophins and their receptors during embryonic rat development. Eur. J. Neurosci. 4, 1140–1158 (1992).
Scarisbrick, A., Jones, E.G. & Isackson, P.J. Coexpression of mRNAs for NGF, BDNF, and Nt3 in the cardiovascular system of the pre- and postnatal rat. J. Neurosci. 13, 875–893 (1993).
Poole, T.J. & Coffin, J.D. Morphogenetic mechanisms in avian vascular development. in The Development of the Vascular System 14 (eds Feinberg, R.N., Sherer, G.K. & Auerbach, R.) 25–36 (Karger Press, Basel, 1991).
Icardo, J.M. & Manasek, F.J. Development mechanisms and embryology. in The Heart and Cardiovascular System, 2nd edn (eds Fozzard, H.A. et al.) 1563–1586 (Raven Press, New York, 1992).
Kirby, M.L., Kumiski, D.H., Myers, T., Cerjan, C., & Mishima, N. Back transplantation of chick cardiac neural crest cells cultured in LIF rescues heart development. Develop. Dyn. 198, 296–311 (1993).
Friedman, W.F. Congenital heart disease in infancy and childhood. in Heart Disease: A Textbook of Cardiovascular Medicine 3rd edn (ed. Braunwald, E.) 895–913 (Saunders Press, Philadelphia, 1988).
Bronner-Fraser, M. Neural crest cell formation and migration in the developing embryo. FASEB J. 8, 699–706 (1994).
Jacks, T., Shin, T.S., Schmitt, E.M., Branson, R.T., Bernards, A. & Weimberg, R.A. Tumour predisposition in mice heterozygous for a targeted mutation in Nf1 . Nature Genet. 7, 353–361 (1994).
Brannan, C.I. et al. Targeted disruption of the neurofibromatosis type-1 gene leads to developmental abnormalities in heart and various neural crest-derived tissues. Genes Dev. 8, 1019–1029 (1994).
Kalcheim, C., Carmeli, C. & Rosenthal, A. Neurotrophin 3 is a mitogen for cultured neural crest cells. Proc. Natl. Acad. Sci. USA 89, 1661–1665 (1992).
Brown, N.A. Routine assesment of morphology and growth: scoring systems and measurement of size. in Postimplantation mammalian embryos: a practical approach. (eds Copp, A.J. & Cockroft, D.L) 930–1108 (IRL/Oxford University Press, New York, 1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Donovan, M., Hahn, R., Tessarollo, L. et al. Identification of an essential nonneuronal function of neurotrophin 3 in mammalian cardiac development. Nat Genet 14, 210–213 (1996). https://doi.org/10.1038/ng1096-210
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1096-210
This article is cited by
-
The role of neurotrophins in psychopathology and cardiovascular diseases: psychosomatic connections
Journal of Neural Transmission (2019)
-
Engineered chromosome-based genetic mapping establishes a 3.7 Mb critical genomic region for Down syndrome-associated heart defects in mice
Human Genetics (2014)
-
Kidins220/ARMS is an essential modulator of cardiovascular and nervous system development
Cell Death & Disease (2011)
-
Optimization of the reconstruction of dermal papilla like tissues employing umbilical cord mesenchymal stem cells
Biotechnology and Bioprocess Engineering (2010)
-
Developing a novel serum-free cell culture model of skeletal muscle differentiation by systematically studying the role of different growth factors in myotube formation
In Vitro Cellular & Developmental Biology - Animal (2009)