Abstract
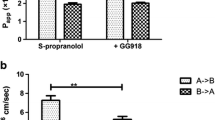
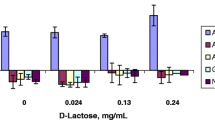
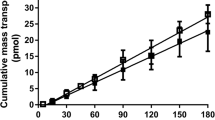
A series of O-cyclopropane carboxylic acid ester prodrugs of various β-blocking agents was synthesized. All prodrugs were hydrolyzed to give their parent compounds in aqueous phosphate buffer of pH 7.4 and in 80% human plasma. The half-lives in buffer solutions varied from 4 hours for the timolol prodrug to about 1 day for the prodrug of alprenolol. In human plasma the half-lives were shorter, ranging from 1 to 7 hours. The formation of the O-cyclopropane carboxylic acid ester derivatives significantly increased the lipophilicities of the β-blockers as measured by the distribution coefficient between n-octanol and aqueous phosphate buffer of pH 7.4. To characterize the biomembrane permeability characteristics of the β-blockers, transport properties across Caco-2 cell monolayers were investigated. An increase in lipophilicity resulted in a higher permeability of the prodrugs as compared to the parent compounds. Hence, acebutolol experienced an increment of a factor 17 on the apparent permeability coefficient, Papp, whereas Papp for the more lipophilic drug propranolol was increased by a factor of only 1.26. Some conversion of the prodrugs to their parent compounds was observed during the transport and appeared to be due to enzymatic intracellular metabolism.
Similar content being viewed by others
REFERENCES
R. D. Schoenwald and H.-S. Huang. Corneal penetration behavior of β-blocking agents I: Physicochemical factors. J. Pharm. Sci. 72:1266–1272 (1983).
J. M. Cruickshank. The clinical importance of cardioselectivity and lipophilicity in beta blockers. Am. Heart J. 100:160–178 (1980).
A. Buur, H. Bundgaard and V. H. L. Lee. Prodrugs of propranolol: Hydrolysis and intramolecular aminolysis of various propranolol esters and an oxazolidin-2-one derivative. Int. J. Pharm. 42:51–60 (1988).
H. Bundgaard, A. Buur, S.-C. Chang and V. H. L. Lee. Prodrugs of timolol for improved ocular delivery: Synthesis, hydrolysis kinetics and lipophilicity of various timolol esters. Int. J. Pharm. 33:15–26 (1986).
H. Bundgaard, A. Buur, S.-C. Chang and V. H. L. Lee. Timolol prodrugs: synthesis and lipophilicity of various alkyl, cycloalkyl and aromatic esters of timolol. Int. J. Pharm. 46:77–88 (1988).
N. Bodor and A. Elkoussi. Improved delivery through biological membranes. LVI. Pharmacological evaluation of alprenoxime—a new potential antiglaucoma agent. Pharm. Res. 8:1389–1395 (1991).
A. R. Hilgers, R. A. Conradi and P. S. Burton. Caco-2 cell monolayers as a model for drug transport across the intestinal mucosa. Pharm. Res. 7:902–910 (1990).
P. Artursson. Epithelial transport of drugs in cell culture. I. A model for studying the passive diffusion of drugs over intestinal absorptive (Caco-2) cells. J. Pharm. Sci. 79:476–482 (1990).
M. Pinto, S. Robine Leon, M.-D. Appay, M. Kedinger, N. Triadou, E. Dussaulux, B. Lacroix, P. Simon-Assmann, K. Haffen, J. Fogh and A. Zweibaum. Enterocyte-like differentiation and polarization of the human colon carcinoma cell line Caco-2 in culture. Bio. Cell. 47:323–330 (1983).
I. H. Hidalgo, T. J. Raub and R. Borchardt. Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology. 96:736–749 (1989).
G. Wilson, I. F. Hassan, C. J. Dix, I. Williamson, R. Shah, M. Mackay and P. Artursson. Transport and permeability properties of human Caco-2 cells: In-vitro model of the intestinal epithelial cell barrier. J. Controlled Release. 11:25–40 (1990).
I. J. Hidalgo, K. M. Hillgren, G. M. Grass and R. T. Borchardt. Characterization of the unstirred water layer in Caco-2 cell monolayers using a novel diffusion apparatus. Pharm. Res. 8:222–227 (1991).
A. Buur and N. Mørk. Metabolism of testosteron during in-vitro transport across Caco-2 cell monolayers: Evidence for β-hydroxysteroid dehydrogenase activity in differentiated Caco-2 cells. Pharm. Res. 9:1290–1294 (1992).
J. M. Quigley, C. G. M. Jordan and R. F. Timoney. The synthesis, hydrolysis kinetics and lipophilicity of O-acyl esters of propranolol. Int. J. Pharm. 101:145–163 (1993).
P. Nicklin, B. Irwin, I. Hassan, I. Williamson and M. Mackay. Permeable support type influences the transport of compounds across Caco-2 cells. Int. J. Pharm. 83:197–209 (1992).
D. C. Taylor, R. Pownall and W. Burke. The absorption of β-adrenoreceptor antagonists in rat in-situ small intestine; the effect of lipophilicity. J. Pharm. Pharmacol. 37:280–283 (1985).
P. Artursson and J. Karlsson. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochem. Biophys. Res. Comm. 175:880–885 (1991).
J. R. Pappenheimer. Physiological regulation of transepithelial impedance in the intestinal mucosa of rats and hamsters. J. Membr. Biol. 100:137–148 (1987).
M. Narawane, S. K. Podder, H. Bundgaard and V. H. L. Lee. Segmental differences in drug permeability, esterase activity and ketone reductase activity in the albino rabbit intestine. J. Drug Targ. 1:29–39 (1993).
Author information
Authors and Affiliations
Additional information
Deseaced.
Rights and permissions
About this article
Cite this article
Hovgaard, L., Brøndsted, H., Buur, A. et al. Drug Delivery Studies in Caco-2 Monolayers. Synthesis, Hydrolysis, and Transport of O-Cyclopropane Carboxylic Acid Ester Prodrugs of Various β-Blocking Agents. Pharm Res 12, 387–392 (1995). https://doi.org/10.1023/A:1016204602471
Issue Date:
DOI: https://doi.org/10.1023/A:1016204602471