Abstract
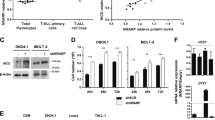
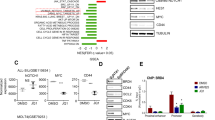
PI3K, mTOR and NOTCH pathways are frequently dysregulated in T-cell acute lymphoblastic leukaemia (T-ALL). Blockade of PI3K and mTOR with the dual inhibitor PI-103 decreased proliferation in all 15 T-ALL cell lines tested, inducing cell death in three. Combined PI3K/mTOR/NOTCH inhibition (with a γ-secretase inhibitor (GSI)) led to enhanced cell-cycle arrest and to subsequent cell death in 7/11 remaining NOTCH mutant cell lines. Commitment to cell death occurred within 48–72 h and was maximal when PI3K, mTOR and NOTCH activities were inhibited. PI-103 addition led to upregulation of c-MYC, which was blocked by coincubation with a GSI, indicating that PI3K/mTOR inhibition resulted in activation of the NOTCH-MYC pathway. Microarray studies showed a global increase in NOTCH target gene expression upon PI3K/mTOR inhibition. NOTCH-MYC-induced resistance to PI3K/mTOR inhibition was supported by synergistic cell death induction by PI-103 and a small molecule c-MYC inhibitor, and by reduction of the cytotoxic effect of PI-103+GSI by c-MYC overexpression. These results show that drugs targeting PI3K/mTOR can upregulate NOTCH-MYC activity, have implications for the use of PI3K inhibitors for the treatment of other malignancies with activated NOTCH, and provide a rational basis for the use of drug combinations that target both the pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Shuttleworth SJ, Silva FA, Cecil AR, Tomassi CD, Hill TJ, Raynaud FI et al. Progress in the preclinical discovery and clinical development of class I and dual class I/IV phosphoinositide 3-kinase (PI3K) inhibitors. Curr Med Chem 2011; 18: 2686–2714.
Zoncu R, Efeyan A, Sabatini DM . mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol 2011; 12: 21–35.
Ihle NT, Powis G . Take your PIK: phosphatidylinositol 3-kinase inhibitors race through the clinic and toward cancer therapy. Mol Cancer Ther 2009; 8: 1–9.
Maira SM, Stauffer F, Schnell C, Garcia-Echeverria C . PI3K inhibitors for cancer treatment: where do we stand? Biochem Soc Trans 2009; 37: 265–272.
Marone R, Cmiljanovic V, Giese B, Wymann MP . Targeting phosphoinositide 3-kinase: moving towards therapy. Biochim Biophys Acta 2008; 1784: 159–185.
Kraszewska MD, Dawidowska M, Szczepanski T, Witt M . T-cell acute lymphoblastic leukaemia: recent molecular biology findings. Br J Haematol 2012; 156: 303–315.
Gutierrez A, Look AT . NOTCH and PI3K-AKT pathways intertwined. Cancer Cell 2007; 12: 411–413.
Palomero T, Sulis ML, Cortina M, Real PJ, Barnes K, Ciofani M et al. Mutational loss of PTEN induces resistance to NOTCH1 inhibition in T-cell leukemia. Nat Med 2007; 13: 1203–1210.
Palomero T, Dominguez M, Ferrando AA. . The role of the PTEN/AKT pathway in NOTCH1-induced leukemia. Cell Cycle 2008; 7: 965–970.
Silva A, Yunes JA, Cardoso BA, Martins LR, Jotta PY, Abecasis M et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability. J Clin Invest 2008; 118: 3762–3774.
Larson Gedman A, Chen Q, Kugel Desmoulin S, Ge Y, Lafiura K, Haska CL et al. The impact of NOTCH1, FBW7 and PTEN mutations on prognosis and downstream signaling in pediatric T-cell acute lymphoblastic leukemia: a report from the Children’s Oncology Group. Leukemia 2009; 23: 1417–1425.
Mavrakis KJ, Van Der Meulen J, Wolfe AL, Liu X, Mets E, Taghon T et al. A cooperative microRNA-tumor suppressor gene network in acute T-cell lymphoblastic leukemia (T-ALL). Nat Genet 2011; 43: 673–678.
Gutierrez A, Sanda T, Grebliunaite R, Carracedo A, Salmena L, Ahn Y et al. High frequency of PTEN, PI3K, and AKT abnormalities in T-cell acute lymphoblastic leukemia. Blood 2009; 114: 647–650.
Chan SM, Weng AP, Tibshirani R, Aster JC, Utz PJ . Notch signals positively regulate activity of the mTOR pathway in T-cell acute lymphoblastic leukemia. Blood 2007; 110: 278–286.
