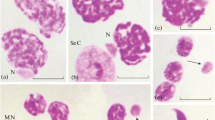
Abstract
It was shown that during ontogenesis, the mice prone to (SAMP1) and resistant (SAMR1) against accelerates senescence did not differ substantially in the frequency of cytogenetic aberrations in the hepatocytes and spermatogenic cells (spermatogonia and round spermatids). These data suggest that in the mice of both lines, the processes of appearance, development, and functioning of complex biological systems, such as liver and spermatogonic epithelium take place against the background of high genetic instability. The role of genetic instability in senescence is discussed.
Similar content being viewed by others
REFERENCES
Auerbach, Ch., Problemy mutageneza (Problems of Mutagenesis), Moscow: Mir, 1978.
Boekelheide, K., Lee, J., Shipp, E.B., et al., Expression of Fas System-Related Genes in the Testis during Development and after Toxicant Exposure, Toxicol. Lett., 1998, vol. 102, pp. 503-508.
Bohr, V.A. and Anson, R.M., DNA Damage, Mutation and Fine Structure DNA Repair in Aging, Mutat. Res., 1995, vol.338, pp. 25-34.
Carp, R.I., Meeker, H.C., Kozlowski, P., and Sersen, E.A., An Endogenous Retrovirus and Exogenous Scrapie in a Mouse Model of Aging, Trends Microbiol., 2000, vol. 8, pp. 39-42.
Curtis, H.J., Biological Mechanism Underlying the Aging Process, Science, 1963, vol. 141, pp. 686-694.
Cutler, R.G., Genetic Stability and Oxidative Stress: Common Mechanisms in Aging and Cancer, EXS, 1992, vol. 62, pp. 31-46.
Davidovich, M., Genetic Stability: The Key to Longevity? Med. Hypotheses, 1999, vol. 53, pp. 329-332.
Dolle, M.E., Giese, H., van Steeg, H., and Vijg, J., Mutation Accumulation in vivo and the Importance of Genome Stability in Aging and Cancer, Results Probl. Cell Differ., 2000, vol. 29, pp. 165-180.
Furuchi, T., Masuko, K., Nishimune, Y., et al., Inhibition of Testicular Germ Cells Apoptosis and Differentiation in Mice Misexpressing Bcl-2 In Spermatogonia, Development (Cambridge, UK), 1996, vol. 122, pp. 1703-1709.
Guo, Z., Toichi, E., Hosono, M., et al., Genetic Analysis of Lifespan in Hybrid Progeny Derived from the SAMP1 Mouse Strain with Accelerated Senescence, Mech. Ageing Dev., 2000, vol. 118, pp. 35-44.
He, P. and Yasumoto, K., Dietary Butylated Hydroxytoluene Counteracts with Paraquat to Reduce the Rate of Hepatic DNA Single Strand Breaks in Senescence-Accelerated Mice, Mech. Ageing Devel., 1994, vol. 76, pp. 43-48.
Higami, Y., Shimokawa, I., Okomoto, T., et al., Effect of Ageing and Dietary Restriction on Hepatocyte Proliferation and Death in Male F334 Rats, Cell Tissue Res., 1997, vol.288, pp. 69-77.
Higuchi, K., Wang, J., Kitagawa, K., et al., Accelerated Senile Amyloidosis Induced by Amyloidogenic Apoa-II Gene Shortens the Life Span of Mice But Does Not Accelerate the Rate of Senescence, J. Gerontol., 1996, vol. 51, pp.295-302.
Hosokawa, M. and Fuisawa, H., Zhu Bing-Hua, et al., In vitro Study of the Mechanisms of Senescence Acceleration, Exp. Gerontol., 1997, vol. 32, pp. 197-203.
Hosokawa, M., Fujisawa, H., Ax, S., et al., Age-Associated DNA Damage Is Accelerated in the Senescence-Accelerated Mice, Mech. Ageing Devel., 2000, vol. 118, pp. 61-70.
Johnson, F.B., Sinclair, D.A., and Guarente, L., Molecular Biology of Aging, Cell (Cambridge, Mass.), 1999, vol. 96, pp. 291-302.
Kipling, D. and Faragher, R.G.A., Ageing Hard Or Hardly Ageing?, Nature (London), 1999, vol. 398, pp. 191-192.
