Abstract
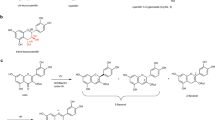
STUDYING the fate of exogenously applied indole-3-acetic acid (IAA) in pea epicotyls, we observed, besides indoleacetylaspartic acid1 (IAAsp), a trace amount of a compound which showed definitely indole reactive properties and which was split on alkaline hydrolysis into IAA. The amount of this metabolite proved too small for identification, therefore we carried out a rough survey on one species from 20 plant families using 2-14CIAA in the hope of finding an accumulation of this metabolite. This unknown compound was found in the leaves of the monocotyledonous plant Colchicum neapolitanum Ten. to about 65 per cent of the total amount of IAA taken up, the rest being unchanged IAA (20 per cent) and minor components (Fig. 1, top). IAAsp was detected, if at all, only in very small quantities. In order to isolate a larger amount of this compound, 70 gm. of mature Colchicum leaves were cut into pieces and incubated with 1 litre of a 4 × 10−4 M solution of IAA for 24 hr. with good aeration. The compound was extracted from the leaves with boiling 80 per cent aqueous ethanol; the extract was then concentrated to a small volume and made up to a powder with ‘Hyflo supercel’ and then extracted in a Soxhlet-type extractor for 6 hr. with ether. The ether containing only the IAA was discarded, and extraction continued with ethyl acetate for another 6 hr. This solution was taken to dryness and the residue containing the compound chromatographed as streaks on Whatman No. 3 paper in n-butanol / glacial acetic acid / water (5 : 1 : 2.2, R F 0.56) and in isopropanol / benzine / water (55 : 30 : 11, R F 0.39). By elution and rechromatography between the two solvents, the derivative could be purified and resulted finally in 6.05 mgm. of a highly purified, non-crystalline material. The compound was located on the paper by spraying with p-dimethylaminobenzaldehyde.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Andreae, W. A., and Good, N. E., Plant Phys., 30, 380 (1955).
Zenk, M. H., Naturwiss. (in the press).
Tang, Y. W., and Bonner, J., Arch. Biochem. Biophys., 13, 11 (1947).
Aronoff, S., Techniques of Radiobiochemistry (Iowa State College Press, 1957).
Lipmann, F., and Tuttle, L. C., J. Biol. Chem., 159, 21 (1945).
Jepson, J. B., Biochem. J., 60, 22P (1958).
Andreae, W. A., and van Ysselstein, M. W., Plant Phys., 35, 225 (1960).
Birkofer, L., Kaiser, C., and Kosmol, H., Naturwiss., 47 469 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ZENK, M. I-(Indole-3-acetyl)-β-D-Glucose, a New Compound in the Metabolism of Indole-3-acetic Acid in Plants. Nature 191, 493–494 (1961). https://doi.org/10.1038/191493a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/191493a0
This article is cited by
-
Analytical History of Auxin
Journal of Plant Growth Regulation (2015)
-
Transgenic Tomato Plants with a Modified Ability to Synthesize Indole-3-acetyl-β-1-O-D -glucose
Journal of Plant Growth Regulation (2005)
-
Plant hormone conjugation
Plant Molecular Biology (1994)
-
In vivo metabolism of labelled indole-3-acetic acid during polar transport in etiolated hypocotyls of Lupinus albus: Relationship with growth
Plant Growth Regulation (1988)
-
T-DNA-encoded auxin formation in crown-gall cells
Planta (1985)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.