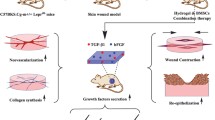
Abstract
Purpose. To accelerate diabetic wound healing with TGF-β1 gene delivery system using a thermosensitive hydrogel made of a triblock copolymer, PEG-PLGA-PEG.
Methods. Two 7 × 7 mm full thickness excisional wounds were created in parallel at the back of each genetically diabetic mouse. The hydrogel containing plasmid TGF-β1 was administered to the wound and formed an adhesive film in situ. Controls were either untreated or treated with the hydrogel without DNA. We used a commercial wound dressing, Humatrix®, either with or without DNA, to compare the therapeutic effect with the thermosensitive hydrogel.
Results. We found that thermosensitive hydrogel alone is slightly beneficial for reepithealization at early stage of healing (day 1-5), but significantly accelerated repithelializaion, increased cell proliferation, and organized collagen were observed in the wound bed treated with thermosensitive hydrogel containing plasmid TGF-β1. The accelerated reepithelialization was accompanied with enhanced collagen synthesis and more organized extracellular matrix deposition. Humatrix® alone or with plasmid TGF-β1, had little effect.
Conclusions. Thermosensitive hydrogel made of PEG-PLGA-PEG triblock copolymer provides excellent wound dressing activity and delivers plasmid TGF-β1 to promote wound healing in a diabetic mouse model.
Similar content being viewed by others
REFERENCES
Lanning DA, Diegelmann RF, Yager DR, Wallace ML, Bagwell CE, and Haynes JH. Myofibroblast induction with transforming growth factor-beta1 and-beta3 in cutaneous fetal excisional wounds. J. Pediatr. Surg. 35(2):183-187; discussion 187-188, (2000).
G. S. Sidhu, H. M. Mani, J. P. Gaddipati, A. K. Singh, P. Seth, K. K. Banaudha, G. K. Patnaik, and R. K. Maheshwari. Curcumin enhances wound healing in stretozocin induced diabetic rats and genetically diabetic mice. Wound Rep. Reg. 7:362-374 (1999).
E. B. Jude, R. Blakytny, J. Bulmer, A. J. Boulton, and M. W. Ferguson. Transforming growth factor-beta 1, 2, 3 and receptor type I and II in diabetic foot ulcers. Diabet. Med. 19:440-447 (2002).
H. D. Beer, M. T. Longaker, and S. Werner. Reduced expression of PDGF and PDGF receptors during impaired wound healing. J. Invest. Dermatol. 109:132-138 (1997).
D. G. Greenhalgh, K. H. Sprudel, M. J. Murray, and R. Ross. PDGF and FGF stimulate wound healing in the genetically diabetic mouse. Am. J. Pathol. 136:1235-1246 (1990).
S. Chesnoy, P. Y. Lee, and L. Huang. Intradermal injection of transforming growth factor-β1 gene enhances wound healing in genetically diabetic mice. Pharm. Res. 20:345-350 (2003).
J. Wang, P. C. Zhang, H. Q. Mao, and K. W. Leong. Enhanced gene expression in mouse muscle by sustained release of plasmid DNA using PPE-EA as a carrier. Gene Ther. 9:1254-1261 (2002).
J. Bonadio, E. Smiley, P. Patil, and S. Goldstein. Localized, direct plasmid gene delivery in vivo: prolonged therapy results in reproducible tissue regeneration. Nat. Med. 5:753-759 (1999).
W. A. Border and N. A. Noble. TGF β in tissue fibrosis. N. Engl. J. Med. 10:1286-1292 (1994).
Z. Li, W. Ning, J. Wang, A. Choi, P. Y. Lee, P. Tyagi, and L. Huang. Control gene delivery system based on thermosensitive biodegradable hydrogel. Pharm. Res. 20(6):884-888 (2003).
B. Jeong, Y. H. Bae, and S. W. Kim. In situ gelation of PEG-PLGA-PEG triblock copolymer aqueous solutions and degradation thereof. J. Biomed. Mater. Res. 50:171-177 (2000).
J. Byeongmoon, H. You, and W. Sung. Biodegradable thermosenstive micelles of PEG-PLGA-PEG triblock copolymers. Colloids and Surfaces B: Biointerfaces 16:185-193 (1999).
F. Sweat, H. Puchtler, and S. Rosenthal. Sirius red F3BA as stain for connective tissue. Archs. Pathology 78:69-72 (1964).
C. R. Hofman, J. P. Dileo, Z. Li, S. Li, and L. Huang. Efficient in vivo gene transfer by PCR amplified fragment with reduced inflammatory activity. Gene Ther. 8(1):71-74 (2001).
G. E. Zaikov. Quantitative aspects of polymer degradation on the living body. JMS-Rev. Macromol. Chem.Phys. C25(4):551-597 (1985).
S. Frank, M. Madlener, and S. Werner. Transforming growth factors β1, β2, and β3 and their receptors are differentially regulated during normal and impaired wound healing. J. Biol. Chem. 271:10188-10193 (1996).
Y. Ito, P. Sarkar, Q. Mi, N. Wu, P. BringasJr., Y. Liu, S. Reddy, R. Maxson, C. Deng, and Y. Chai. Overexpression of Smad2 reveals its concerted action with Smad4 in regulating TGF-beta-mediated epidermal homeostasis. Dev. Biol. 236:181-194 (2001).
P. J. Christner, C. M. Artlett, R. F. Conway, and S. A. Jimenez. Increased numbers of microchimeric cells of fetal origin are associated with dermal fibrosis in mice following injection of vinyl chloride. Arthritis Rheum. 43:2598-2605 (2000).
D. A. Lanning, B. C. Nwomeh, S. J. Montante, D. R. Yager, R. F. Diegelmann, and J. H. Haynes. TGF-beta1 alters the healing of cutaneous fetal excisional wounds. J. Pediatr. Surg. 34(5):695-700 (1999).
D. A. Bettinger, D. R. Yager, R. F. Diegelmann, and I. K. Cohen. The effect of TGF-beta on keloid fibroblast proliferation and collagen synthesis. Plast. Reconstr. Surg. 98(5):827-833 (1996).
C. L. Theoret, S. M. Barber, T. N. Moyana, and J. R. Gordon. Preliminary observations on expression of transforming growth factors beta1 and beta3 in equine full-thickness skin wounds healing normally or with exuberant granulation tissue. Vet. Surg. 31:266-273 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, PY., Li, Z. & Huang, L. Thermosensitive Hydrogel as a Tgf-β1 Gene Delivery Vehicle Enhances Diabetic Wound Healing. Pharm Res 20, 1995–2000 (2003). https://doi.org/10.1023/B:PHAM.0000008048.58777.da
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000008048.58777.da