Abstract
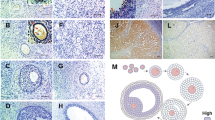
Previously, we prepared an IgM monoclonal antibody(PFG-1) which specifically recognized a cell-membraneglycoprotein (PFG-1 antigen; 55 kD, pI 5.9),immunohistochemically reacted with granulosa cells ofhealthy follicles but not of atretic follicles, andinduced granulosa cell apoptosis. In the presentstudy, an IgM monoclonal antibody (PFG-3) capable ofinducing granulosa cell apoptosis and an IgGmonoclonal antibody (PFG-4) not capable of inducingapoptosis were produced against granulosa cellsprepared from healthy antral follicles of porcineovaries. Two-dimensional Western blotting analysisrevealed that PFG-3 specifically recognized twocell-membrane proteins (named PFG-3-1 andPFG-3-2/PFG-1 antigens; 42 kD, pI 5.2 and 55 kD, pI5.9, respectively) of healthy granulosa cells, andthat PFG-4 recognized the same two cell-membraneproteins. In atretic granulosa cells, PFG-3-2/PFG-1antigen disappeared. Immunochemical reactions of theseantibodies were only detected in follicular granulosacells but not any other ovarian tissues or organs.PFG-3 and PFG-4 immunohistochemically reacted withgranulosa cells of healthy and atretic follicles. Whenthe isolated granulosa cells prepared from healthyfollicles were cultured in medium containing PFG-3,the cells underwent apoptosis, and co-incubation withPFG-4 inhibited PFG-3-inducible apoptosis. Theseobservations suggested that PFG-3-2/PFG-1 antigen isa novel cell death receptor which is different fromthe apoptosis-mediating receptors (Fas/Apo-1/CD95 orTNF receptor), and that PFG-3-1 antigen may act as adecoy receptor and inhibit apoptotic signal transmission.
Similar content being viewed by others
References
Babaloa GO and Shapiro BH (1988) Correlation of follicular steroid hormone profiles with ovarian cyclicity in sows. J Reprod Fertil 84: 79–87.
Coley AJ, Howard HJ, Slanger WD and Ford JJ (1994) Steroidogenesis in the preovulatory porcine follicle. Biol Reprod 51: 655–661.
Dailey RA, Clark JR, First NL, Chapman AB and Casida LE (1975) Loss of follicles during the follicular phase of the estrous cycle of swine as affected by genetic group and level of feed intake. J Anim Sci 41: 835–841.
Emery JG, McDonnell P, Burke MB, Deen KC, Lyn S, Silverman C, Dul E, Appelbaum ER, Eichman C, Diprinzio R, Dodds RA, James IE, Rosenberg M, Lee JC and Young PR (1998) Osteoprotegerin is a receptor for the cytotoxic ligand TRAIL. J Biol Chem 273: 14363–14367.
Flemming W (1885) Ñber die Bildung von Richtungsfiguren in Säugethiereiern beim Untergang Graaf'scher Follikel. Arch Anat EntGesch 221–244.
Guthrie HD, Cooper BS, Welch GR, Zakaria AD and Johnson LA (1995) Atresia in follicles grown after ovulation in the pig: Measurement of increased apoptosis in granulosa cells and reduced follicular fluid estradiol-17β. Biol Reprod 52: 920–927.
Hakuno N, Koji T, Yano T, Kobayashi N, Tsutsumi O, Taketani Y and Nakane PK (1996) Fas/APO-1/CD95 system as a mediator of granulosa cell apoptosis in ovarian follicle atresia. Endocrinology, 137: 1938–1948.
Hirshfield AN (1989) Rescue of atretic follicles in vitro and in vivo. Biol Reprod 40: 181–190.
Hirshfield AN (1991) Development of follicles in the mammalian ovary. Int Rev Cytol 124: 43–101.
Hughes Jr FM and Gorospe WC (1991) Biochemical identification of apoptosis (programmed cell death) in granulosa cells: evidence for a potential mechanism underlying follicular atresia. Endocrinology 129: 2799–2801.
Imai Y, Manabe N, Uchio K, Kinoshita A, Kimura Y, Nakayama M, Nishihara S, Wada S, Sugimoto M and Miyamoto H (1999) Interactions of apoptosis and extracellular matrices in granulose cells of atretic follicles in porcine ovaries. J Mamm Ova Res 16: 59–66.
Itoh N, Yonehara S, Ishii A, Yonehara M, Mizushima S-I, Sameshima M, Hase A, Seto Y and Nagata S (1991) The polypeptide encoded by the cDNA for human cell surface antigen Fas can mediate apoptosis. Cell 66: 233–243.
