Abstract
Purpose. The aim of this study was to develop a method based upon electrokinetic chromatography (EKC) using oppositely charged surfactant vesicles as a buffer modifier to estimate hydrophobicity (log P) for a range of neutral and charged compounds.
Methods. Vesicles were formed from cetyltrimethylammonium bromide (CTAB) and sodium n-octyl sulfate (SOS). The size and polydispersity of the vesicles were characterized by electron microscopy, dynamic light scattering, and pulsed-field gradient NMR (PFG-NMR). PFG-NMR was also used to determine if ion-pairing between cationic analytes and free SOS monomer occurred. The CTAB/SOS vesicles were used as a buffer modifier in capillary electrophoresis (CE). The capacity factor (log k′) was calculated by determining the mobility of the analytes both in the presence and absence of vesicles. Log k′ was determined for 29 neutral and charged analytes.
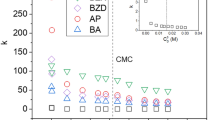
Results. There was a linear relationship between the log of capacity factor (log k′) and octanol/water partition coefficient (log P) for both neutral and basic species at pH 6.0, 7.3, and 10.2. This indicated that interaction between the cation and vesicle was dominated by hydrophobic forces. At pH 4.3, the log k′ values for the least hydrophobic basic analytes were higher than expected, indicating that electrostatic attraction as well as hydrophobic forces contributed to the overall interaction between the cation and vesicle. Anionic compounds could not be evaluated using this system.
Conclusion. Vesicular electrokinetic chromatography (VEKC) using surfactant vesicles as buffer modifiers is a promising method for the estimation of hydrophobicity.
Similar content being viewed by others
REFERENCES
O. H. Chan and B. H. Stewart. Physicochemical and drug-delivery considerations for oral drug bioavailability. Drug Discovery Today 1:461-473 (1996).
T. Fujita, J. Iwasa, and C. Hansch. A new substituent constant, π, derived from partition coefficients. J. Am. Chem. Soc. 86:5175-5180 (1964).
W. P. Purcell, G. E. Bass, and J. M. Clayton. Strategy of Drug Design, Wiley, New York, 1973, pp. 126-143.
Y. Ishihama, Y. Oda, K. Uchikawa, and N. Asakawa. Correlation of octanol-water partition coefficients with capacity factors measured by micellar electrokinetic chromatography. Chem. Pharm. Bull. 42:1525-1527 (1994).
M. Adlard, G. Okafo, E. Meenan, and P. Camilleri. Rapid estimation of octanol-water partition coefficients using deoxycholate micelles in capillary electrophoresis. J. Chem. Soc., Chem. Commun. 2241-2243 (1995).
B. J. Herbert and J. G. Dorsey. n-Octanol-water partition coefficient estimation by micellar electrokinetic capillary chromatography. Anal. Chem. 67:744-749 (1995).
J. T. Smith and D. V. Vinjamoori. Rapid determination of logarithmic partition coefficients between n-octanol and water using micellar electrokinetic capillary chromatography. J. Chromatogr. B 669:59-66 (1995).
S. Yang, J. G. Bumgarner, L. F. R. Kruk, and M. G. Khaledi. Quantitative structure-activity relationships studies with micellar electrokinetic chromatography: Influence of surfactant type and mixed micelles on estimation of hydrophobicity and bioavailability. J. Chromatogr. A 721:323-335 (1996).
M. Hanna, V. de Biasi, B. Bond, C. Salter, A. J. Hutt, and P. Camilleri. Estimation of the partitioning characteristics of drugs: A comparison of a large and diverse drug series utilizing chromatographic and electrophoretic methodology. Anal. Chem. 70:2092-2099 (1998).
Y. Ishihama, Y. Oda, K. Uchikawa, and N. Asakawa. Evaluation of solute hydrophobicity by microemulsion electrokinetic chromatography. Anal. Chem. 67:1588-1595 (1995).
Y. Ishihama, Y. Oda, and N. Asakawa. A hydrophobicity scale based on the migration index from microemulsion electrokinetic chromatography of anionic solutes. Anal. Chem. 68:1028-1032, (1996).
Y. Ishihama, Y. Oda, and N. Asakawa. Hydrophobicity of cationic solutes measured by electrokinetic chromatography with cationic microemulsions. Anal. Chem. 68:4281-4284, (1996).