Kirkwood, T.B.L. and Cremer, T., Cytogerontology Since 1881: A Reappraisal of August Weismann and a Review of Modern Progress, Hum. Genet., 1982, vol. 60, pp. 101-121.
Kuro-o, M., Matsumura, Y., Aizawa, H., et al., Mutation of the Mouse Klotho Gene Leads to a Syndrome Resembling Ageing, Nature (London), 1998, vol. 390, pp. 45-51.
Kuzovatov, S.I., Krivtsov, V.Yu., and Vakhtin, Yu.B., Internuclear Chromosome Bridges and Nuclei with Protrusions in Populations of Rat Rhabdosarcoma RA-23 Cells, Tsitologiya, 2000, vol. 42, pp. 1097-1102.
Lee, J.W., Righburg, J.H., Shipp, E., et al., The Fas System, a Regulator of Testicular Germ Cell Apoptosis, Is Differentially Up-Regulated in Sertoli Cell Versus Germ Cell Injury of the Testis, Endocrinology, 1999, vol. 140, pp. 852-858.
Lindahl, T., Suppression of Spontaneous Mutagenesis in Human Cells by DNA Base Excision-Repair, Mutat. Res., 2000, vol. 462, pp. 129-135.
Lundberg, A.S., Hahn, W.C., Gupta, P., and Weinberg, R.A., Genes Involved in Senescence and Immortalization, Current Opinion Cell Biol., 2000, vol. 12, pp. 705-709.
Lutz, W.K., Endogenous Genotoxic Agents and Processes as a Basis of Spontaneous Carcinogenesis, Mutat. Res., 1990, vol. 275, pp. 305-315.
McClearn, G.E., Biogerontologic Theories, Exp. Gerontol., 1997, vol. 32, pp. 3-10.
Medvedev, Z.A., On the Immortality of the Germ Line: Genetic and Biochemical Mechanism. A Review, Mech. Ageing Devel., 1981, vol. 17, pp. 331-359.
Medvedev, Z.A., An Attempt at a Rational Classification of Theories of Ageing, Biol. Rev., 1990, vol. 65, pp. 375-398.
Muller, W.U. and Streffer, C., Micronucleus Assays, Advances in Mutagenesis Research, Obe, G., Ed., Berlin: Springer, 1994, pp. 4-134.
Nagley, P. and Yau-Huei Wei, Ageing and Mammalian Mitochondrial Genetics, Trends. Genet., 1998, vol. 14, pp. 513-517.
Nehlin, J.O., Skovgaard, G.L., and Bohr, V.A., The Werner syndrome. A Model for the Study of Human Aging, Ann. Acad., 2000, vol. 908, pp. 167-179.
Nisitani, S., Hosokawa, M., Sasaki, M.S., et al., Acceleration of Chromosome Aberrations in the Senescence-Accelerated Stains of Mice, Mutat. Res., 1990, vol. 237, pp. 221-228.
Odagiri, Y., Uchida, H., Hosokawa, M., et al., Accelerated Accumulation of Somatic Mutations in the Senescence-Accelerated Mouse, Nature Genetics, 1998, vol. 19, pp. 116-117.
Pichierri, P., Franchitto, A., Mosesso, P., and Palitti, F., Werner's Syndrome Cell Lines Are Hypersensitive to Camptothecin-Induced Chromosomal Damage, Mutat. Res., 2000, vol.456, pp. 45-57.
Print, C.G., Loveland, K.L., Gibson, L., et al., Apoptosis Regulator Bcl-W Is Essential for Spermatogenesis but Appears Otherwise Redundant, Proc. Natl. Acad. Sci. USA, 1998, vol. 95, pp. 12421-12431.
Rapoport, I.A., Mikrogenetika (Microgenetics), Moscow: Nauka, 1965.
Rapoport, I.A., Enzymatic Control of Mutagenesis in the Cell, Khimicheskii mutagenez i selektsiya (Chemical Mutagenesis and Selection), Moscow: Nauka, 1971, pp. 113-129.
Rapoport, I.A., Discovery of Chemical Mutagenesis, Izbrannye trudy (Selected Works), Moscow: Nauka, 1993.