Aster JC, Pear WS, Blacklow SC . Notch signaling in leukemia. Annu Rev Pathol 2008; 3: 587–613.
Weng AP, Ferrando AA, Lee W, JPt Morris, Silverman LB, Sanchez-Irizarry C et al. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science 2004; 306: 269–271.
Breit S, Stanulla M, Flohr T, Schrappe M, Ludwig WD, Tolle G et al. Activating NOTCH1 mutations predict favorable early treatment response and long-term outcome in childhood precursor T-cell lymphoblastic leukemia. Blood 2006; 108: 1151–1157.
Mansour MR, Duke V, Foroni L, Patel B, Allen CG, Ancliff PJ et al. Notch-1 mutations are secondary events in some patients with T-cell acute lymphoblastic leukemia. Clin Cancer Res 2007; 13: 6964–6969.
Mansour MR, Linch DC, Foroni L, Goldstone AH, Gale RE . High incidence of Notch-1 mutations in adult patients with T-cell acute lymphoblastic leukemia. Leukemia 2006; 20: 537–539.
Park MJ, Taki T, Oda M, Watanabe T, Yumura-Yagi K, Kobayashi R et al. FBXW7 and NOTCH1 mutations in childhood T cell acute lymphoblastic leukaemia and T cell non-Hodgkin lymphoma. Br J Haematol 2009; 145: 198–206.
O'Neil J, Grim J, Strack P, Rao S, Tibbitts D, Winter C et al. FBW7 mutations in leukemic cells mediate NOTCH pathway activation and resistance to gamma-secretase inhibitors. J Exp Med 2007; 204: 1813–1824.
Asnafi V, Buzyn A, Le Noir S, Baleydier F, Simon A, Beldjord K et al. NOTCH1/FBXW7 mutation identifies a large subgroup with favorable outcome in adult T-cell acute lymphoblastic leukemia (T-ALL): a Group for Research on Adult Acute Lymphoblastic Leukemia (GRAALL) study. Blood 2009; 113: 3918–3924.
Zhu YM, Zhao WL, Fu JF, Shi JY, Pan Q, Hu J et al. NOTCH1 mutations in T-cell acute lymphoblastic leukemia: prognostic significance and implication in multifactorial leukemogenesis. Clin Cancer Res 2006; 12: 3043–3049.
De Keersmaecker K, Lahortiga I, Mentens N, Folens C, Van Neste L, Bekaert S et al. In vitro validation of gamma-secretase inhibitors alone or in combination with other anti-cancer drugs for the treatment of T-cell acute lymphoblastic leukemia. Haematologica 2008; 93: 533–542.
Lewis HD, Leveridge M, Strack PR, Haldon CD, O'Neil J, Kim H et al. Apoptosis in T cell acute lymphoblastic leukemia cells after cell cycle arrest induced by pharmacological inhibition of notch signaling. Chem Biol 2007; 14: 209–219.
Tatton L, Morley GM, Chopra R, Khwaja A . The Src-selective kinase inhibitor PP1 also inhibits Kit and Bcr-Abl tyrosine kinases. J Biol Chem 2003; 278: 4847–4853.
Irizarry RA, Hobbs B, Collin F, Beazer-Barclay YD, Antonellis KJ, Scherf U et al. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 2003; 4: 249–264.
Weng AP, Millholland JM, Yashiro-Ohtani Y, Arcangeli ML, Lau A, Wai C et al. c-Myc is an important direct target of Notch1 in T-cell acute lymphoblastic leukemia/lymphoma. Genes Dev 2006; 20: 2096–2109.
Palomero T, Lim WK, Odom DT, Sulis ML, Real PJ, Margolin A et al. NOTCH1 directly regulates c-MYC and activates a feed-forward-loop transcriptional network promoting leukemic cell growth. Proc Natl Acad Sci USA 2006; 103: 18261–18266.
Wang H, Zou J, Zhao B, Johannsen E, Ashworth T, Wong H et al. Genome-wide analysis reveals conserved and divergent features of Notch1/RBPJ binding in human and murine T-lymphoblastic leukemia cells. Proc Natl Acad Sci USA 2011; 108: 14908–14913.
Moellering RE, Cornejo M, Davis TN, Del Bianco C, Aster JC, Blacklow SC et al. Direct inhibition of the NOTCH transcription factor complex. Nature 2009; 462: 182–188.
Chadwick N, Zeef L, Portillo V, Fennessy C, Warrander F, Hoyle S et al. Identification of novel Notch target genes in T cell leukaemia. Mol Cancer 2009; 8: 35.
