Abstract
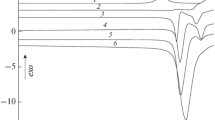
The compound 2-(2-benzofuryl) Δ-2 imidazoline, has been studied by DSC, TG, X-ray diffraction and thermomicroscopy.
We shall see that in the case of a study by DSC this compound presents a strange behaviour, which apparently is in contradiction with the thermodynamic rules. In a case of monotropy, if we have the α-phase (stable) and the γ-phase (metastable), after melting and cooling only theα-phase could crystallise. But this compound can give, according to the rate of cooling, theγ-phase metastable. The rate of cooling is of fundamental importance and the monotropic behaviour of this compound will be explained using the Gibbs function G=f(T) for P=1 atm.
Similar content being viewed by others
References
C. B. Chapleo, P. L. Myers, R. C. M. Butler, J. A. Davis, C. J. Coxey, S. D. Higgins, M. Myers, A. G. Roach, C. F. Smith, M. R. Stillings and A. P. Welbourn, J. Med. Chem., 27 (1984) 570.
M. Anastassiadou, S. Danoun, L. Crane, G. Baziard-Mouysset, M. Payard, D. H. Caignard, P. Renard and M. C. Rettori, Bioorganic and Medicinal Chem., 9 (2001) 585.
J. K. Haleblian, R. T. Koda and J. A. Biles, J. Pharm. Sci., 60 (1971) 1488.
Note of the Editor, Bull. Alloy Phases Diagrams, 7 (1986) 602.
Hultgren et al., 1973. 'Selected Values of the Thermodynamic Properties of the elements', A. S. M. Materials Park 1973.
B. Fraisse, 1985. Thèse Univ. Montpellier II.
G. M. Sheldrick, 1993. SHELXS93. Program for refinement of crystal structures. Univ. of Göttingen, Germany.
M. Volner and A. Weber, Z. Phys. Chem., 119 (1925) 277.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Legendre, B., Baziard-Mouysset, G., Anastassiadou, M. et al. Polymorphic Study of 2-(2-benzofuryl) Δ-2 Imidazoline. Journal of Thermal Analysis and Calorimetry 66, 659–673 (2001). https://doi.org/10.1023/A:1013102025734
Issue Date:
DOI: https://doi.org/10.1023/A:1013102025734