Riggs, J.E., Aging, Increasing Genomic Entropy, and Neurodegenerative Disease, Neurol. Clin., 1999, vol. 16, pp. 757-770.
Rodriguez, I., Ody, C., Araki, K., et al., An Early and Massive Wave of Germinal Cell Apoptosis Is Required for the Development of Functional Spermatogenesis, EMBO J., 1997, vol. 16, pp. 2262-2270.
Ross, A., Waimare, K., Moss, J., et al., Testicular Degeneration in Bcl-W-Deficient Mice, Nature Genetics, 1998, vol.18, pp. 251-256.
Rotter, V., Schwartz, D., Almon, E., et al., Mice with Induced Level of Protein Exhibit the Testicular Giant-Cell Degenerative Syndrome, Proc. Natl. Acad. Sci. USA, 1993, vol. 90, pp. 9075-9079.
Schwartz, D., Goldfinger, N., Kam, Z., and Rotter, V., p53 Control Low DNA Damage-Dependent Premeiotic Checkpoint and Facilitates DNA Repair during Spermatogenesis, Cell Growth Differ., 1999, vol. 10, pp. 665-675.
Shen, J.C. and Loeb, L.A., The Werner Syndrome Gene. The Molecular Basis of RecQ Helicase-Deficiency Diseases, Trends. Genet., 2000, vol. 16, pp. 213-220.
Shimizu, N., Itoh, N., Utiyama, H., and Wahl, G.M., Selective Entrapment of Extrachromosomally Amplified DNA by Nuclear Budding and Micronucleation during S Phase, J. Cell Biol., 1998, vol. 140, pp. 1307-1320.
Shimura, M., Tanaka, Y., Nakamura, S., et al., Micronuclei Formation and Aneuploidy Induced by Vpr, an Accessory Gene of Human Immunodeficiency Virus Type 1, FASEB J., 1999, vol. 13, pp. 621-637.
Simons, J.W., Coming of Age: “Dysgenetics”-a Theory Connecting Induction of Persistent Delayed Genomic Instability with Disturbed Cellular Ageing, Int. J. Radiat. Biol., 2000, vol. 76, pp. 1533-1543.
Slagboom, P.E., The Aging Genome: Determinant or Target?, Mutat. Res., 1990, vol. 237, pp. 183-187.
Strehler, B.L., Deletional Mutations Are the Basic Cause of Aging: Historical Perspectives, Mutat. Res., 1995, vol. 338, pp. 3-17.
Szilard, L., On the Nature of the Aging Process, Proc. Natl. Acad. Sci. USA, 1959, vol. 45, pp. 30-45.
Takeda, T., Senescence-Accelerated Mouse (SAM): With Special Reference to Age-Associated Pathologies and Their Modulation, Nippon Eiseigaku Zasshi, 1996, vol. 51, pp. 569-578.
Takeda, T., Senescence-Accelerated Mouse (SAM): A Biogerontological Resource in Aging Research, Neurobiol. Ageing, 1999, vol. 20, pp. 105-110.
Takeda, T., Hosokawa, M., Takeshita, S., et al., A New Murine Model of Accelerated Senescence, Mech. Ageing Devel., 1981, vol. 17, pp. 183-194.
Takeda, T., Hosokawa, M., and Huguchi, K., Senescence-Accelerated Mouse (SAM). A Novel Murine Model of Aging, The SAM Model of Senescence, Takeda, T., Ed., Amsterdam: Excerpta Medica, 1994, pp. 15-22.
Takeda, T., Hosokawa, M., and Higuchi, K., Senescence-Accelerated Mouse (SAM): A Novel Murine Model of Senescence, Exp. Gerontol., 1997a, vol. 32, pp. 105-109.
Takeda, T., Matsushita, T., Kurozumi, M., et al., Pathobiology of the Senescence-Accelerated Mouse (SAM), Exp. Gerontol., 1997b, vol. 32, pp. 117-127.
Thakur, M.K., Oka, T., and Natori, Y., Gene Expression and Ageing, Mech. Ageing Devel., 1993, vol. 66, pp. 283-298.
Tobita, M., Nakamura, S., Nagano, I., et al., DNA Single-Strand Breaks Hippocampal Regions of Senescence-Accelerated Mice (SAMP8/Ta) Detected by Modified in situ Nick Translation Procedure, The SAM Model of Senescence, Takeda, T., Ed., Amsterdam: Excerpta Medica, 1994, pp. 125-128.
