Abstract
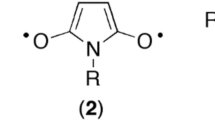
Diazomethane and ethyl diazoacetate are highly reactive and highly versatile synthetic reagents that undergo numerous related reactions. However, while the former is highly dangerous because of its toxicity and explosive behavior; the latter is much more benign. This is usually ascribed to resonance stabilization in ethyl diazoacetate involving an extra carbonyl group that is absent in diazomethane, cf.
The additional resonance stabilization is derived using a recent literature measurement of the enthalpy of an ethyl diazoacetate/aldehyde reaction, key enthalpies of formation, also from the literature, and some simplifying assumptions. The resonance stabilization is deduced to be but 16 kJ/mol, merely 4 kcal/mol. But, oh how grateful we are for this!
Similar content being viewed by others
REFERENCES
Oneil, M. J.; Smith, A., Eds. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals; 13 edn.; Merck Co.: Rahway, NJ, 2001.
Fieser, M.; Fieser, L. F. Reagents for Organic Synthesis; Wiley: New York, 1967; Vol. 1.
Chen, H.-M.; Hosmane, R. S.; Baldisseri, D. M. J. Heterocyclic. Chem. 2000,37, 951.
Qiu, Y. L.; Ksebati, M. B.; Ptak, R. G.; Fan, B. Y.; Breitenbach, J. M.; Lin, J. S.; Cheng, Y. C.; Kern, E. R.; Drach, J. C.; Zemlicka, J. J. Med. Chem. 1998,41 10.
Kovtonyuk, V. N.; Kobrina, L. S.; Bagryanskaya, I. Y.; Gatilov, Y. V. Russ. J. Org. Chem. 1999,35, 67.
Bachmann, W. E.; Struve, W. S. Org. Reactions 1942,1, 38.
Alguacil, R.; Farina, F.; Martin, M. V.; Paredes, M. C. Tetrahedron 1999,55, 229.
Mock, W. L.; Hartman, M. E. J. Amer. Chem. Soc. 1970,92, 5767.
Cram, D. J.; Helgeson, R. C. J. Amer. Chem. Soc. 1966,88, 3515.
Armour, M.-A. Hazardous Laboratory Chemicals Disposal Guide; 2 edn.; Lewis Publishers, CRC Press: Boca Raton, FL, 1996. For contemporary discussion of the thermal stability and lack of detonation (as opposed to “merely” explosive) properties of ethyl diazoacetate, see Clark, J. D.; Shah, A. S.; Peterson, J. C.; Patelis, L.; Kersten, R. J. A.; Grogan, F. M.; Heemskerk, A. H. Thermochim. Acta,in press, and Clark, J. D.; Shah, A. S.; Peterson, J. C.; Patelis, L.; Kersten, R. J. A.; Heemskerk. A. H.; Grogan, M.; Camden, S. Thermochim. Acta, in press. The authors thank Jerry Clark for helpful discussions and informing us of these new studies.
Aldrich Catalogue; Aldrich Chemical, Inc.: Milwaukee, WI, 2000.
Womack, E. B.; Nelson, A. B. Org. Syn. 1944,24, 56.
Lancaster Catalogue; Lancaster Synthesis, Inc.: Windham, NH, 2000.
Clark, J. D.; Shah, A. S.; Peterson, J. C.; Grogan, F. M.; Camden, S. K. Thermochim. Acta 2001,367/368, 75.
Unless otherwise said, all requisite enthalpies of formation in this paper are taken from Pedley, J. B.; Naylor, R. D.; Kirby, S. P. Thermochemical Data of Organic Compounds; Chapman & Hall: New York, 1986.
Lauer, A. H.; Okabe, H. J. Amer. Chem. Soc. 1971,93, 4137.
Paulett, G. S.; Ettinger, R. J. Chem. Phys. 1963,39, 825.
Wagman, D. D.; Evans, W. H.; Parker, V. B.; Schumm, R. H.; Halow, I.; Bailey, S. M.; Churney, K. L.; Nuttall, R. L. The NBS Tables of Chemical Thermodynamic Properties: Selected Values for Inorganic and C 1 and C 2 Organic Substances in SI Units, J. Phys. Chem. Ref. Data 1982,11, Suppl. 2.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hosmane, R.S., Liebman, J.F. Paradigms and Paradoxes: Diazomethane and Ethyl Diazoacetate: The Role of Substituent Effects on Stability. Structural Chemistry 13, 501–503 (2002). https://doi.org/10.1023/A:1020573723147
Issue Date:
DOI: https://doi.org/10.1023/A:1020573723147