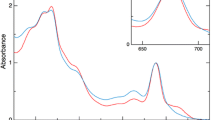
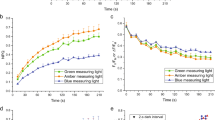
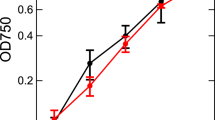
Abstract
When far red light preincubated cells of Euglena gracilis are transferred to dark or light, chlorophyll fluorescence (F0 and Fm) decreases. Non-photochemical quenching in the dark is suggested to be induced partly by chlororespiration and partly by changes in the distribution of excitation energy between the photosystems. Depending on the light intensities it was possible to resolve the non-photochemical quenching into at least three different components. The slowest relaxation phase of non-photochemical quenching occurred only after exposure to high light and was assigned to photoinhibition. The other two components were an energy-dependent quenching (qE), and the one which we attribute to a spill over mechanism. We suggest that both photosystems use a common antenna system consisting of LHC I and LHC II proteins. In contrast to higher plants, qE in Euglena gracilis is independent of the xanthophyll cycle and an aggregation of LHC II.
Similar content being viewed by others
References
Allen JF (1992) Protein phosphorylation in regulation of photosynthesis. Biochim Biophys Acta 1098: 275–335
Anderson JM (1980) P-700 content and polypeptides profile of chlorophyll complexes of spinach and barley thylakoids. Biochim Biophys Acta 591: 113–126
Barber J and De Las Rivas P (1993) A functional model for the role of cytochrome b595 in the protection against donor and acceptor side photoinhibition. Proc Natl Acad Sci USA 90: 10942–10946
Bennoun P (1982) Evidence for a respiratory chain in the chloroplast. Proc Natl Acad Sci USA 79: 4352–4356
Britton G (1985) General carotenoid methods. Methods Enzymol 111: 113–149
Brown J (1980) Absorption and fluorescence spectra of chlorophyllproteins isolated from Euglena gracilis. Biochim Biophys Acta 591: 9–21
Brown J and Michel-Wolwertz MR (1968) Chlorophyll fluorescence near 720 mμ in Euglena extracts. Biochim Biophys Acta 153: 288–290
Bulté L, Gans P, Rébeillé F and Wollmann FA (1990) ATP control on state transitions in vivo in Chlamydomonas reinhardtii. Biochim Biophys Acta 1020: 72–80
Casper-Lindley C and Björkman O (1998) Fluorescence quenching in four unicellular algae with different light-harvesting and xanthophyll-cycle pigments. Photosynth Res 56: 277–289
Cunningham Jr FX and Schiff JA (1986a) Chlorophyll-protein complexes from Euglena gracilis and mutants deficient in chlorophyll b. 1. Pigment composition. Plant Physiol 80: 223–230
Cunningham Jr FX and Schiff JA (1986b) Chlorophyll-protein complexes from Euglena gracilis and mutants deficient in chlorophyll b. 2. Polypeptide composition. Plant Physiol 80: 231–238
Delphin E, Duval JC, Etienne AL and Kirilovsky D (1996) State transitions or 1pH-dependent quenching of Photosystem II fluorescence in red algae. Biochemistry 35: 9435–9445
Demmig-Adams B (1990) Carotenoids and photoprotection in plants: A role for the xanthophyll zeaxanthin. Biochim Biophys Acta 1020: 1–24
Dreyfuss BW and Thornber JP (1994) Assembly of the lightharvesting complexes (LHCs) of Photosystem II. Plant Physiol 106: 829–839
Endo T and Asada K (1996) Dark induction of the nonphotochemical quenching of chlorophyll fluorescence by acetate in Chlamydomonas reinhardtii. Plant Cell Physiol 37: 551–555
Frank HA, Cua A, Chynwat V, Young A, Gosztola D and Wasielewski MR (1996) The lifetime and energies of the excited singlet states of diadinoxanthin and diatoxanthin: the role of these molecules in excess energy dissipation in algae. Biochim Biophys Acta 1277: 243–252