Uryvaeva, I.V. and Delone, G.V., An Improved Method of Mouse Liver Micronucleus Analysis: An Application to Age-Related Genetic Alteration and Polyploidy Study, Mutat. Res., 1995, vol. 334, pp. 71-80.
Uryvaeva, I.V. and Delone, G.V., Estimation of the Level of Genetic Damages Accumulated with Age and Induced Genetic Age Damages in the Cells According to the Production of Micronuclei, Ontogenez, 1992, vol. 23, pp. 370-377.
Uryvaeva, I.V. and Faktor, V.M., Fraction of Liver Growth, Its Ploidy Composition, and Changes during Senescence, Ontogenez (Moscow), 1975, vol. 6, pp. 458-465.
Uryvaeva, I.V., Marshak, T.L., Zakhidov, S.T., et al., Age-Related Accumulation of Micronuclear Aberrations in Hepatocytes of Senescence-Accelerated SAM mice, Dokl. Ross. Akad. Nauk, 1999, vol. 368, pp. 703-705.
Uryvaeva, I.V., Marshak, T.L., Zakhidov, S.T., et al., Genomic Disturbances, Elevated Death Rate and Proliferation of Hepatocytes in SAM (Senescence-Accelerated Mouse) Mice, IV Mezhdunar. simpozium “Biologicheskie mekhanizmy stareniya” (IV International Symposium “Biological Mechanisms of Aging”), Kharkov, 2000, p. 53.
Vijg, J., Somatic Mutations and Aging: A Re-Evaluation, Mutat. Res., 2000, vol. 447, pp. 117-135.
Xia, C., Higuchi, K., Shimizu, M., et al., Genetic Typing of the Senescence-Accelerated Mouse (SAM) Strains with Microsatellite Markers, Mamm. Genome, 1999, vol. 10, pp. 235-238.
Yan, W., Samson, M., Jegou, B., and Toppori, J., Bcl-W Forms Complexes with Bax and Bak, and Elevated Ratios of Spermatocyte Apoptosis in the Testis, Molec. Endocrinol., 2000, vol. 14, pp. 682-699.
Yin, Y., Stahl, B.C., DeWolf, W.C., and Morgentaler, F., p53-Mediated Germ Cell Quality Control in Spermatogenesis, Dev. Biol., 1998, vol. 204, pp. 165-171.
Zakhidov, S.T., Processes of Normal and Atypical Spermatogenesis in Animals, Doctoral (Biol.) Dissertation, Moscow: Mosk. Gos. Univ., 1993.
Zakhidov, S.T., Genetic Theory of Aging, Sinergetika (Synergetics), Moscow: Mosk. Gos. Univ., 1999, vol. 2, pp. 185-193.
Zakhidov, S.T., Semenova, M.L., Gordeeva, O.F., and Belyaeva, A.A., Spermatogenesis in SAMP1 Mice Prone to Accelerated Senescence, Dokl. Ross. Akad. Nauk, 1999, vol. 365, pp. 403-405.
Zakhidov, S.T., Gopko, A.V., Marshak, T.L., et al., Quantitative Patterns of Mutational Process during Spermatogenesis in SAM (Senescence-Accelerated Mouse) Mice, IV Mezhdunar. simpozium “Biologicheskie mekhanizmy stareniya” (IV International Symposium “Biological mechanisms of Aging”), Kharkov, 2000.
Zakhidov, S.T., Gordeeva, O.F., and Marshak, T.L., Biological Model of Accelerated Senescence. 1. Rate of Spontaneous Mutational Process during Spermatogenesis of SAM (Senescence-Accelerated Mouse) Mice, Izv. Akad. Nauk, Ser. Biol., 2001, no. 1, pp. 23-30.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zakhidov, S.T., Marshak, T.L., Uryvaeva, I.V. et al. Cytogenetic Aberrations in the Cells of Liver and Spermatogenic Epithelium in Senescence Accelerated SAMP1 and SAMR1 Mice. Russian Journal of Developmental Biology 33, 362–373 (2002). https://doi.org/10.1023/A:1021158727040
Issue Date:
DOI: https://doi.org/10.1023/A:1021158727040