Abstract
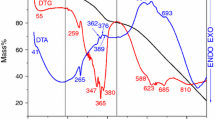
The adsorption of the anionic dye congo-red (CR) by Na-, Cs-, Mg-, Al- and Fe-montmorillonite was studied by simultaneous DTA-TG. Thermal analysis curves of adsorbed CR were compared with those of neat CR. The oxidation of neat CR is completed below 570°C. Thermal analysis curves of adsorbed CR show three regions representing dehydration of the clay, oxidation of the organic dye and dehydroxylation of the clay together with the oxidation of residual organic matter. The oxidation of the dye begins at about 250°C with the transformation of organic H atoms into water and carbon into charcoal. Two types of charcoal are obtained, low-temperature and high-temperature stable charcoal. The former gives rise to an exothermic peak in the second region of the thermal analysis and the latter in the third region. The exchangeable metallic cation determines the ratio between the low-temperature and high-temperature stable charcoal, which is formed. With increasing acidity of the exchangeable metallic cation higher amounts of high-temperature stable charcoal are obtained. It was suggested that aromatic compounds p bonded to the oxygen plane of the clay framework are converted into charcoal, which is burnt at about 550-700°C. With increasing surface acidity of the clay more species of CR are protonated. Only protonated dye species can form p bonds with oxygen plane and are converted to high-temperature stable charcoal during the thermal analysis. The thermal behavior of the dye complex of Cu-montmorillonite is different probably due to the catalytic effect of Cu.
Similar content being viewed by others
References
A. Langier-Kuźniarowa, 'Organo Clay Complexes and Interactions' (S. Yariv and H. Cross, Eds), Marcel Dekker, New York 2002, p. 273.
S. Yariv, 'Lecture Notes in Earth Sciences 38, Thermal Analysis in the Geoscience' (W. Smykatz-Kloss and S. St. J. Warne, Eds), Springer-Verlag, Berlin 1991, p. 328.
R. Greene-Kelly, in 'The Differential Thermal Investigation of Clays' (R. C. Mackenzie, Ed.), Mineralogical Society, London 1957, p. 140.
R. C. Mackenzie, in 'Differential Thermal Analysis' (R. C. Mackenzie, Ed.), 1. Academic Press, London 1970, p. 553.
W. Smykatz-Kloss, Differential Thermal Analysis. Application and Results in Mineralogy, Springer Verlag, Berlin 1974.
S. Yariv, Thermochim. Acta, 88 (1985) 49.
S. Yariv, J. Thermal Anal., 36 (1990) 1953.
W. Bodenheimer and L. Haller, Israel J. Chem., 6 (1968) 409.
S. Yariv, L. Heller, Z. Sofer and W. Bodenheimer, Israel J. Chem., 6 (1968) 741.
S. Yariv, L. Heller, Y. Deutsch and Y. Bodenheimer, Proc. 3rd Int. Conf. Thermal Anal., 3 (1971) 663.
S. Yariv, in 'Organo Clay Complexes and Interactions' (S. Yariv and H. Cross, Eds), Marcel Dekker, New York 2002, p. 463.
Z. Yermiyahu, I. Lapides and S. Yariv, J. Therm. Anal. Cal., 69 (2002) 317.
Z. Yermiyahu, I. Lapides and S. Yariv, submitted for publication.
S. Yariv, M. Müller-Vonmoos, G. Kahr and A. Rub, Thermochim. Acta, 148 (1989) 457.
S. Yariv, M. Müller-Vonmoos, G. Kahr and A. Rub, J. Thermal Anal., 35 (1989) 1941.
S. Yariv, M. Müller-Vonmoos, G. Kahr and A. Rub, J. Thermal Anal., 35 (1989) 1997.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yermiyahu, Z., Landau, A., Zaban, A. et al. Monoionic montmorillonites treated with Congo-Red. Journal of Thermal Analysis and Calorimetry 72, 431–441 (2003). https://doi.org/10.1023/A:1024596726087
Issue Date:
DOI: https://doi.org/10.1023/A:1024596726087