Abstract
Objective:
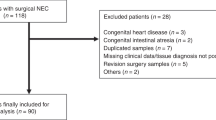
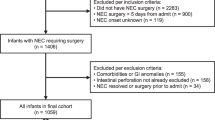
Up to a third of all infants who develop necrotizing enterocolitis (NEC) require surgical resection of necrotic bowel. We hypothesized that the histopathological findings in surgically resected bowel can predict the clinical outcome of these infants.
Study Design:
We reviewed the medical records and archived pathology specimens from all patients who underwent bowel resection/autopsy for NEC at a regional referral center over a 10-year period. Pathology specimens were graded for the depth and severity of necrosis, inflammation, bacteria invasion and pneumatosis, and histopathological findings were correlated with clinical outcomes.
Result:
We performed clinico-pathological analysis on 33 infants with confirmed NEC, of which 18 (54.5%) died. Depth of bacterial invasion in resected intestinal tissue predicted death from NEC (odds ratio 5.39 per unit change in the depth of bacterial invasion, 95% confidence interval 1.33 to 21.73). The presence of transmural necrosis and bacteria in the surgical margins of resected bowel was also associated with increased mortality.
Conclusion:
Depth of bacterial invasion in resected intestinal tissue predicts mortality in surgical NEC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 2010; 126 (3): 443–456.
Berrington JE, Hearn RI, Bythell M, Wright C, Embleton ND . Deaths in preterm infants: changing pathology over 2 decades. J Pediatrics 2012; 160 (1): 49–53 e41.
Moss RL, Kalish LA, Duggan C, Johnston P, Brandt ML, Dunn JC et al. Clinical parameters do not adequately predict outcome in necrotizing enterocolitis: a multi-institutional study. J Perinatol 2008; 28 (10): 665–674.
Ballance WA, Dahms BB, Shenker N, Kliegman RM . Pathology of neonatal necrotizing enterocolitis: a ten-year experience. J Pediatr 1990; 117 (1 Pt 2): S6–13.
Santulli TV, Schullinger JN, Heird WC, Gongaware RD, Wigger J, Barlow B et al. Acute necrotizing enterocolitis in infancy: a review of 64 cases. Pediatrics 1975; 55 (3): 376–387.
Walsh MC, Kliegman RM . Necrotizing enterocolitis: treatment based on staging criteria. Pediatr Clin North Am 1986; 33 (1): 179–201.
Gilbert-Barness E, Spicer DE, Steffensen TS . Handbook of Pediatric Autopsy Pathology. Springer Science: New York, NY, USA, 2014.
Cohen J . A coefficient of agreement for nominal scales. Educ Psychol Meas 1960; 20 (1): 37–46.
Smirnov N . Table for estimating the goodness of fit of empirical distributions. Ann Math Stat 1948; 19: 279–281.
Shapiro SS, Wilk MB . An analysis of variance test for normality (complete samples). Biometrika 1965; 52 (3-4): 591–611.
Levene H . Contributions to Probability and Statistics: Essays in Honor of Harold Hotelling. Stanford University Press: Redwood City, CA, USA, 1960.
Mann HB, Whitney DR . On a test of whether one of two random variables is stochastically larger than the other. Ann Math Stat 1947; 18 (1): 50–60.
Student. The probable error of a mean. Biometrika 1908; 6: 1–25.
Fisher RA . On the interpretation of χ2 from contingency tables, and the calculation of P. J R Stat Soc 1922; 85 (1): 87–94.
Nagelkerke NJD . A note on a general definition of the coefficient of determination. Biometrika 1991; 78 (3): 691–692.
Spearman C . The proof and measurement of association between two things. Am J Psychol 1904; 15: 72–101.
Mela CF, Koppale PK . The impact of colinearity on regression analysis: the asymmetric eVect of negative and positive correlations. J Appl Econ 2002; 34: 667–677.
Wald A . Tests of statistical hypotheses concerning several parameters when the number of observations is large. Trans Am Math Soc 1943; 54: 426–482.