Rao SS, O'Neil J, Liberator CD, Hardwick JS, Dai X, Zhang T et al. Inhibition of NOTCH signaling by gamma secretase inhibitor engages the RB pathway and elicits cell cycle exit in T-cell acute lymphoblastic leukemia cells. Cancer Res 2009; 69: 3060–3068.
Efeyan A, Sabatini DM . mTOR and cancer: many loops in one pathway. Curr Opin Cell Biol 2010; 22: 169–176.
Sicinska E, Aifantis I, Le Cam L, Swat W, Borowski C, Yu Q et al. Requirement for cyclin D3 in lymphocyte development and T cell leukemias. Cancer Cell 2003; 4: 451–461.
Joshi I, Minter LM, Telfer J, Demarest RM, Capobianco AJ, Aster JC et al. Notch signaling mediates G1/S cell-cycle progression in T cells via cyclin D3 and its dependent kinases. Blood 2009; 113: 1689–1698.
Chiarini F, Fala F, Tazzari PL, Ricci F, Astolfi A, Pession A et al. Dual inhibition of class IA phosphatidylinositol 3-kinase and mammalian target of rapamycin as a new therapeutic option for T-cell acute lymphoblastic leukemia. Cancer Res 2009; 69: 3520–3528.
Fala F, Blalock WL, Tazzari PL, Cappellini A, Chiarini F, Martinelli G et al. Proapoptotic activity and chemosensitizing effect of the novel Akt inhibitor (2S)-1-(1H-Indol-3-yl)-3-[5-(3-methyl-2H-indazol-5-yl)pyridin-3-yl]oxyprop an2-amine (A443654) in T-cell acute lymphoblastic leukemia. Mol Pharmacol 2008; 74: 884–895.
Faber AC, Li D, Song Y, Liang MC, Yeap BY, Bronson RT et al. Differential induction of apoptosis in HER2 and EGFR addicted cancers following PI3K inhibition. Proc Natl Acad Sci USA 2009; 106: 19503–19508.
Turke AB, Engelman JA . PIKing the right patient. Clin Cancer Res. 2010; 16: 3523–3525.
Liu Q, Kirubakaran S, Hur W, Niepel M, Westover K, Thoreen CC et al. Kinome-wide selectivity profiling of ATP-competitive mTOR (mammalian target of rapamycin) inhibitors and characterization of their binding kinetics. J Biol Chem 2012; 287: 9742–9752.
Wang H, Hammoudeh DI, Follis AV, Reese BE, Lazo JS, Metallo SJ et al. Improved low molecular weight Myc-Max inhibitors. Mol Cancer Ther 2007; 6: 2399–2408.
Rahl PB, Lin CY, Seila AC, Flynn RA, McCuine S, Burge CB et al. c-Myc regulates transcriptional pause release. Cell 2010; 141: 432–445.
Prochownik EV, Vogt PK. . Therapeutic targeting of Myc. Genes Cancer 2010; 1: 650–659.
Mansur MB, Hassan R, TC Barbosa, Splendore A, Jotta PY, Yunes JA et al. Impact of complex NOTCH1 mutations on survival in paediatric T-cell leukaemia. BMC Cancer 2012; 12: 9.
Evangelisti C, Ricci F, Tazzari P, Tabellini G, Battistelli M, Falcieri E et al. Targeted inhibition of mTORC1 and mTORC2 by active-site mTOR inhibitors has cytotoxic effects in T-cell acute lymphoblastic leukemia. Leukemia 2011; 25: 781–791.
Chiarini F, Grimaldi C, Ricci F, Tazzari PL, Evangelisti C, Ognibene A et al. Activity of the novel dual phosphatidylinositol 3-kinase/mammalian target of rapamycin inhibitor NVP-BEZ235 against T-cell acute lymphoblastic leukemia. Cancer Res 2010; 70: 8097–8107.
Uddin S, Hussain A, Al-Hussein K, Platanias LC, Bhatia KG . Inhibition of phosphatidylinositol 3′-kinase induces preferentially killing of PTEN-null T leukemias through AKT pathway. Biochem Biophys Res Commun 2004; 320: 932–938.
Knight ZA, Gonzalez B, Feldman ME, Zunder ER, Goldenberg DD, Williams O et al. A pharmacological map of the PI3-K family defines a role for p110alpha in insulin signaling. Cell 2006; 125: 733–747.
Guha S, Cullen JP, Morrow D, Colombo A, Lally C, Walls D et al. Glycogen synthase kinase 3 beta positively regulates Notch signaling in vascular smooth muscle cells: role in cell proliferation and survival. Basic Res Cardiol 2011; 106: 773–785.
