Abstract
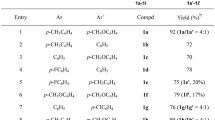
The reaction of 5-anilino(toluidino-, morpholino)-1,3,4-thiadiazoline-2-thiones at 80°C with allyl bromide and benzyl chloride in alcohol, acetonitrile or DMF in the presence of KOH and also with phenoxymethyloxirane in alcohol in the absence of base gives the corresponding novel allyl-, benzyl-, and 2-hydroxy-3-phenoxypropyl products substituted at the exocyclic S atom. Alkylation of the indicated thiones with benzyl chloride at 150-153°C in DMF in the presence of KOH occurs similarly. Under these conditions, allyl bromide forms alkylation products at the endocyclic N(3) atom as a result of an S→N thio-Claisen rearrangement of the initially formed product which is allyl substituted at the exocyclic S atom.
Similar content being viewed by others
REFERENCES
E. D. Shtefan and Yu. V. Vvedenskii, Usp. Khim., 65, 326 (1996)
J. Sandstrom and J. Wennerbeek, Acta Chem. Scand., 20, 57 (1966)
K. Arvidsson and J. Sandstrom, Acta Chem. Scand., 15, 1179 (1961).
M. B. Kolesova, L. I. Maksimova, and A. V. El'tsov, Zh. Org. Khim., 9, 2613 (1973).
A. K. Ramrakhyani and R. S. Shukla, J. Indian Chem. Soc., 57 856 (1980).
S. K. Sangal and A. Kumar, J. Indian Chem. Soc., 65, 811 (1988)
Y. Makisumi, Tetrahedron Lett., 6399 (1966)
A. C. Fabretti, G. C. Franchini, and G. Peyronel, Spectrochim. Acta, 36A, 1289 (1980).
A. R. Katritzky, Z. Wang, and R. J. Ofterman, J. Heterocycl. Chem., 27, 139 (1990)
V. Ya. Kazakov and I. Ya. Postovskii, Izv. Vuzov. Khimiya i Khim. Tekhnol., 4, 238 (1961)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Polvonov, K., Sabirov, Q. & Shakhidoyatov, K.M. The Route of Alkylation of 5-Substituted 1,3,4-Thiadiazoline-2-thiones. Chemistry of Heterocyclic Compounds 39, 228–232 (2003). https://doi.org/10.1023/A:1023776610073
Issue Date:
DOI: https://doi.org/10.1023/A:1023776610073