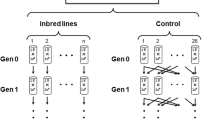
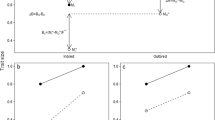
Abstract
Most mutations with observable phenotypic effects are deleterious. Studies of Drosophila and inbred plant populations suggest that a new individual may have a mean number of new deleterious mutations that exceeds one-half. Most of these have relatively small homozygous effects and reduce fitness by 1–2% when heterozygous. Several striking features of present-day organisms have apparently evolved in response to the constant input of deleterious alleles by recurrent mutation. For example, the adaptations of hermaphroditic organisms for outcrossing have been widely interpreted in terms of the benefits of avoiding the reduced fitness of inbred progeny, which is partly due to deleterious mutations. Population genetic models of modifiers of the breeding system in the presence of genome-wide deleterious mutation are reviewed and their predictions related to genetic and comparative data. The evolution of degenerate Y chromosomes is a phenomenon that may be caused by the accumulation of deleterious mutations. The population genetic mechanisms that can drive this degeneration are reviewed and their significance assessed in the light of available data.
Similar content being viewed by others
References
Aide, T.M., 1986. The influence of wind and animal pollination on variation in outcrossing rates. Evolution 40: 434-435.
Antonovics, J., 1968. Evolution in closely adjacent plant populations. V. Evolution of selffertility. Heredity 23: 219-238.
Ashburner, M., 1989. Drosophila. A Laboratory Handbook. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Baker, H.G., 1959. Reproductive methods as a factor in speciation in flowering plants. Cold Spr. Harb. Symp. Quant. Biol. 24: 9-24.
Barrett, S.C.H. & C.G. Eckert, 1990. Variation and evolution of plant mating systems, pp. 229-254 in biological approaches and evolutionary trends in plants, edited by S. Kawano. Academic Press, New York.
Barrett, S.C.H. & D. Charlesworth, 1991. Effects of a change in the level of inbreeding on the genetic load. Nature 352: 522-524.
Barrett, S.C.H. L.D. Harder & A.C. Worley, 1996. Comparative biology of plant reproductive traits. Phil. Trans. Roy. Soc. Lond. B 351: 1272-1280.
Barton, N.H., 1995. Linkage and the limits to natural selection. Genetics 140: 821-84.
Bawa, K.S., 1980. Evolution of dioecy in flowering plants. Ann. Rev. Ecol. Syst. 11: 15-39.
Berry, A.J.& M. Kreitman, 1993. Molecular analysis of an allozyme cline: alcohol dehydrogenase in Drosophila melanogaster on the East Coast of North America. Genetics 129: 869-893.
Berry, A.J., J.W. Ajioka & M. Kreitman, 1991. Lack of polymorphism on the Drosophila fourth chromosome resulting from selection. Genetics 129: 1111-1117.
Birky, C.W., 1988. Evolution and variation in plant chloroplast and mitochondrial DNA, pp. 23-53 in plant evolutionary biology, edited by L. D. Gottlieb & S.K. Jain. Chapman and Hall, London.
Birky, C.W. & J.B. Walsh, 1988. Effects of linkage on rates of molecular evolution. Proc. Natl. Acad. Sci. USA 85: 6414-6418.
Brunet, J., 1990. Gender specialization of flowers within inflorescences of hermaphroditic plants. Ph.D. Dissertation, State University of New York, Stony Brook.
Bryant, E.H., L.M. Meffert & L.M. McCommas, 1990. Fitness rebound in serially bottlenecked populations of the house fly. Amer. Nat. 136: 542-549.
Bull, J.J., 1983. Evolution of Sex Determining Mechanisms, Benjamin Cummings, Menlo Park, CA.
Campbell, R.B., 1986. The interdependence of mating structure and inbreeding depression. Theoret. Pop. Biol. 30: 232-244.
Campbell, D.R. & N.M. Waser, 1987. The evolution of plant mating systems: multilocus simulations of pollen dispersal. Amer. Nat. 129: 593-609.
Charlesworth, B., 1978. Model for evolution of Y chromosomes and dosage compensation. Proc. Natl. Acad. Sci. USA 75: 5618-5622.
Charlesworth, B., 1980. The cost of sex in relation to mating system. J. Theoret. Biol. 84: 655-671.
Charlesworth, B., 1987. The heritability of fitness, pp. 21-40 in Sexual Selection: testing the alternatives. Dahlem Conference, edited by J.W. Bradbury & M. Andersson. Springer Verlag, Berlin.
Charlesworth, B., 1990. Mutationselection balance and the evolutionary advantage of sex and recombination. Genet. Res. 55: 199-221.
Charlesworth, B., 1991. The evolution of sex chromosomes. Science 251: 1030-1033.
Charlesworth, B., 1994. The effect of background selection against deleterious alleles on weakly selected, linked variants. Genet. Res. 63: 213-228.
Charlesworth, B., 1996. The evolution of chromosomal sex determination and dosage compensation. Curr. Biol. 6: 149-162.
Charlesworth, B.,1998. The effect of synergistic epistasis on the inbreeding load. Genet. Res. (in press).
Charlesworth, B. & D. Charlesworth, 1997. Rapid fixation of deleterious alleles can be caused by Muller's ratchet. Genet. Res. 70: 63-73.
Charlesworth, B. & D.S. Guttman, 1996. Reductions in genetic variation in Drosophila and E. coli caused by selection at linked sites. J. Genet. 75: 49-61.
Charlesworth, B. & K.A. Hughes, 1996. Agespecific inbreeding depression and components of genetic variance in relation to the evolution of senescence. Proc. Natl. Acad. Sci. USA 93: 6140-6145.
Charlesworth, B. & K.A. Hughes, 1998. The maintenance of genetic variation in lifehistory traits, pp.xxx-xxx in evolutionary genetics from molecules to morphology, edited by Singh, R.S. & C.B. Krimbas. Cambridge University Press, Cambridge (in press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Charlesworth, B., Charlesworth, D. Some evolutionary consequences of deleterious mutations. Genetica 102, 3–19 (1998). https://doi.org/10.1023/A:1017066304739
Issue Date:
DOI: https://doi.org/10.1023/A:1017066304739