Jin YH, Kim H, Oh M, Ki H, Kim K . Regulation of Notch1/NICD and Hes1 expressions by GSK-3alpha/beta. Mol Cells 2009; 27: 15–19.
Espinosa L, Ingles-Esteve J, Aguilera C, Bigas A . Phosphorylation by glycogen synthase kinase-3 beta down-regulates Notch activity, a link for Notch and Wnt pathways. J Biol Chem 2003; 278: 32227–32235.
Foltz DR, Santiago MC, Berechid BE, Nye JS . Glycogen synthase kinase-3beta modulates notch signaling and stability. Curr Biol 2002; 12: 1006–1011.
Song J, Park S, Kim M, Shin I . Down-regulation of Notch-dependent transcription by Akt in vitro. FEBS Lett 2008; 582: 1693–1699.
Mo JS, Ann EJ, Yoon JH, Jung J, Choi YH, Kim HY et al. Serum- and glucocorticoid-inducible kinase 1 (SGK1) controls Notch1 signaling by downregulation of protein stability through Fbw7 ubiquitin ligase. J Cell Sci 2011; 124: 100–112.
Li X, Sanda T, Look AT, Novina CD, von Boehmer H . Repression of tumor suppressor miR-451 is essential for NOTCH1-induced oncogenesis in T-ALL. J Exp Med 2011; 208: 663–675.
Guo W, Lasky JL, Chang CJ, Mosessian S, Lewis X, Xiao Y et al. Multi-genetic events collaboratively contribute to Pten-null leukaemia stem-cell formation. Nature 2008; 453: 529–533.
Gutierrez A, Grebliunaite R, Feng H, Kozakewich E, Zhu S, Guo F et al. Pten mediates Myc oncogene dependence in a conditional zebrafish model of T cell acute lymphoblastic leukemia. J Exp Med 2011; 208: 1595–1603.
Ilic N, Utermark T, Widlund HR, Roberts TM . PI3K-targeted therapy can be evaded by gene amplification along the MYC-eukaryotic translation initiation factor 4E (eIF4E) axis. Proc Natl Acad Sci USA 2011; 108: E699–E708.
Liu P, Cheng H, Santiago S, Raeder M, Zhang F, Isabella A et al. Oncogenic PIK3CA-driven mammary tumors frequently recur via PI3K pathway-dependent and PI3K pathway-independent mechanisms. Nat Med 2011; 17: 1116–1120.
Muellner MK, Uras IZ, Gapp BV, Kerzendorfer C, Smida M, Lechtermann H et al. A chemical-genetic screen reveals a mechanism of resistance to PI3K inhibitors in cancer. Nat Chem Biol 2011; 7: 787–793.
Puente XS, Pinyol M, Quesada V, Conde L, Ordonez GR, Villamor N et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 2011; 475: 101–105.
Robinson DR, Kalyana-Sundaram S, Wu YM, Shankar S, Cao X, Ateeq B et al. Functionally recurrent rearrangements of the MAST kinase and Notch gene families in breast cancer. Nat Med 2011; 17: 1646–1651.
Acknowledgements
This work was undertaken at the UCLH/UCL which received a proportion of funding from the NIHR Biomedical Research Centres funding scheme of the UK Department of Health. The work was supported by Leukaemia and Lymphoma Research, UK (LB and CWC), The Kay Kendall Leukaemia Fund (CS), the Medical Research Council UK (MRM) and the Tapner ALL Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Shepherd, C., Banerjee, L., Cheung, C. et al. PI3K/mTOR inhibition upregulates NOTCH-MYC signalling leading to an impaired cytotoxic response. Leukemia 27, 650–660 (2013). https://doi.org/10.1038/leu.2012.285
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2012.285
Keywords
This article is cited by
-
Cardiac-specific LRP6 knockout induces lipid accumulation through Drp1/CPT1b pathway in adult mice
Cell and Tissue Research (2020)
-
Memantine potentiates cytarabine-induced cell death of acute leukemia correlating with inhibition of Kv1.3 potassium channels, AKT and ERK1/2 signaling
Cell Communication and Signaling (2019)
-
Notch1 signaling pathway promotes invasion, self-renewal and growth of glioma initiating cells via modulating chemokine system CXCL12/CXCR4
Journal of Experimental & Clinical Cancer Research (2019)
-
Outlook on PI3K/AKT/mTOR inhibition in acute leukemia
Molecular and Cellular Therapies (2015)
-
Targeting PI3K/AKT/mTOR network for treatment of leukemia
Cellular and Molecular Life Sciences (2015)