Abstract
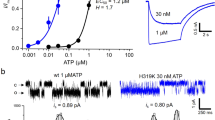
P2X receptors are ligand-gated ion channels activated by the binding of extracellular adenosine 5´-triphosphate (ATP). Brief (< 1 s) applications of ATP to nodose ganglion neurons or to cells transfected with P2X2 or P2X4 receptor cDNAs induce the opening of a channel selectively permeable to small cations within milliseconds. We now show that, during longer ATP application (10–60 s), the channel also becomes permeable to much larger cations such as N-methyl-D-glucamine and the propidium analog YO-PRO-1. This effect is enhanced in P2X2 receptors carrying point mutations in the second transmembrane segment. Progressive dilation of the ion-conducting pathway during prolonged activation reveals a mechanism by which ionotropic receptors may alter neuronal function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
North, R. A. & Barnard, E. A. Nucleotide receptors. Curr. Opin. Neurobiol. 7, 346–357 (1997).
North, R. A. P2X purinoceptor plethora. Semin. Neurosci. 8, 187–194 (1996).
Evans, R. J. & Surprenant, A. P2X receptors in autonomic and sensory neurons. Semin. Neurosci. 8, 217–223 (1996).
Evans, R. J. & Surprenant, A. Vasoconstriction of guinea-pig submucosal arterioles following sympathetic nerve stimulation is mediated by the release of ATP. Br. J. Pharmacol. 106, 242–249 (1992).
Evans, R. J., Derkach, V. & Surprenant, A. ATP mediates fast synaptic transmission in mammalian neurones. Nature 357, 503– 505 (1992).
Edwards, F. A., Gibb, A. J. & Colquhoun, D. ATP receptor-mediated synaptic currents in the central nervous system. Nature 359, 144– 147 (1992).
Gu, J. G. & MacDermott, A. B. Activation of ATP P2X receptors elicits glutamate release from sensory neuron synapses. Nature 389, 749–753 ( 1997).
Khakh, B. S. & Henderson, G. ATP receptor-mediated enhancement of fast excitatory neurotransmitter release in the brain. Mol. Pharmacol. 54, 372–378 ( 1998).
Le, K. T. et al. Sensory presynaptic and widespread somatodendritic immunolocalization of central ionotropic P2X ATP receptors. Neuroscience 83, 177–190 (1998).
Lewis, C. et al. Coexpression of P2X2 and P2X3 receptor subunits can account for ATP-gated currents in sensory neurons. Nature 377, 432–435 ( 1995).
Benham, C. D. & Tsien R. W. A novel receptor-operated Ca2+-permeable channel activated by ATP in smooth muscle. Nature 328, 275–278 ( 1987).
Virginio, C., North, R. A. & Surprenant, A. Calcium permeability and block at homomeric and heteromeric P2X2 and P2X3 receptors, and P2X receptors in rat nodose neurones. J. Physiol. (Lond.) 510, 27– 35 (1998).
Evans, R. J. et al. Ionic permeability and divalent cation effects on two ATP-gated cation channels (P2X receptors) expressed in heterologous cells. J. Physiol. (Lond.) 497, 413–422 (1996).
Valera, S. et al. A new class of ligand-gated ion channel defined by P2X receptor for extracellular ATP. Nature 371, 516– 519 (1994).
Garcia-Guzman, M. et al. Characterization of recombinant human P2X4 receptor reveals pharmacological differences to the rat homologue. Mol. Pharmacol. 51, 109–118 (1997).
Surprenant, A., Rassendren, F., Kawashima, E., North, R. A. & Buell, G. The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science 272, 735–738 (1996).
Collo, G. et al. Tissue distribution of the P2X7 receptor. Neuropharmacology 36 1277–1284 (1997).
Rassendren, F. et al. The permeabilizing ATP receptor (P2X7): cloning and expression of a human cDNA. J. Biol. Chem. 272, 5482–5486 (1997).
Di Virgilio, F. The P2Z purinoceptor: an intriguing role in immunity, inflammation and cell death. Immunol. Today 16, 524– 528 (1995).
Humphreys, B. D., Virginio, C., Surprenant, A., Rice, J. & Dubyak, G. R. Isoquinolines as antagonists of the P2X7 nucleotide receptor: high sensitivity for the human versus rat receptor homologues. Mol. Pharmacol. 54, 22–32 (1998).
