Abstract
Purpose. The current formulation of paclitaxel contains ethanol and Cremophor EL and has been reported to cause serious adverse reactions. The purpose of the present work was to develop an improved emulsion vehicle for paclitaxel and to study the physicochemical properties of such a system.
Methods. Emulsions were prepared by either microfluidization or sonication method and the droplet size characterized by dynamic light scattering and light microscopy.
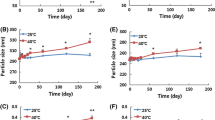
Results. Stable emulsions could be made using mixtures of lecithin/sodium deoxycholate as the emulsifiers. The formulation was further improved by using a combination of free acid and the sodium salt. Paclitaxel could be loaded into the emulsions at 2.5 mg/ml without the formation of drug crystals. While these emulsions were stable on storage, they flocculated when mixed with plasma. Steric stabilization of the emulsion droplets with poloxamer 188 increased the stability of the emulsions in plasma but promoted the crystallization of paclitaxel. The crystallization tendency could be reduced by using PEG5000PE (1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine-N-[poly (ethylene glycol) 5000]), a less water-soluble stabilizer
Conclusions. Emulsions with good stability characteristics containing 2.5 mg/ml paclitaxel could be made using bile salt/acid and lecithin, and the excellent stability of these emulsions in plasma was achieved by steric stabilization using PEG5000PE.
Similar content being viewed by others
REFERENCES
E. K. Rowinsky and R. C. Donehower. Paclitaxel (taxol). N. Engl. J. Med. 332:1004-1014 (1995).
D. B. Wilson, T. M. Beck, and C. A. Gundlach. Paclitaxel formulation as a cause of ethanol intoxication. Ann. Pharmacother. 31:873-875 (1997).
R. Weiss, R. C. Donehower, P. H. Wiernik, T. Ohnuma, R. A. Gralla, D. L. Trump, J. R. Baker, D. A. VanEcho, D. D. Von-Hoff, and B. Leyland-Jones. Hypersensitivity reactions from taxol. J. Clin. Oncol. 8:1263-1268 (1990).
H. Chen, Z. Zhang, C. McNulty, C. Olbert, H. Yoon, J. Lee, S. Kim, M. Seo, H. Oh, A. Lemmo, S. Ellis, and K. Heimlich. A high-throughput combinatorial approach for the discovery of a Cremophor EL-free paclitaxel formulation. Pharm. Res. 20:1302-1308 (2003).
H. Alkan-Onyuksel, S. Ramakrishnan, H. Chai, and J. M. Pezzuto. A mixed micellar formulation suitable for the parenteral administration of taxol. Pharm. Res. 11:206-212 (1994).
A. Krishnadas, I. Rubinstein, and H. Onyuksel. Sterically stabilized phospholipid mixed micelles: In vitro evaluation as a novel carrier for water-insoluble drugs. Pharm. Res. 20:297-302 (2003).
X. Zhang, J. K. Jackson, and H. M. Burt. Development of amphiphilic diblock copolymers as micellar carriers of taxol. Int. J. Pharm. 132:195-206 (1996).
A. Sharma, R. M. Straubinger, I. Ojima, and R. J. Bernacki. Antitumor efficacy of taxane liposomes on a human ovarian tumor xenograft in nude athymic mice. J. Pharm. Sci. 84:1404 (1995).
R. Perez-Soler and Y. Zou. Liposomes as carriers of lipophilic antitumor agents. In D. D. Lasicand D. Papahadjopoulos (eds.), Medical Applications of Liposomes, Elsevier Science B. V., Amsterdam, 1998.
M. Immordino, P. Brusa, S. Arpicco, B. Stella, F. Dosio, and L. Cattel. Preparation, characterization, cytotoxicity and pharmaco-kinetics of liposomes containing docetaxel. J. Control. Release 91:417-429 (2003).
P. Kan, Z. B. Chen, C. J. Lee, and I. M. Chu. Development of nonionic surfactant/phospholipid O/W emulsion as a paclitaxel delivery system. J. Control. Release 58:271-278 (1999).
B. B. Lundberg. A submicron lipid emulsion coated with amphiphathic polyethylene glycol for parenteral administration of paclitaxel (taxol). J. Pharm. Pharmaco. 49:16-21 (1997).