K. Jinno and Y. Sawada. Relationships between capacity factors and hydropobicity of polycyclic aromatic hydrocarbons in cyclodextrin-modified micellar electrokinetic chromatography using surface treated capillaries. J. Liq. Chromatogr. 18:3719-3727 (1995).
M. G. Khaledi, S. C. Smith, and J. K. Strasters. Micellar electrokinetic capillary chromatography of acidic solutes: Migration behavior and optimization strategies. Anal. Chem. 63:1820-1830 (1991).
J. K. Strasters and M. G. Khaledi. Migration behavior of cationic solutes in micellar electrokinetic capillary chromatography. Anal. Chem. 63:2503-2508 (1991).
K. L. Herrington, E. W. Kaler, D. D. Miller, J. A. Zasadzinski, and S. Chiruvolu. Phase behavior of aqueous mixtures of dodecyltrimethylammonium bromide (DTAB) and sodium dodecyl sulfate (SDS). J. Phys. Chem. 97:13792-13802 (1993).
M. T. Yatcilla, K. L. Herrington, L. L. Brasher, E. W. Kaler, S. Chiruvolu, and J. A. Zasadzinski. Phase behavior of aqueous mixtures of cetyltrimethylammonium bromide (CTAB) sodium octyl sulfate (SOS). J. Phys. Chem. 100:5874-5879 (1996).
M. Hong, B. Weekley, S. J. Grieb, and J. P. Foley. Electrokinetic chromatography using thermodynamically stable vesicles and mixed micelles formed from oppositely charged surfactants. Anal. Chem. 70:1394-1403 (1998).
R. G. Bates. Determination of pH: Theory and Practice, Wiley, New York, 1964, pp. 219-220.
S. J. Gibbs and C. S. Johnson, Jr. A PFG NMR experiment for accurate diffusion and flow studies in the presence of eddy currents. J. Magn. Reson. 93:395-402 (1991).
D. Wu, A. Chen, and C. S. Johnson, Jr. An improved diffusion-ordered spectroscopy experiment incorporating bipolar-gradient pulses. J. Magn. Reson. Ser. A 115:260-264 (1995).
M. Lin, D. A. Jayawickrama, R. A. Rose, J. A. DelViscio, and C. K. Larive. Nuclear magnetic resonance spectroscopic analysis of the selective complexation of the cis and trans isomers of phenylalanylproline by β-cyclodextrin. Anal. Chim. Acta 307: 449-457 (1995).
K. F. Morris, B. J. Cutak, A. M. Dixon, and C. K. Larive. Analysis of diffusion coefficient distributions in humic and fulvic acids by means of diffusion ordered NMR spectroscopy. Anal. Chem. 71:5315-5321 (1999).
M. M. Bushey and J. W. Jorgenson. Effects of methanol-modified mobile phase on the separation of isotopically substituted compounds by micellar electrokinetic chromatography. J. Microcolumn Sep. 1:125-130 (1989).
W. S. Price. Pulsed-field gradient nuclear magnetic resonance as a tool for studying translational diffusion part I: Basic theory. Concepts Magn. Reson. 9:299-336 (1997).
S. W. Provencher. A constrained regularization method for inverting data represented by linear algebraic or integral equations. Comput. Phys. Commun. 27:213-227 (1982).
D. P. Hinton and C. S. Johnson, Jr. Diffusion ordered 2D NMR spectroscopy of phospholipid vesicles: Determination of vesicle size distributions. J. Phys. Chem. 97:9064-9072 (1993).
P. N. Craig. Cumulative subject index and drug compendium, Vol. 6. In C. Hansch, P. G. Sammes, and J. B. Taylor (eds.), Comprehensive Medicinal Chemistry, Pergamon Press, Oxford, 1990, pp. 237-965.
A. C. Moffat, J. V. Jackson, M. S. Moss, and B. Widdop (eds.). Clarke's Isolation and Identification of Drugs, The Pharmaceutical Press, London, 1986.
R. P. Austin, A. M. Davies, and C. N. Manners. Partitioning of ionizing molecules between aqueous buffers and phospholipid vesicles. J. Pharm. Sci. 84:1180-1183 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Razak, J.L., Cutak, B.J., Larive, C.K. et al. Correlation of the Capacity Factor in Vesicular Electrokinetic Chromatography with the Octanol: Water Partition Coefficient for Charged and Neutral Analytes. Pharm Res 18, 104–111 (2001). https://doi.org/10.1023/A:1011039129664
Issue Date:
DOI: https://doi.org/10.1023/A:1011039129664