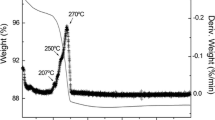
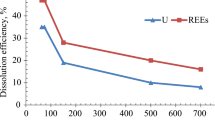
Abstract
The effects of iron on uranium oxidation states during sample dissolution were studied. A mineral acid mixture in anaerobic conditions was used for the dissolution the sample and the uranium oxidation states were determined by ion exchange. The first experiments were performed with pure iron chloride compounds. In the second stage, study was made of common iron-containing minerals. Uranium oxidation states were affected when the content of iron compound was as low as 10-5M. In the case of the natural minerals, pyrite caused uranium to change to an increasingly reduced state, whereas goethite caused it to change to an increasingly oxidized state as the amount of mineral was increased. The interferences of the silicates fell between those of pyrite and goethite. The results indicate that a wide range of common bulk rocks with less than 20 wt% of iron-containing minerals can be reliable chemically analyzed for uranium oxidation state.
Similar content being viewed by others
References
E. Rodrigues, F. Z. El Aamrani, J. Gimenez, I. Casas, M. E. Torrero, J. de Pablo, L. Duro, K-H. Hellmuth, Mat. Res. Symp. Proc., 506 (1998) 321.
A. Abdelouas, Y. Lu, W. Lutze, H. E. Nuttal, J. Contam. Hydrol., 35 (1998) 217.
L. L. Barton, K. Choudhury, B. M. Thomson, K. Steenhoudt, A. R. Groffman, Radioact. Waste Managem. Environ. Restor., 20 (1996) 141.
P. A. Bertrand, G. R. Choppin, Radiochim. Acta, 31 (1982) 135.
S. C. Foti, E. C. Freiling, Talanta, 11 (1964) 385.
I. Hodara, I. Balouka, Anal. Chem., 43 (1971) 1213.
A. Saito, G. R. Choppin, Anal. Chem., 55 (1983) 2454.
M. Fattahi, R. Guillamont, Radiochim. Acta, 61 (1993) 155.
M. Hussonnois, R. Guillamont, L. Brillard, M. Fattahi, Mater. Res. Soc., 127 (1989) 979.
K. A. Kraus, G. E. Moore, F. J. Nelson, Am. Chem. Soc., 78 (1956) 2692.
J. Bruno, I. Casas, L. Puigdomenech, Geochim. Cosmochim. Acta, 55 (1991) 647.
D. E. Grandstaff, Econ. Geol., 71 (1976) 1493.
R. F. Anderson, Nucl. Instr. Meth., 223 (1984) 213.
D. Von Borstel, P. Halbach, Fresenius Z. Anal. Chem., 310 (1982) 431.
A. Kobashi, T. Tominaga, Radiochim. Acta, 30 (1982) 205.
R. S. Clarke Jr., Z. S. Altschuler, Geochim. Cosmochim. Acta, 13 (1958) 127.
P. Wersin, M. F. Hochella Jr., P. Persson, G. Redden, J. O. Leckie, D. W. Harris, Geochim. Cosmochim. Acta, 58 (1994) 2829.
F. Z. El Aamrani, I. Casas, J. de Pablo, L. Duro, M. Grive, J. Bruno, Experimental and Modelling Study of the Interaction between Uranium(VI) and Magnetite, Report SKB TR-99-21, 1999.
B. Grambow, E. Smailos, H. Gerkeis, R. Muller, H. Hentschel, Radiochim. Acta, 74 (1996) 149.
K. Idemitsu, K. Obata, H. Furuya, Y. Inagaki, Mater. Res. Soc. Symp. Proc., 353 (1995) 981.
E. Liger, L. Charlet, P. van Cappellen, Geochim. Cosmochim. Acta, 63 (1999) 2939.
H. F. Steger, W. S. Bowman, Reference Uranium-Thorium Ore DL-1a, Certificate of Analysis, CANMET Report 80-10E, CANMET, Energy, Mines and Resources Canada, 1980.
H. Ervanne, J. Suksi, Radiochemistry, 38 (1996) 306.
D. Langmuir, Geochim. Cosmochim. Acta, 42 (1978) 547.
J. Acker, P. Bricker, Geochim. Cosmochim. Acta, 56 (1992) 3073.
J. Jolivet, E. Tronc, J. Coll. Interface Sci., 125 (1988) 688.
A. F. White, M. L. Peterson, Geochim. Cosmochim. Acta, 60 (1996) 3799.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ervanne, H. Interferences in uranium oxidation states during dissolution of solid phases. Journal of Radioanalytical and Nuclear Chemistry 256, 497–500 (2003). https://doi.org/10.1023/A:1024503916943
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1024503916943