Abstract
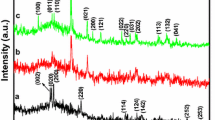
Uniform crystals of ZnWO4 have been synthesised from the equimolar mixtures of ZnO and WO3 by conventional solid state method. For the first time the sample has been characterised detailedly to confirm the formation of pure single phase of perovskite ZnWO4. The formation of ZnWO4 has been confirmed by sintering the mixtures of ZnO and WO3 at two different temperatures one at 900 °C and other at 1000 °C. It is observed that the sample sintered at 1000 °C for 24 h shows complete formation of the single phase of ZnWO4. The crystallinity and the phase formation has been confirmed by X-ray diffraction technique. X-ray photoelectron spectroscopy measurements have been carried out for the bulk ZnWO4 sintered at 1000 °C for 24 h, showing 16% of Zn, 16% of W and 68% of O indicating stoichiometric ZnWO4. Surface morphology studies by scanning electron microscopy showed uniform crystals of ZnWO4. The purity of the compound has also been checked in depth by Energy Dispersive X-ray method indicating the absence of foreign ions apart from that, the ratio of Zn : W has been calculated and found to be 1 : 1 confirming the stoichiometric ZnWO4 inside the crystals.
Similar content being viewed by others
References
M. Furuya and A. Ochi, Jpn. J. Appl. Phys. 33 (1994) 5482.
T. S. Rao, V. R. K. Murthy and B. Viswanathan, Ferroelectrics 102 (1990) 155.
K. Wakino, ibid. 91 (1989) 69.
B. C. H. Steele, “Electronic Ceramics” (Elsevier Applied Science, London and New York, 1991).
V. L. Gurevich and A. K. Tagantsev, Sov. Phys. JETP 64 (1986) 142.
D. A. Sagala and S. Nambu,J. Am. Ceram. Soc. 75 (1992) 2573.
B. D. Silverman, Phys. Rev. 125 (1962) 1921.
K. Matsumoto, T. Hiuga, K. Takada and H. Ichimura, in Proc. 6th IEEE Int. Symp. Appl. Ferroelectrics (1986) p. 118.
M. P. Harmer, J. Chen, P. Peng, H. M. Chan and D. M. Smyth, Ferroelectrics 97 (1989) 263.
C. H. Lu and C. C. Tsai, J. Mater. Res. 11 (1996) 1219.
X. M. Chen, Y. Suzuki and N. Sato, J. Mater. Sci. 5 (1994) 244.
S. Nomura, Ferroelectrics 49 (1983) 61.
K. H. Yoon, D. P. Kim and E. S. Kim,J. Am. Ceram. Soc. 77 (1994) 1062.
F. Izumi, in “The Rietveld Method,” edited by R. A. Young (Oxford University Press, New York, 1993) p. 236.
F. P. J. M. Kerkhof and J. A. Moulijn, J. Electron Spectroscopy and Related Phenomena 14 (1978) 453.
D. A. Sagala and S. Nambu, J. Phys. Soc. Jpn. 61 (1992) 1791.
K.-M. Lee, H. M. Jang and W.-J. Park, J. Mater. Res. 12 (1997) 1603.
K.-M. Lee and H. M. Jang, J. Am. Ceram. Soc. 81 (1998) 2586.
D. Briggs and M. P. Seah, “Auger and X-ray Photoelectron Spectroscopy” (John Wiley and Sons, 1990).
R. J. Colton and J. W. Rabais, Inorg. Chem. 15 (1976) 236.
F. Izumi, M. Mitomo and Y. Bando, J. Mater. Sci. 19 (1984) 3115.
R. A. Young and D. B. Wiles, J. Appl. Cryst. 15 (1982) 435.
R. D. Shannon and C. T. Prewitt, Acta Cryst. B25 (1969) 925.
W. Piece and A. Weiss, in “Landolt-Börnstein-Numerical Data and Functional Relationships in Science and Technology, New Series,” edited by K. H. Helwege and A. M. Helwege (Springer-Verlag,1973), Group III: Crystal and Solid State Physics. 7 [e].
G. V. Samsonov, “The Oxide Handbook” (IFI/PLENUM, 1982).
N.-H. Chan, R. K. Sharma and D. M. Smyth, J. Am. Ceram. Soc. 65 (1982) 167.
I. M. Reaney, J. Petzelt, V. V. Voitsekhovskii, F. Chu and N. Setter, J. Appl. Phys. 76 (1994) 2086.
C. A. Randall, D. J. Barber, R. W. Whatmore and P. Groves, J. Mater. Sci. 21 (1986) 4456.
G. Koschek and E. Kubalek,J. Am. Ceram. Soc. 68 (1985) 582.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Phani, A.R., Passacantando, M., Lozzi, L. et al. Structural characterization of bulk ZnWO4 prepared by solid state method. Journal of Materials Science 35, 4879–4883 (2000). https://doi.org/10.1023/A:1004809804206
Issue Date:
DOI: https://doi.org/10.1023/A:1004809804206