Abstract
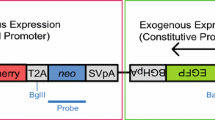
The efficiency of transgenic animal production would increase if microinjected embryos with a successfully integrated transgene could be identified prior to transfer. It is possible to detect microinjected DNA in embryos. However, no reliable system is able to distinguish between transgenes merely present as extrachromosomal DNA and those that have been integrated into chromatin. The experiments reported here were designed to determine if the inclusion of matrix attachment regions (MARs) would enhance the efficiency of transgenic embryos identification using a selection scheme based on the expression of green fluorescent protein (GFP). Pronuclei of mouse embryos were microinjected with GFP reporter gene under the control of three different promoters and flanked or not by three different MAR elements. Transgene expression profiles were followed by direct visualization of GFP in cultured microinjected embryos. Embryos at different developmental stages were classified according to their GFP expression and groups with the same expression pattern were transferred into oviducts of pseudopregnant female mice. Fetuses were collected between days 12–15, and their genomic DNA was purified and analyzed to detect transgene integration. We did not find any statistically significant difference between the percentage of transgenic fetuses produced from GFP-positive or GFP-negative embryos transferred at 4-cell, morula, or blastocyst stage. However, when MAR elements were included in the construct, we found that GFP-positive embryos transferred at the 2-cell stage produced a significantly higher percentage of transgenic fetuses than GFP-negative embryos, but MAR sequences did not completely eliminate false positives.
Article PDF
Similar content being viewed by others
References
Baskar JF, Smith PP, Nilaver G, Jupp RA, Hoffmann S, Peffer NJ, Tenney DJ, Colberg-Poley AM, Ghazal P and Nelson JA (1996) The enhancer domain of the human cytomegalovirus major immediate-early promoter determines cell type-specific expression in transgenic mice. J Virol 70: 3207–3214.
Bishop JO (1996) Chromosomal insertion of foreign DNA. Reprod Nutr Dev 36: 607–618.
Bode J and Maass K (1988) Chromatin domain surrounding the human interferon-beta gene as defined by scaffold-attached regions. Biochemistry 27: 4706–4711.
Bokman SH and Ward WW (1981) Renaturation of Aequorea green fluorescent protein. Biochem Biophys Res Commun 101: 1372–1380.
Bondioli KR and Wall RJ (1996) Positive selection of bovine embryos in culture. Theriogenolgy 45: 345.
Burdon TG and Wall RJ (1992) Fate of microinjected genes in preimplantation mouse embryos. Mol Reprod Dev 33: 436–442.
Erbach GT, Lawitts JA, Papaioannous VE and Biggers JD (1994) Differential growth of the mouse preimplantation embryo in chemically defined media. Biol Reprod 50: 1027–1033.
Fink PS (1991) Using sodium chloride step gradients to fractionate DNA fragments. BioTechniques 10: 437–440.
Gordon J, Scangos G, Plotkin D, Barbosa J and Ruddle F (1980) Genetic transformation of mouse embryos by microinjection of purified DNA. Proc Natl Acad Sci USA 77: 7380–7384.
Horvat S, Medrano JF, Behboodi E, Anderson GB and Murray JD (1993) Sexing and detection of gene construct in microinjected bovine blastocysts using the polymerase chain reaction. Transgenic Res 2: 134–140.
Kalos M and Fournier REK (1995) Position-independent transgene expression mediated by boundary elements from the apolipoprotein B chromatin domain. Mol Cellular Biol 15: 189–207.
Kim DW, Uetsuki T, Kaziro Y, Yamaguchi N and Sugano S (1990) Use of the human elongation factor 1α promoter as a versatile and efficient expression system. Gene 91: 217–223.
Klehr D, Maass K and Bode J (1991) Scaffold-attached regions from the human interferon (domain can be used to enhance the stable expression of genes under the control of various promoters. Biochemistry 30: 1264–1270.
Menck MC, Mercier Y, Campion E, Lobo RB, Heyman Y, Renard JP and Thompson EM (1998) Prediction of transgene integration by noninvasive bioluminiscent screening of microinjected bovine embryos. Transgenic Res 7: 331–341.
Nakamura A, Okumura J-I and Muramatsu T (1998) Quantitative analysis of luciferase activity of viral and hybrid promoters in bovine preimplantation embryos. Mol Reprod Dev 49: 368–373.
Poljak L, Seum C, Mattioni T and Laemmli U (1994) SARs stimulate but do not confer position independent gene expression. Nucleic Acids Res 22: 4386–4394.
Powell AM, Bondioli KR and Rexroad CE (1996) The sheep uterus as host for IVM-IVF bovine embryos from day 7– day 15. Theriogenology 45: 217.
Pursel VG and Rexroad CE (1993) Status of research with transgenic farm animals. J Anim Sci 71(3): 10–15.
Schnieke AE, Kind AJ, Ritchie KM, Scott AR, Ritchie M, Wilmut I, Colman A and Campbell HS (1997) Human factor IX transgenic sheep produced by transfer of nuclei from transfected fetal fibroblasts. Science 278: 2130–2133.
Stief A, Winter DM, Stratling WH and Sippel AE (1989) A nuclear DNA attachment element mediates elevated and position-independent gene activity. Nature 341: 343–345.
Tada N, Sato M, Hayashi K, Kasai K and Ogawa S (1995) In vitro selections of transgenic mouse embryos in the presence of G-418. Transgenics 1: 535–540.
Takada T, Lida K, Awaji T, Itoh K, Takahashi R, Shibui A, Yoshida K, Sugano S, and Tsujimoto G (1997). Selective production of transgenic mice using green fluorescent protein as a marker. Nature Biotech 15: 458–461.
Thompson EM, Adenot P, Tsuji FI and Renard J-P (1995) Real time imaging of transcriptional activity in live mouse preimplantation embryos using a secreted luciferase. Proc Natl Acad Sci USA 92: 1317–1321.
Thompson EM, Christians E, Stinnakre M-G and Renard J-P (1994) Scaffold attachment regions stimulate HSP70.1 expression in mouse preimplantation embryos but not in differentiated tissues. Mol Cellular Biol 14: 4694–4703.
Wall RJ (1996) Transgenic livestock: progress and prospects for the future. Theriogenology 46: 57–68.
Wang D-M, Taylor S and Levy-Wilson B (1996) Evaluation of the function of the human apolipoprotein B gene nuclear matrix association regions in transgenic mice. J Lipid Res 37: 2117–2124.
Whitelaw CBA, Springbett AJ, Webster J, Clark AJ (1993) The majority of G0 transgenic mice are derived from mosaic embryos. Transgenic Res 2: 29–32.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gutiérrez-Adán, A. Effect of flanking matrix attachment regions on the expression of microinjected transgenes during preimplantation development of mouse embryos. Transgenic Res 9, 81–89 (2000). https://doi.org/10.1023/A:1008926022370
Issue Date:
DOI: https://doi.org/10.1023/A:1008926022370