Gilmore AM and Yamamoto HY (1991) Zeaxanthin formation and energy-dependent fluorescence quenching in pea chloroplasts under artificially mediated linear and cyclic electron transport. Plant Physiol 96: 635–643
Gilmore AM and Yamamoto HY (1993) Linear models relating xanthophyll and lumen acidity to non-photochemical fluorescence quenching. Evidence that antheraxanthin explain zeaxanthin independent quenching. Photosynth Res 35: 67–78
Goss R, Böhme K and Wilhelm C (1998) The xanthophyll cycle of Mantoniella squamata converts violaxanthin into antheraxanthin but not to zeaxanthin: Consequences for the mechanism of enhanced non-photochemical energy dissipation. Planta 205, 613–621
Green BR and Durnford DG (1996) The chlorophyll-carotenoid proteins of oxygenic photo-synthesis. Annu Rev Plant Physiol Mol Biol 47: 685–714
Hager A (1980) The reversible, light-induced conversion of xanthophylls in the chloroplast. In: Czygan F-C (ed) Pigments in Plants, pp 57–79. Akademie-Verlag Berlin
Harrison MA and Allen JF (1993) Differential phosphorylation of individual LHC-II polypeptides during short-term and long-term acclimation to light regime in the green alga Dunaliella salina. Biochim Biophys Acta 1141: 37–44
Horton P, Ruban AV and Walters RG (1996) Regulation of light harvesting in green plants. Annu Rev Plant Physiol Plant Mol Biol 47: 655–684
Horton P, Ruban AV, Rees D, Pascal AA, Noctor G and Young AJ (1991) Control of the light-harvesting function of chloroplast membranes by aggregation of the LHCII chlorophyll-protein complex. FEBS Lett 292: 1–4
Houlné G and Schantz R (1988) Characterization of cDNA sequences for LHCI apoproteins in Euglena gracilis The mRNA encodes a large precursor containing several consecutive divergent polypeptides. Mol Gen Genet 213: 479–486
Jahns P and Krause GH (1994) Xanthophyll cycle and energydependent fluorescence quenching in leaves from pea plants grown under intermittent light. Planta 192: 176–182
Kooten van O and Snel JFH (1990) The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth Res 25: 147–150
Kramer HJM, Westerhuis WHJ and Amesz J (1985) Low temperature spectroscopy of intact algae. Physiol Veg 23: 534–543
Krieger A and Weis E (1992) Energy dependent quenching of chlorophyll-a-fluorescence. The involvement of proton-calcium exchange at Photosystem II. Photosynthetica 27: 89–98
Krieger A, Moya I and Weis E (1992) Energy dependent quenching of chlorophyll a fluorescence: Effect of chlorophyll a fluorescence and picosecond-relaxation kinetics in thylakoid membranes. Biochim Biophys Acta 1102: 167–176
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Lokstein H, Härtel H and Hoffmann P (1993) Comparison of chlorophyll fluorescence quenching in leaves of wild-type with a chlorophyll-b-mutant of barley (Hordeum vulgare L.). J Photochem Photobiol B: Biol 19: 217–225
Mantoura RFC and Llewellyn CA (1983) The rapid determination of alga chlorophyll and carotenoid pigments and their breakdown products in natural waters by reverse-phase high performance liquid chromatography. Analytica Chimica Acta 151: 297–314
Muchhal U and Schwartzbach SD (1992) Characterization of an Euglena gene encoding a polyprotein precursor to the light harvesting chlorophyll a/b binding protein of Photosystem II. Plant Mol Biol 18: 287–299
Olaizola M and Yamamoto HY (1994) Short-term response of the diadinoxanthin cycle and fluorescence yield of high irradiance in Chaetoceros muelleri (Bacillariophyceae). J Phycol 30: 606–612