Gill, C. H., Peters, J. A. & Lambert, J. J. An electrophysiological investigation of the properties of a murine recombinant 5-HT3 receptor stably expressed in HEK293 cells. Br. J. Pharmacol. 114, 1211–1221 (1995).
Brandle, U. et al. Desensitization of the P2X(2) receptor controlled by alternative splicing. FEBS Lett. 404, 294– 298 (1997).
Simon, J. et al. Localization and functional expression of splice variants of the P2X2 receptor. Mol. Pharmacol. 52, 237–248 (1997).
Collo, G. et al. Cloning of P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J. Neurosci. 16, 2495–2507 (1996).
Li, C., Peoples, R. W. & Weight, F. F. Ethanol-induced inhibition of a neuronal P2X purinoceptor by an allosteric mechanism. Br. J. Pharmacol. 123, 1–3 (1998).
Harden, T. K. et al. in P2 Nucleotide Receptors (eds. Turner, J. T., Weisman, G. A. & Fedan, J. S.) 109–134 (Humana, Totowa, New Jersey, 1998).
Buell, G., Lewis, C., Collo, G., North, R. A. & Surprenant, A. An antagonist-insensitive P2X receptor expressed in epithelia and brain. EMBO J. 15, 55– 62 (1996).
Rassendren, F., Buell, G., Newbolt, A., North, R. A. & Surprenant A. Identification of amino acid residues contributing to the pore of a P2X receptor. EMBO J. 16, 3446–3454 (1997).
Egan, T. M., Haines, W. R. & Voigt, M. M. A domain contributing to the ion channel of ATP-gated P2X2 receptors identified by the substituted cysteine accessibility method. J. Neurosci. 18, 2350– 2359 (1998).
Khakh, B. S., Humphrey, P. P. A. & Surprenant, A. Electrophysiological properties of P2X-purinoceptors in rat superior cervical, nodose and guinea-pig coeliac neurones. J. Physiol. (Lond.) 484, 385–395 (1995).
Li, C., Peoples, R. W. & Weight, F. F. Mg2+ inhibition of ATP-activated current in rat nodose ganglion neurons: evidence that Mg2+ decreases the agonist affinity of the receptor. J. Neurophysiol. 77, 3391–3395 (1997).
Evans, R. J. et al. Pharmacological characterization of heterologously expressed ATP-gated cation channels (P2X purinoceptors). Mol. Pharmacol. 48, 178–183 ( 1995).
Brake, A. J., Wagenbach, M. J. & Julius, D. New structural motif for ligand-gated ion channels defined by an ionotropic ATP receptor. Nature 371, 519–523 (1994).
Tiairi, A. et al. Ligand binding to the serotonin 5-HT3 receptor studied with a novel fluorescent ligand. Biochemistry (in press).
Fenwick, E. M., Marty, A. & Neher, E. A patch-clamp study of bovine chromaffin cells and of their sensitivity to acetylcholine. J. Physiol. (Lond.) 331, 577–597 (1982).
Clements, J. D. Rapid solution exchange using a piezoelectric translator. Axobits 14, 9–11 (1993).
Neher, E. in Methods in Enzymology, Ion Channels vol. 207, 123– 130 (1992).
Acknowledgements
We are grateful to Daniele Estoppey and Denis Fahmi and Alison Newbolt for assistance with cell culture, transfections and mutagenesis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Virginio, C., MacKenzie, A., Rassendren, F. et al. Pore dilation of neuronal P2X receptor channels. Nat Neurosci 2, 315–321 (1999). https://doi.org/10.1038/7225
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/7225
This article is cited by
-
P2X7 Receptor: an Emerging Target in Alzheimer’s Disease
Molecular Neurobiology (2024)
-
The functional change of the P2X7R containing the Ala348 to Thr polymorphism is associated with the pathogenesis of gout
Scientific Reports (2023)
-
P2X7 receptors and pannexin1 hemichannels shape presynaptic transmission
Purinergic Signalling (2023)
-
Involvement of P2X7 receptors in chronic pain disorders
Purinergic Signalling (2022)
-
Unravelling the intricate cooperativity of subunit gating in P2X2 ion channels
Scientific Reports (2020)