Kapuscinski J and Skoczylas B (1977) Simple and rapid fluorimetric method for DNA microassay. Anal Biochem 83: 252–257.
Kerr JFR, Wyllie AH and Currie AR (1972) Apoptosis: A basic biological phenomenon with wide ranging implication in tissue kinetics. Br J Cancer 26: 239–257.
Kim JM, Boone DL, Auyeung A and Tsang BK (1998) Granulosa cell apoptosis induced at the penultimate stage of follicular development is associated with increased levels of Fas and Fas ligand in the rat ovary. Biol Reprod 58: 1170–1176.
Kimura Y, Manabe N, Matsushita H, Tajima C, Myoumoto A and Miyamoto H (1998) Examination of granulosa cell glycoconjugates which change during follicular atresia in the pig ovaries. J Reprod Dev 44: 35–44.
Kimura Y, Manabe N, Nishihara S, Matsushita H, Tajima C, Wada S and Miyamoto H (1999) Up-regulation of the α2,6-sialyltransferase messenger ribonucleic acid increases glycoconjugates containing α2,6-linked sialic acid residues in granulose cells during follicular atresia of porcine ovaries. Biol Reprod 60: 1475–1482.
Macfarlane M, Ahmad M, Srinivasula SM, Fernandes AT, Cohen GM, Alnemri ES (1997) Identification and molecular cloning of two novel receptors for the cytotoxic ligand TRAIL. J Biol Chem 272: 25417–25420.
Manabe N, Imai Y, Ohno H, Takahagi Y, Sugimoto M and Miyamoto H (1996a) Apoptosis occurs in granulosa cells but not cumulus cells in the atretic antral follicles in the pig ovaries. Experientia 52: 647–651.
Manabe N, Imai Y, Kimura Y, Myoumoto A, Sugimoto M, Miyamoto H, Okamura Y and Fukumoto M (1996b) Ca2+/Mg2+-dependent endonuclease but not Ca2+-dependent, Mg2+-dependent or cation-independent endonuclease is involved in granulosa cell apoptosis of pig atretic follicles. J Reprod Dev 42: 247–253.
Manabe N, Imai Y, Kimura Y, Myoumoto A, Sugimoto M, Miyamoto H, Okamura Y and Fukumoto Y (1996c) Signal transmission of granulosa cell apoptosis in the atretic follicles in the pig ovaries. J Reprod Dev 42: 135–141.
Manabe N, Kimura Y, Myoumoto A, Imai Y, Sakamaki K, Sugimoto M and Miyamoto H (1997a) Apoptosis occurs in granulosa cells but not cumulus cells in the atretic antral follicles in the porcine ovaries. J Reprod Dev 43: 179–180.
Manabe N, Imai Y, Myoumoto A, Kimura Y, Sugimoto M, Okamura Y, Fukumoto M, Sakamaki K and Miyamoto H (1997b) Apoptosis occurs in granulosa cells but not cumulus cells in the atretic graafian follicles in multiparous pig ovaries. Acta Histochem Cytochem 30: 85–92.
Manabe N, Myoumoto A, Kimura Y, Imai Y, Sugimoto M and Miyamoto H (1997c) A monoclonal antibody against pig ovarian follicular granulosa cells recognizes a novel cell death receptor and induces granulosa cell apoptosis. In: Kawashima S (ed.) Advances in Comparative Endocrinology (pp. 1793–1797). Monduzzi, Editore Spa, Bologna.
Manabe N, Myoumoto A, Kimura Y, Imai Y, Sugimoto M, Sakamaki K, Okamura Y, Fukumoto M and Miyamoto H (1998a) Regulatory mechanisms of granulosa cell apoptosis in porcine ovarian follicle atresia. In: Miyamoto H and Manabe N (eds) Reproductive Biology Update (pp. 23–35). Nakanishi Pub Co, Kyoto.
Manabe N, Kimura Y, Myoumoto A, Matsushita H, Tajima C, Sugimoto M and Miyamoto H (1998b) Role of granulosa cell apoptosis in ovarian follicle atresia. In: Yamada T and Hashimoyto Y (eds) Apoptosis: Its Roles and Mechanism (pp. 97–111). Jpn Acad Soc Book Cent, Tokyo.
Marsters SA, Sheridan JP, Pitti RM, Huang A, Skubatch M, Baldwin D, Yuan J, Gurney A, Goddard AD, Godowski P and Ashkenazi A (1997) A novel receptor for Apo2L/TRAIL contains a truncated death domain. Cur Biol 7: 1003–1006.
