Abstract
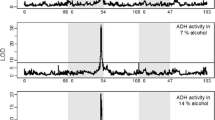
Acetone formation from propan-2-ol, a saturated secondary alcohol, has been analysed in flies of three different Adh-gentypes of D. melanogaster. The in vivo oxidation of propan-2-ol was mainly mediated through ADH activity. It could be demonstrated that flies homozygous for the Adh71k allele produced more acetone than flies homozygous for AdhF. This difference in metabolic flux mediated through the cryptic allozymes under non-saturated ADH-substrate conditions seems to be based on their different kinetic properties in vivo. Product inhibition of ADH monitored by means of ADH-isozymes conversion as observed after electrophoresis was similar for both cryptic allozymes.
ADH-71k and ADH-F showed immunological identity, and the in vivo protein levels of ADH-71k were 25–30 per cent higher than ADH-F.
The population-genetic implications of our findings have been evaluated.
Similar content being viewed by others
Article PDF
References
Benyajati, C, Place, A, and Sofer, W. 1983. Formaldehyde mutagenesis in Drosophila: Molecular analysis of ADH-negative mutants. Mutation Res, 111, 1–7.
Briscoe, D A, Malpica, J M, and Robertson, A. 1975. Dominance at the Adh locus in response of adult Drosophila melanogaster to environmental alcohol. Nature, 255, 148–149.
David, J R, Van Herrewege, J, Scheemaeker-Louis, M, and Pla, E. 1981. Drosophila alcohol dehydrogenase: Detoxification of isopropanol and acetone, substances not used in energy metabolism. Heredity, 47, 263–268.
David, J R, and Van Herrewege, J. 1983. Adaptation to alcoholic fermentation in Drosophila species: Relationship between alcohol tolerance and larval habitat. Comp Biochem Physiol, 74A, 283–288.
Eisses, K TH, Schoonen, W G E J, Aben, W, Scharloo, W, and Thörig, G E W. 1985. Dual function of the alcohol dehydrogenase of Drosophila melanogaster: Ethanol and acetaldehyde oxidation by two allozymes ADH-71k and ADH-F. Mol Gen Genet, 199, 76–81.
Geer, B W, and McKechnie, S W. 1984. Dietary ethanol and lipid synthesis in Drosophila melanogaster. Genetics, 107, s36.
Gibson, J B, Chambers, G K, Wilks, A V, and Oake-Shott, J G. 1980. An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. I. Activity ratios, thermostability, genetic localization and comparison with two other thermostable variants. Aust J Biol Sci, 33, 387–398.
Grell, E H, Jacobson, K B, and Murphy, J B. 1968. Alterations of genetic material for analysis of alcohol dehydrogenase isozymes of Drosophila melanogaster. Ann NY Acad Sci, 151, 441–455.
Heinstra, P W H, Eisses, K TH, Schoonen, W G E J, Aben, W, De Winter, A J, Van Der Horst, D J, Van Marrewijk, W J A, Beenakkers, A M TH, Scharloo, W, and Thörig, G E W. 1983. A dual function of alcohol dehydrogenase in Drosophila. Genetica 60, 129–137.
Heinstra, P W H, Eisses, K TH, Scharloo, W, and Thörig, G E W. 1986a. Metabolism of secondary alcohols in Drosophila melanogaster: Effects on alcohol dehydrogenase. Comp Biochem Physiol, 83B, 403–408.
Heinstra, P W H, Scharloo, W, and Thörig, G E W. 1986b. Alcohol dehydrogenase of Drosophila: Conversion and retroconversion of isozyme patterns. Comp Biochem Physiol, 83B, 409–414.
Hovik, R, Winberg, J-O, and McKinley-McKee, J S. 1984. Drosophila melanogaster alcohol dehydrogenase: Substrate stereospecificity of the AdhF alleloenzyme. Insect Biochem, 14, 345–351.
Kricka, L J, and Clark, P M S. 1979. Biochemistry of Alcohol and Alcoholism. John Wiley & Sons. New York.
Mancini, G, Carbonara, G O, and Heremans, J F. 1965. Immunochemical quantification of antigens by Single radial immunodiffusion. Immunochemistry, 2, 235–254.
Maroni, G, Laurie-Ahlberg, C C, Adams, D A, and Wilton, A N. 1982. Genetic Variation in the expression of ADH in Drosophila melanogaster. Genetics, 101, 431–436.
McKechnie, S W, and Morgan, P. 1982. Alcohol dehydrogenase polymorphism of Drosophila melanogaster: Aspects of alcohol and temperature Variation in the larval environment. Aust J Biol Sci, 35, 85–93.
McKechnie, S W, and Geer, B W. 1984. Regulation of alcohol dehydrogenase in Drosophila melanogaster by dietary alcohol and carbohydrate. Insect Biochem, 14, 231–242.
Middleton, R J, and Kacser, H. 1983. Enzyme Variation, metabolic flux and fitness: alcohol dehydrogenase in Drosophila melanogaster. Genetics, 105, 633–650.
Moxom, L N, Holmes, R S, Parsons, P A, Irving, M G, and Doddrell, D M. 1985. Purification and molecular properties of alcohol dehydrogenase from Drosophila melanogaster: Evidence from NMR and kinetic studies for function as an aldehyde dehydrogenase. Comp Biochem Physiol, 80B, 525–535.
Ouchterlony, O. 1953. Antigen-antibody reactions in gels. IV Types of reactions in coordinated Systems of diffusion. Acta Pathol Microbiol Scand, 22, 231–240.
Scharloo, W, Van Duken, F R, Hoorn, A J W, De Jong, G, and Thörig, G E W. 1977. Functional aspects of genetic Variation Christiansen, F. B. and Fenchel, T. M. (eds.) In Measuring Selection in Natural Populations. Springer. Berlin.
Schwartz, M, and Sofer, W. 1976. Diet-induced alterations in distribution of multiple forms of alcohol dehydrogenase in Drosophila. Nature, 263, 129–131.
Segel, I H. 1975. Enzyme kinetics. John Wiley & Sons. New York.
Thörig, G E W, Schoone, A A, and Scharloo, W. 1975. Variation between electrophoretically identical alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem Genet, 13, 721–731.
Van Delden, W. 1982. The alcohol dehydrogenase polymorphism in Drosophila melanogaster: Selection at an enzyme locus. Evol Biol, 15, 187–222.
Winberg, J-O, Thatcher, D R, and McKinley-McKee, J S. 1982. Alcohol dehydrogenase from the fruitfly, Drosophila melanogaster: Substrate speeificities of the alleloenzymes AdhS and AdhUF. Biochem Biophys Acta, 704, 7–16.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heinstra, P., Aben, W., Scharloo, W. et al. Alcohol dehydrogenase of Drosophila melanogaster: metabolic differences mediated through cryptic allozymes. Heredity 57, 23–29 (1986). https://doi.org/10.1038/hdy.1986.82
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1986.82
This article is cited by
-
A novel ancestral protein ofDrosophila alcohol dehydrogenase inStreptomyces?
Biochemical Genetics (1993)
-
A novel ancestral protein ofDrosophila alcohol dehydrogenase inStreptomyces?
Biochemical Genetics (1993)
-
Evolutionary genetics of the Drosophila alcohol dehydrogenase gene-enzyme system
Genetica (1993)
-
Participation of Drosophila melanogaster alcohol dehydrogenase (ADH) in the detoxification of 1-pentene-3-ol and 1-pentene-3-one
Heredity (1988)