Abstract
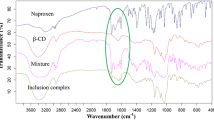
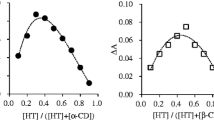
Nifluminic acid (N) is a frequently used anti-inflammatory drug,but its poor aqueous solubility is a disadvantageous property. Inclusion complexation with cyclodextrinderivatives (CDs) affords a possibility to increase its solubility properties. For thispurpose, different CDs were used, primarily hydroxypropyl-β-CD (HP-β-CD), to prepareproducts by powder mixing or kneading in four molecular ratios. The dissolution and in vitromembrane diffusion of the products were investigated. The wetting angles of pure N andHP-β-CD and of the products, and the n-octanol/water partition coefficients were determined.The interaction, leading to complex formation between the components of the products was examined by thermoanalytical methods.
Similar content being viewed by others
REFERENCES
The Merck Index: 11th edn., Merck & Co., Rahway, NJ (1989).
Martindale: The Extra Pharmacopoeia, 31st edn., The Royal Pharm. Society, London (1996).
USP: 23rd edn., US Pharm. Conv. Inc., Rockville, MD (1994).
H. Stricker: Pharm. Ind. 31, 794 (1969).
Booklet of Sartorius Resorption Model, SM 16750, Göttingen, p. 15 (1976).
F. Giordano, Cs. Novak, and J.R. Moyano: Thermochim. Acta 380, 123 (2001).
Rights and permissions
About this article
Cite this article
Kata, M., Ambrus, R. & Aigner, Z. Preparation and Investigation of Inclusion Complexes Containing Nifluminic Acid and Cyclodextrins. Journal of Inclusion Phenomena 44, 123–126 (2002). https://doi.org/10.1023/A:1023074025175
Issue Date:
DOI: https://doi.org/10.1023/A:1023074025175