Murdoch WJ (1995) Immunolocalization of a gonadotropinreleasing hormone receptor site in murine endometrium that mediates apoptosis. Cell Tissue Res 282: 527–529.
Myoumoto A, Manabe N, Imai Y, Kimura Y, Sugimoto M, Okamura Y, Fukumoto M, Sakamaki K, Yonehara S, Niwano Y and Miyamoto H (1997) Monoclonal antibodies against pig ovarian follicular granulosa cells induce apoptotic cell death in cultured granulosa cells. J Vet Med Sci 59: 641–649.
O'Farrell PH (1975) High resolution two-dimentional electrophoresis of proteins. J Biol Chem 250: 4007–4021.
Ogasawara J, Watanabe-Fukunaga R, Adachi M, Matsuzawa A, Kasugai T, Kitamura Y, Itoh N, Suda T and Nagata S (1993) Lethal effect of the anti-Fas antibody in mice. Nature 364: 806–809.
Oi VT and Herzenberg LA (1980) Immunoglobulin producing hybrid cell lines. In: Mishell BB and Shiiji SM (eds) Selected Methods in Cellular Immunology (pp. 351–372). Freeman Pub, San Francisco.
Pan G, Ni J, Wei YF, Yu GL, Gentz RA and Dixit VM (1997) An antagonist decoy receptor and a death domain-containing receptor for TRAIL. Science 277: 815–818.
Quirk SM, Cowan RG, Huber SH (1997) Fas antigen-mediated apoptosis of ovarian surface epithelial cells. Endocrinology 138: 4558–4566.
Rieger J, Naumann U, Glaser T, Ashkenazi A and Weller M (1998) APO2 ligand: A novel lethal weapon against malignant glioma? FEBS Lett 427: 124–128.
Sakamaki K, Yoshida H, Nishimura Y, Nishikawa SI, Manabe N and Yonehara S (1997) Involvement of Fas antigen in ovarian follicular atresia and leuteolysis. Mol Reprod Dev 47: 11–18.
Sheikh MS, Burns TF, Huang Y, Wu GS, Amundson S, Brooks KS, Fornace Jr AJ and El Deiry WS (1998) p53-dependent and-independent regulation of the death receptor Killer/DR5 gene expression in response to genotoxic stress and tumor necrosis factor alpha. Cancer Res 58: 1593–1598.
Sheridan JP, Marsters SA, Pitti RM, Gurney A, Skubatch M, Baldwin D, Ramakrishnan L, Gray CL, Baker K, Wood WI, Goddard AD, Godowski P and Ashkenazi A (1997) Control of TRAIL induced apoptosis by a family of signaling and decoy receptors. Science 277: 818–821.
Stauber GB, Aiyer RA and Aggatwal BB (1988) Human tumor necrosis factor-a receptor: Purification by immunoaffinity chromatography and initial characterization. J Biol Chem 263: 190–198.
Sugimoto M, Manabe N, Kimura Y, Myomoto A, Imai Y, Ohno H and Miyamoto H (1998) Ultrastructural changes in granulosa cells in porcine antral follicles undergoing atresia indicate apoptotic cell death. J Reprod Dev 44: 7–14.
Tilly JL, Kowalski KI, Johnson AL and Hsueh AJW (1991) Involvement of apoptosis in ovarian follicular atresia and postovulatory regression. Endocrinology 129: 2799–2801.
Tilly JL and Hsueh AJW (1992) Apoptosis as the basis of ovarian follicular atresia. In: Hillier SG (ed.) Gonadal Development and Function (pp. 157–165). Raven Press, New York.
Yamada T, Horiuchi M and Dzau VJSO (1996) Angiotensin II type 2 receptor mediates programmed cell death. Prod Natl Acad Sci USA 93: 156–160.
Yonehara S, Ishii A and Yonehara M (1989) A cell-killing monoclonal antibody (Anti-Fas) to a cell surface antigen co-downregulated with the receptor of tumor necrosis factor. J Exp Med 169: 1747–1756.
Yoo J, Stone RT and Beattie CW (1996) Cloning and characterization of the bovine Fas. DNA Cell Biol 15: 227–234.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Manabe, N., Myoumoto, A., Tajima, C. et al. Immunochemical characteristics of a novel cell death receptor and a decoy receptor on granulosa cells of porcine ovarian follicles. Cytotechnology 33, 189–201 (2000). https://doi.org/10.1023/A:1008146119761
Issue Date:
DOI: https://doi.org/10.1023/A:1008146119761