Friedman JH, Meulman JJ . Multiple additive regression trees with application in epidemiology. Stat Med 2003; 22 (9): 1365–1381.
Zweig MH, Campbell G . Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 1993; 39 (4): 561–577.
Kurundkar AR, Killingsworth CR, McIlwain RB, Timpa JG, Hartman YE, He D et al. Extracorporeal membrane oxygenation causes loss of intestinal epithelial barrier in the newborn piglet. Pediatr Res 2010; 68 (2): 128–133.
Neu J, Walker WA . Necrotizing enterocolitis. N Engl J Med 2011; 364 (3): 255–264.
Wang Y, Hoenig JD, Malin KJ, Qamar S, Petrof EO, Sun J et al. 16S rRNA gene-based analysis of fecal microbiota from preterm infants with and without necrotizing enterocolitis. ISME J 2009; 3 (8): 944–954.
Brower-Sinning R, Zhong D, Good M, Firek B, Baker R, Sodhi CP et al. Mucosa-associated bacterial diversity in necrotizing enterocolitis. PLoS One 2014; 9 (9): e105046.
Morrow AL, Lagomarcino AJ, Schibler KR, Taft DH, Yu Z, Wang B et al. Early microbial and metabolomic signatures predict later onset of necrotizing enterocolitis in preterm infants. Microbiome 2013; 1 (1): 13.
Torrazza RM, Ukhanova M, Wang X, Sharma R, Hudak ML, Neu J et al. Intestinal microbial ecology and environmental factors affecting necrotizing enterocolitis. PLoS One 2013; 8 (12): e83304.
Hsueh W, Caplan MS, Tan X, MacKendrick W, Gonzalez-Crussi F . Necrotizing enterocolitis of the newborn: pathogenetic concepts in perspective. Pediatr Dev Pathol 1998; 1 (1): 2–16.
Gonzalez-Rivera R, Culverhouse RC, Hamvas A, Tarr PI, Warner BB . The age of necrotizing enterocolitis onset: an application of Sartwell's incubation period model. J Perinatol 2011; 31: 519–523.
Bury RG, Tudehope D . Enteral antibiotics for preventing necrotizing enterocolitis in low birthweight or preterm infants. Cochrane Database Syst Rev 2001; (1): CD000405.
Krimmel GA, Baker R, Yanowitz TD . Blood transfusion alters the superior mesenteric artery blood flow velocity response to feeding in premature infants. Am J Perinatol 2009; 26 (2): 99–105.
Nankervis CA, Giannone PJ, Reber KM . The neonatal intestinal vasculature: contributing factors to necrotizing enterocolitis. Semin Perinatol 2008; 32 (2): 83–91.
Touloukian RJ, Smith GJ . Normal intestinal length in preterm infants. J Pediatr Surg 1983; 18 (6): 720–723.
Ewer AK, Al-Salti W, Coney AM, Marshall JM, Ramani P, Booth IW . The role of platelet activating factor in a neonatal piglet model of necrotising enterocolitis. Gut 2004; 53 (2): 207–213.
Caplan MS, Sun XM, Hseuh W, Hageman JR . Role of platelet activating factor and tumor necrosis factor-alpha in neonatal necrotizing enterocolitis. J Pediatr 1990; 116 (6): 960–964.
Qu XW, Thaete LG, Rozenfeld RA, Zhu Y, De Plaen IG, Caplan MS et al. Tetrahydrobiopterin prevents platelet-activating factor-induced intestinal hypoperfusion and necrosis: Role of neuronal nitric oxide synthase. Crit Care Med 2005; 33 (5): 1050–1056.
Keyburn AL, Boyce JD, Vaz P, Bannam TL, Ford ME, Parker D et al. NetB, a new toxin that is associated with avian necrotic enteritis caused by Clostridium perfringens. PLoS Pathogens 2008; 4 (2): e26.
Holman RC, Stoll BJ, Clarke MJ, Glass RI . The epidemiology of necrotizing enterocolitis infant mortality in the United States. Am J Public Health 1997; 87 (12): 2026–2031.
