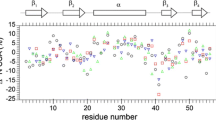
Abstract
Many important proteins contain multiple domains connected by flexible linkers. Inter-domain motion is suggested to play a key role in many processes involving molecular recognition. Heteronuclear NMR relaxation is sensitive to motions in the relevant time scales and could provide valuable information on the dynamics of multi-domain proteins. However, the standard analysis based on the separation of global tumbling and fast local motions is no longer valid for multi-domain proteins undergoing internal motions involving complete domains and that take place on the same time scale than the overall motion.
The complexity of the motions experienced even for the simplest two-domain proteins are difficult to capture with simple extensions of the classical Lipari–Szabo approach. Hydrodynamic effects are expected to dominate the motion of the individual globular domains, as well as that of the complete protein. Using Pin1 as a test case, we have simulated its motion at the microsecond time scale, at a reasonable computational expense, using Brownian Dynamic simulations on simplified models. The resulting trajectories provide insight on the interplay between global and inter-domain motion and can be analyzed using the recently published method of isotropic Reorientational Mode Dynamics which offer a way of calculating their contribution to heteronuclear relaxation rates. The analysis of trajectories computed with Pin1 models of different flexibility provides a general framework to understand the dynamics of multi-domain proteins and explains some of the observed features in the relaxation rate profile of free Pin1.
Similar content being viewed by others
References
Allen, M.P. and Tildesley, D.J. (1990) Computer Simulations of Liquids, Oxford, Claredon Press.
Antosiewicz, J. and McCammon, J.A. (1995) Biophys. J., 69, 57–65.
Antosiewicz, J., Briggs, J.M. and McCammon, J.A. (1996) Eur. Biophys. J., 24, 137–141.
Avigan, M.I., Stober, B. and Levens, D.A. (1990) J. Biol. Chem., 265, 18538–18545.
Baber, J., Szabo, A. and Tjandra, N. (2001) J. Am. Chem. Soc., 123, 3953–3959.
Bernadó, P., Åkerud, T., García de la Torre, J., Akke, M. and Pons, M. (2003) J. Am. Chem. Soc. 125, 916–923.
Bernadó, P., García de la Torre, J. and Pons, M. (2002) J. Biomol. NMR, 23, 139–150.
Braddock, D.T., Louis, J.M., Baber, J.L., Levens, D. and Clore, M.G. (2002) Nature, 415, 1051–1056.
Brune, D. and Kim, S. (1994) Proc. Natl. Acad. Sci. USA, 91, 2930–2934.
Brüschweiler, R., Liao, X. and Wright, P. (1995) Science, 268, 886–889.
Burd, C.G. and Dreyfuss, G. (1994) Science, 265, 615–621.
Campbell, I.D. and Downing, A.K. (1998) Nat. Struct. Biol., 5, 496–499.
Carrasco, B. and García de la Torre, J. (1999a) Biophys. J., 75, 3044–3057.
Carrasco, B. and García de la Torre, J. (1999b) J. Chem. Phys., 110, 4817–4826.
Chang, S.-L. and Tjandra, N. (2001) J. Am. Chem. Soc., 123, 11484–11485.
Chou, J.J., Li, S., Klee, C.B. and Bax, A. (2001) Nat. Struct. Biol., 8, 990–997.
Clore, G.M., Szabo, A., Bax, A., Kay, L.E., Driscoll, P.C. and Gronenborn, A.M. (1990) J. Am. Chem. Soc., 112, 4989–4991.
Dagget, V. (2000) Curr. Opin. Struct. Biol., 10, 160–164.
Ermak, D.L. and MacCammon, J.A. (1978) J. Chem. Phys., 69, 1352–1360.
Fernandes, M.X., Bernadó, P., Pons, M. and García de la Torre, J. (2001) J. Am. Chem. Soc., 123, 12037–12047.
Fischer, M.W.F., Losonczi, J.A., Weaver, J.L. and Prestegard, J.H. (1999) Biochemistry, 38, 9013–9022.
Fushman, D., Xu, R. and Cowburn, D. (1999) Biochemistry, 38, 10225–10230.
García Bernal, J.M. and García de la Torre, J. (1980) Biopolymers, 19, 751–766.
