Abstract
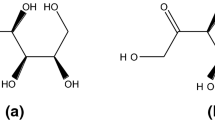
Isomalt, a commercial sugar alcohol widely used as a sweetener, is approximately a 1:1 mixture of two diastereomers, α-D-glucopyranosyl-1-6-mannitol (GPM) and α-D-glucopyranosyl-1-6-sorbitol (GPS). A calorimetric investigation has been carried out on mixtures of isomalt with GPS, in the (molar) composition range 0.45<xGPS<1, for both crystalline and amorphous states.
The GPS-rich portion of the solid-liquid GPM/GPS phase diagram was established and discussed in light of the existing literature. New evidence was given for the non-ideality of GPM/GPS mixtures, by indicating excess interactions in the melt and/or in the solid state. The commonly accepted hypothesis of a simple GPM/GPS eutectic forming isomalt was refuted in favour of more complicated mixture behaviour with possible formation of a complex.
Glass transition and physical ageing of isomalt and GPS were re-visited, with peculiar attention given to the measurement conditions. Standard thermal histories were adapted to each sample and the fictive temperature was used for the characterisation of the structural glass states. A linear increase of the fictive temperature was found upon passing from pure GPS to xGPS=0.45. GPS showed a slightly higher enthalpy relaxation rate than isomalt.
Similar content being viewed by others
References
A. Collet, M. J. Brienne and J. Jacques, Chem. Rev., 80 (1980) 215.
R. J. Prankerd and M. Z. Elsabee, Thermochim. Acta, 248 (1995) 147.
T. Ushio, R. Tamura, H. Takahashi, N. Azuma and K. Yamamoto, Angew. Chem. Int. Ed. Engl., 35 (1996) 2372.
H. E. Gallis, P. J. van Ekeren, J. C. van Miltenburg and H. A. J. Oonk, Thermochim. Acta, 326 (1999) 83.
A. Reisman, Phase Equilibria. Basic Principles, Applications, Experimental Techniques, Academic Press, New York 1970.
R. Koningsveld, W. H. Stockmayer and E. Nies, Polymer Phase Diagrams, Oxford University Press 2001.
A. Cesàro, in Thermodynamic Data for Biochemistry and Biotechnology, (H. J. Hinz Ed.) Springer Verlag, Berlin 1986, p. 177.
M. Siniti, S. Jabrane and J. M. Létoffé, Thermochim. Acta, 325 (1999) 171.
H. Schiweck, M. Munir, K. M. Rapp, B. Schneider and M. Vogel, in Carbohydrates as Organic Raw Materials, F. W. Lichtenthaler, Weinheim 1991, p. 57.
H. Schiweck, Alimenta, 19 (1980) 5.
B. Zielasko, Ermittlung physikalisch-chemischer Daten von Isomalt und seinen Komponenten. PhD thesis, University of Braunschweig 1997.
B. Borde and A. Cesàro, J. Therm. Anal. Cal., 66 (2001) 179.
H. K. Cammenga and B. Zielasko, Thermochim. Acta, 271 (1996) 149.
H. K. Cammenga, L. O. Figura and B. Zielasko, J. Therm. Anal., 47 (1996) 427.
C. T. Moynihan, A. J. Easteal, M. A. DeBolt and J. Tucker, J. Am. Ceram. Soc., 59 (1976) 12.
C. C. Huang and Y. P. Chen, Chem. Eng. Sci., 55 (2000) 3175.
I. M. Hodge, J. Non-Cryst. Solids, 169 (1994) 211.
P. R. Couchmann and F. E. Karasz, Macromolecules, 11 (1978) 117.
N. Lebrun and J. C. van Miltenburg, J. Alloys Compd., 320 (2001) 320.
M. Siniti, J. Carré, J. M. Létoffé, J. P. Bastide and P. Claudy, Thermochim. Acta, 224 (1993) 97.
F. Sussich and A. Cesàro, J. Therm. Anal. Cal., 62 (2000) 757.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Borde, B., Cesàro, A. Thermal properties of isomalt: A diastereomer mixture. Journal of Thermal Analysis and Calorimetry 69, 267–280 (2002). https://doi.org/10.1023/A:1019914512980
Issue Date:
DOI: https://doi.org/10.1023/A:1019914512980