Olaizola M, La Roche J, Kolber Z and Falkowski PG (1994) Nonphotochemical fluorescence quenching and the diadinoxanthin cycle in a marine diatom. Photosynth Res 41: 357–370
Peltier G, Ravenel J and Verméglio A (1987) Inhibition of a respiratory activity by short saturating flashes in Chlamydomonas: evidence for a chlororespiration. Biochim Biophys Acta 893: 83–90
Pfündel E and Bilger W (1994) Regulation and possible function of violaxanthin cycle. Photosynth Res 42: 89–109
Pogson BJ, Niyogi KK, Björkman O and DellaPenna D (1998) Altered xanthophyll compositions adversely affect chlorophyll accumulation and nonphotochemical quenching in Arabidopsis mutants. Proc Natl Acad Sci USA 95: 13324–13329
Pospišil P (1997) Mechanisms of non-photochemical chlorophyll fluorescence quenching in higher plants. Photosynthetica 34: 343–355
Pringsheim EG and Wiessner W (1961) Ernährung and Stoffwechsel von Chlamydobotrys (Volvocales). Arch Mikrobiol 40: 231–244
Quick WP and Stitt M (1989) An examination of factors contributing to non-photochemical quenching of chlorophyll fluorescence in barley leaves. Biochim Biophys Acta 977: 287–296
Ruban AV and Horton P (1992) Mechanism of 1pH-dependent dissipation of absorbed excitation energy by photosynthetic membranes I: Spectroscopic analysis of isolated light-harvesting complexes. Biochim Biophys Acta 1102: 30–38
Ruban AV and Horton P (1995) Regulation of non-photochemical quenching of chlorophyll fluorescence in plants. Aust J Plant Physiol 22: 221–230
Salvador G, Lefort-Tran M, Nigon V and Jourdan F (1971) Structure et évolution du corps prolamellaire dans les proplastes d'Euglena gracilis. Exp Cell Res 64: 457–462
Schiller H, Hühn M and Dau H (1998) On the long-wavelength spectral forms of chlorophyll a in Photosystem I: Spectroscopic and immunological investigation on a greening mutants of the green alga Scenedesmus obliquus. Photosynth Res 55: 95–107
Schreiber U, Endo T, Mi H and Asada K (1995) Quenching analysis of chlorophyll fluorescence by the saturation pulse methods: particular aspects relating to the study of eukaryotic algae and cyanobacteria. Plant Cell Physiol 36: 873–882
Siefermann-Harms D (1988) Fluorescence properties of isolated chlorophyll-protein complexes. In: Lichtenthaler (ed) Application of Chlorophyll Fluorescence, pp 45–53. Kluwer Academic Publishers, Dordrecht, The Netherlands
Thiele A, Winter K and Krause GH (1997) Low inactivation of D1 protein of Photosystem II in young canopy leaves of Anacardium excelsum under high-light stress. J Plant Physiol 151: 286–292
Ting CS and Owens TG (1993) Photochemical and nonphotochemical fluorescence quenching processes in the diatom Phaeodactylum tricornutum. Plant Physiol 101: 1323–1330
Walters RG and Horton P (1991) Resolution of components of non-photochemical chlorophyll fluorescence quenching in barley leaves. Photosynth Res 27: 121–133
Weis E and Berry JA (1987) Quantum efficiency of Photosystem II in relation to ‘energy’-dependent quenching of chlorophyll fluorescence. Biochim Biophys Acta 894: 198–208
Wilhelm C and Duval JC (1990) Fluorescence induction kinetcs as a tool to detect a chlororespiratory activity in the prasinophycean alga Mantoniella squamata. Biochim Biophys Acta 1016: 197–202
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doege, M., Ohmann, E. & Tschiersch, H. Chlorophyll fluorescence quenching in the alga Euglena gracilis. Photosynthesis Research 63, 159–170 (2000). https://doi.org/10.1023/A:1006356421477
Issue Date:
DOI: https://doi.org/10.1023/A:1006356421477