García de la Torre, J. and Bloomfield, V.A. (1981) Q. Rev. Biophys., 14, 81–139.
García de la Torre, J., Huertas, M.L. and Carrasco, B. (2000) J. Magn. Reson., 147, 138–147.
Gerstein, M. and Krebs, W. (1998) Nucl. Acids Res., 26, 4280–4290.
Ikura, M., Clore, G.M., Gronenborn, A.M., Zhu, G., Klee, C.B. and Bax, A. (1992) Science, 256, 632–638.
Iniesta, A. and García de la Torre, J. (1990) J. Chem. Phys., 92, 2015–2018.
Jacobs, D.M., Saxena, K., Vogtherr, M., Bernadó, P., Pons, M. and Fiebig, K. (2003) J. Biol. Chem., 278, 26174–26182.
Levitt, M. (1976) J. Mol. Biol., 104, 59–107.
Lipari, G. and Szabo, A. (1982a) J. Am. Chem. Soc., 104, 4546–4559.
Lipari, G. and Szabo, A. (1982b) J. Am. Chem. Soc., 104, 4559–4570.
Lu, K.P., Liou, Y.-C. and Zhou, X.Z. (2002) Trends Cell Biol., 12, 164–172.
Meador, W.E., Means, A.R. and Quiocho, F.A. (1993) Science, 262, 1718–1721.
Peter, C., Daura, X. and van Gunsteren, W.F. (2001) J. Biomol. NMR, 20, 297–310.
Prompers, J.J. and Brüschweiler, R. (2001) J. Am. Chem. Soc., 123, 7305–7313.
Prompers, J.J. and Brüschweiler, R. (2002a) J. Am. Chem. Soc., 124, 4522–4534.
Prompers, J.J. and Brüshweiler, R. (2002b) Proteins, 46, 177–189.
Prompers, J.J., Lienin, S.F. and Brüschweiler, R. (2001) In Biocomputing: Proceedings of the 2001 Pacific Symposium, Altman, R.B., Dunker, A.K., Hunter, L., Lauderdale, K. and Klein, T.E., World Scientific, Singapore, pp. 79–88.
Ranganathan, R., Lu, K.P., Hunter, T. and Noel, J.P. (1997) Cell, 89, 875–886.
Rotne, J. and Prager, J. (1969) J. Chem. Phys., 50, 4831–4837.
Sekerina, E., Rahfeld, J.U., Müller, J., Fanghänel, C.R., Fischer, G. and Bayer, P.J. (2000) Mol. Biol., 301, 1003–1017.
Sicheri, F. and Kuriyan, J. (1997) Curr. Opin. Struct. Biol., 7, 777–785.
Tjandra, N., Garrett, D.S., Gronenborn, A.M., Bax, A. and Clore, G.M. (1997) Nat. Struct. Biol., 4, 443–449.
Tolman, J.R., Flanagan, J.M., Kennedy, M.A. and Prestegard, J.H. (1995) Proc. Natl. Acad. Sci. USA, 92, 9279–9283.
Varadan, R., Walker, O., Pickart, C. and Fushman, D. (2002) J. Mol. Biol., 324, 637–647.
Verdecia, M.A., Bowman, M.E., Lu, K.P., Hunter, T. and Noel, J.P. (2000) Nat. Struct. Biol., 7, 639–643.
Wade, R.C., Davies, M.E., Luty, B.A., Madura, J.D. and McCammon, J.A. (1993) Biophys. J., 64, 9–15.
Yamakawa, H. (1970) J. Chem. Phys., 53, 436–443.
Yuan, X., Werner, J.M., Lack, J., Knott, V., Handford, P.A., Campbell, I.D. and Downing, A.K. (2002) J. Mol. Biol, 316, 113–125.
Zhou, X.Z., Lu, P.-J., Wulf, G. and Lu, K.P. (1999) Cell. Mol. Life Sci., 56, 788–806.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Bernadó, P., Fernandes, M.X., Jacobs, D.M. et al. Interpretation of NMR relaxation properties of Pin1, a two-domain protein, based on Brownian dynamic simulations. J Biomol NMR 29, 21–35 (2004). https://doi.org/10.1023/B:JNMR.0000019499.60777.6e
Issue Date:
DOI: https://doi.org/10.1023/B:JNMR.0000019499.60777.6e