Guner YS, Friedlich P, Wee CP, Dorey F, Camerini V, Upperman JS . State-based analysis of necrotizing enterocolitis outcomes. J Surg Res 2009; 157 (1): 21–29.
Maheshwari A, Schelonka RL, Dimmitt RA, Carlo WA, Munoz-Hernandez B, Das A et al. Cytokines associated with necrotizing enterocolitis in extremely-low-birth-weight infants. Pediatr Res 2014; 76 (1): 100–108.
Sharma R, Hudak ML, Tepas JJ 3rd, Wludyka PS, Marvin WJ, Bradshaw JA et al. Impact of gestational age on the clinical presentation and surgical outcome of necrotizing enterocolitis. J Perinatol 2006; 26 (6): 342–347.
Bagci S, Eis-Hubinger AM, Yassin AF, Simon A, Bartmann P, Franz AR et al. Clinical characteristics of viral intestinal infection in preterm and term neonates. Eur J Clin Microbiol Infect Dis 2010; 29 (9): 1079–1084.
Weber TR, Lewis JE . The role of second-look laparotomy in necrotizing enterocolitis. J Pediatr Surg 1986; 21 (4): 323–325.
Faingold R, Daneman A, Tomlinson G, Babyn PS, Manson DE, Mohanta A et al. Necrotizing enterocolitis: assessment of bowel viability with color doppler US. Radiology 2005; 235 (2): 587–594.
Vaughan WG, Grosfeld JL, West K, Scherer LR 3rd, Villamizar E, Rescorla FJ . Avoidance of stomas and delayed anastomosis for bowel necrosis: the 'clip and drop-back' technique. J Pediatr Surg 1996; 31 (4): 542–545.
Amin SC, Pappas C, Iyengar H, Maheshwari A . Short bowel syndrome in the NICU. Clin Perinatol 2013; 40 (1): 53–68.
Grimes DA, Schulz KF . Bias and causal associations in observational research. Lancet 2002; 359 (9302): 248–252.
Maheshwari A, Corbin LL, Schelonka RL . Neonatal necrotizing enterocolitis. Res Rep Neonatol 2011; 1: 39–53.
Dorling J, Kempley S, Leaf A . Feeding growth restricted preterm infants with abnormal antenatal Doppler results. Arch Dis Child Fetal Neonatal Ed 2005; 90 (5): F359–F363.
Andrews WW, Goldenberg RL, Faye-Petersen O, Cliver S, Goepfert AR, Hauth JC . The Alabama Preterm Birth study: polymorphonuclear and mononuclear cell placental infiltrations, other markers of inflammation, and outcomes in 23- to 32-week preterm newborn infants. Am J Obstet Gynecol 2006; 195 (3): 803–808.
Acknowledgements
This work was supported in part by the NIH award R01HD059142 (to AM).
Author contributions
AM, JIR, and SCA designed the study; JIR, SCA, SM, SAG and AM collected data; AAL and RR provided critical tools and expertise for data analysis; AM wrote the manuscript. All the authors reviewed and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Remon, J., Amin, S., Mehendale, S. et al. Depth of bacterial invasion in resected intestinal tissue predicts mortality in surgical necrotizing enterocolitis. J Perinatol 35, 755–762 (2015). https://doi.org/10.1038/jp.2015.51
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2015.51
This article is cited by
-
Role of macrophages in fetal development and perinatal disorders
Pediatric Research (2021)
-
Incomplete resection of necrotic bowel may increase mortality in infants with necrotizing enterocolitis
Pediatric Research (2021)
-
Role of platelets in neonatal necrotizing enterocolitis
Pediatric Research (2021)
-
Intestinal dysbiosis and necrotizing enterocolitis: assessment for causality using Bradford Hill criteria
Pediatric Research (2020)
-
Clinical determinants of postoperative outcomes in surgical necrotizing enterocolitis
Journal of Perinatology (2020)