B. D. Tarr, T. G. Sambandan, and S. H. Yalkowsky. A new parenteral emulsion for the administration of taxol. Pharm. Res. 4:162-165 (1987).
D. Rodrigues, C. Covolan, S. Coradi, R. Barboza, and R. Maranhao. Use of a cholesterolrich emulsion that binds to low-density lipoprotein receptors as a vehicle for paclitaxel. J. Pharm. Pharmacol. 54:765-772 (2002).
P. Simamora, R. M. Dannenfelser, S. E. Tabibi, and S. H. Yalkowsky. Emulsion formulations for intravenous administration of paclitaxel. PDA J. Pharm. Sci. Technol. 52:170-172 (1998).
L. He, G. Wang, and Q. Zhang. An alternative paclitaxel micro-emulsion formulation: hypersensitivity evaluation and pharmaco-kinetic profile. Int. J. Pharm. 250:45-50 (2003).
P. P. Constantinides, K. J. Lambert, A. K. Tustian, B. Schneider, S. Lalji, W. W. Ma, B. Wentzel, D. Kessler, D. Worah, and S. C. Quay. Formulation development and antitumour activity of a filter-sterilizable emulsion of paclitaxel. Pharm. Res. 17:175-182 (2000).
S. S. Davis. and J. Han. Taxol emulsion, PCT Int. Appl., WO 99/04787, Danbiosyst UK Limited, UK, 1999.
S. Benita and M. Y. Levy. Submicron emulsions as colloidal drug carriers for intravenous administration: comprehensive physico-chemical characterization. J. Pharm. Sci. 82:1069-1079 (1993).
C. Washington. The electrokinetic properties of phospholipid stabilized fat emulsions.6. Zeta-potentials of Intralipid 20% in TPN mixtures. Int. J. Pharm. 87:167-174 (1992).
D. P. Cistola, J. A. Hamilton, D. Jackson, and D. M. Small. Ionization and phase behavior of fatty acids in water: application of the Gibbs phase rule. Biochem. 27:1881-1888 (1988).
D. F. Driscoll, F. Etzler, T. A. Barber, J. Nehne, W. Niemann, and B. R. Bistrian. Physicochemical assessments of parenteral lipid emulsions: light obscuration versus laser diffraction. Int. J. Pharm. 219:21-37 (2001).
L. Illum, S. S. Davis, C. G. Wilson, N. W. Thomas, M. Frier, and J. G. Hardy. Blood clearance and organ deposition of intravenously administered colloidal particles-the effects of particle size, nature and shape. Int. J. Pharm. 12:135-146 (1982).
R. J. Hunter. Foundations of Colloid Science, Oxford University Press, New York, 1986.
M. Malmsten and J. M. VanAlstine. Adsorption of poly(ethylene glycol) amphiphiles to form coatings which inhibit protein adsorption. J. Colloid Interface Sci. 177:502-512 (1996).
W. Lin, M. C. Garnett, M. C. Davies, F. Bignotti, P. Ferruti, S. S. Davis, and L. Illum. Preparation of surface-modified albumin nanospheres. Biomater. 18:559-565 (1997).
W. Lin, M. C. Garnett, E. Schacht, S. S. Davis, and L. Illum. Preparation and in vitro characterization of HSA-mPEG nano-particles. Int. J. Pharm. 189:161-170 (1999).
K. L. Prime and G. M. Whitesides. Adsorption of proteins onto surfaces containing end-attached oligo(ethylene oxide): a model system using self-assembled monolayers. J. Am. Chem. Soc. 115: 10714-10721 (1993).
P. Alexandridis and T. A. Hatton. Poly(ethylene oxide)-poly (propylene oxide)-poly(ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: thermodynamics, structure, dynamics and modeling. Colloid Surface A 96:1-46 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Han, J., Davis, S.S., Papandreou, C. et al. Design and Evaluation of an Emulsion Vehicle for Paclitaxel. I. Physicochemical Properties and Plasma Stability. Pharm Res 21, 1573–1580 (2004). https://doi.org/10.1023/B:PHAM.0000041451.70367.21
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000041451.70367.21