Abstract
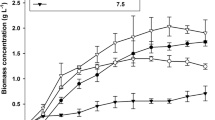
The cell growth and eicosapentaenoic acid (EPA) yields of Nannochloropsis sp. were enhanced in the fed-batch cultures. With feeding glucose solution, the biomass reached 1.1 g dry wt l−1 after 10 days' culture, which was 40% higher than that obtained in the batch culture (0.8 g dry wt l−1). With supplement of nitrate solution, the biomass reached 1 g dry wt l−1, and reached the stationary phase 2 days earlier than the others. The maximum of biomass (1.2 g dry wt l−1) was obtained with the supplement of the mixture of glucose and nitrate solution. The EPA yields of Nannochloropsis sp. after 10 days' growth in the fed-batch cultures were 52 mg l−1, 43 mg l−1 and 56 mg l−1 with, respectively, addition of nitrate, glucose and both together. In batch culture only 35 mg EPA l−1 was obtained.
Similar content being viewed by others
References
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37: 911-917.
Carreau JP, Dudacg JP (1978) Adaptation of a macro-scale method to the micro-scale for fatty acid methyl transesterification of biological lipid extracts. J. Chromatogr. 151: 384-390.
Chen F, Johns MR (1991) Effect of C/N ratio and aeration on the fatty acid composition of heterotrophic Chlorella sorokiniana. J. Appl. Phycol. 3: 203-209.
Chen F, Zhang Y (1997) High cell density mixotrophic culture of Spirulina platensis on glucose for phycocyanin production using a fed-batch system. Enzyme Microb. Technol. 20: 221-224.
Chini Zittelli G, Lavista F, Bastianini A, Rodolfi L, Vincenzini M, Tredici MR (1999) Production of eicosapentaenoic acid by Nan - nochloropsis sp. cultures in outdoor tubular photobioreactors. J. Biotech. 70: 299-312.
Collos Y, Mornet F, Sciandra A, Waser N, Larson A, Harrison PJ (1999) An optical method for the rapid measurement of micro-molar concentration of nitrate in marine phytoplankton cultures. J. Appl. Phycol. 11: 179-184.
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal. Biochem. 28: 350-356.
Markwell MAK, Haas SM, Bieber LL, Tolbert NE (1978) A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal. Biochem. 87: 206-210.
Molina GE, Sánchez PJA (1992) EPA from Isochrysis galbana. Growth conditions and productivity. Proc. Biochem. 27: 299-305.
Suen Y, Hubbard JS, Holzer G, Tornabene TG (1987) Total lipid production of the green algae Nannochloropsis sp. QII under different nitrogen regimes. J. Phycol. 23: 289-296.
Sukenik A, Zmora O, Carmeli Y (1993) Biochemical quality of mar-ine unicellular algae with special emphasis on lipid composition. II. Nannochloropsis sp. Aquaculture 117: 313-326.
Wen ZY, Jiang Y, Chen F (2002) High cell density culture of the diatom Nitzschia laevis for eicosapentaenoic acid production: fed-batch development. Proc. Biochem. 37: 1447-1453.
Xu F, Hu H, Cong W, Cai Z, Ouyang F (2004) Growth characteristics and eicosapentaenoic acid production by Nannochloropsis sp. in mixotrophic conditions. Biotechnol. Lett. 26: 51-53.
Xu N, Zhang X, Fan X, Han L, Tseng CK (2001) Effects of nitrogen source and concentration on growth rate and fatty acid composition of Ellipsoidion sp. (Eustigmatophyta). J. Appl. Phycol. 13: 463-469.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xu, F., Cai, Zl., Cong, W. et al. Growth and fatty acid composition of Nannochloropsis sp. grown mixotrophically in fed-batch culture. Biotechnology Letters 26, 1319–1322 (2004). https://doi.org/10.1023/B:BILE.0000045626.38354.1a
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000045626.38354.1a