Abstract
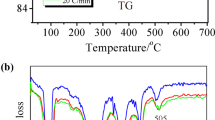
Using thermal analysis as well as isothermal gravimetric measurements, the thermal stability of the zinc nitrate(V) hexahydrate has been determined. Some intermediate products are the results of melting, evaporation, hydrolysis, polycondensation and decomposition processes occurring in the salt - water system up to 300°C. Solid phases were characterized by means of chemical analyses and X-ray powder diffraction method.
Similar content being viewed by others
References
A. Małecki, B. Małecka, R. Gajerski and S. Łabu, J. Therm. Anal. Cal., 72 (2003) 135.
T. Sato, J. Therm. Anal. Cal., 69 (2002) 255.
A. C. Oliveira, G. S. Marchetti and M. Carmo Rangel, J. Therm. Anal. Cal., 73 (2003) 233.
C. Duval, Inorganic Thermogravimetric Analysis, Elsevier, Amsterdam 1963, p. 408.
I. Noshida, J. Chem. Soc. Japan, 48 (1927) 520.
W. W. Wendtland, Texas J. Sci., 10 (1958) 392.
Atlas of Thermoanalytical Curves, Vol. 5, Akadémiai Kiadó, Budapest 1976, p. 144.
A. Małecki, R. Gajerski, S. Łabu, B. Prochowska-Klisch and K. T. Wojciechowski, J. Therm. Anal. Cal., 60 (2000) 17 and lit. cit. herein.
A. J. Kozak, K. Wieczorek-Ciurowa and A. Kozak, XL Scientific Conf. of Polish Chem. Soc. & Assoc. of Engineers & Technicians of Chem. Industry, Gdańsk, Sept. 22-26, 1997, Abstracts S-19, P-5 (in Polish).
M. A. Mohamed and S. A. Halawy, J. Thermal Anal., 41 (1994) 147.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kozak, A.J., Wieczorek-Ciurowa, K. & Kozak, A. The thermal transformations in Zn(No3)2-H2O (1:6) system. Journal of Thermal Analysis and Calorimetry 74, 497–502 (2003). https://doi.org/10.1023/B:JTAN.0000005186.15474.be
Issue Date:
DOI: https://doi.org/10.1023/B:JTAN.0000005186.15474.be