Abstract
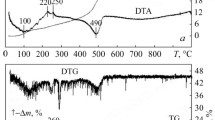
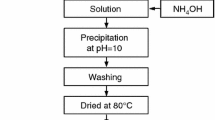
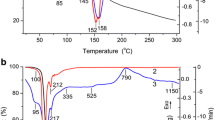
Alumina was prepared from an aqueous salt solution by homogeneous precipitation followed by calcination in air. Dependence of the thermal stability of transition phases on the presence of a zirconia dopant and on autoclave treatment prior to calcination was investigated using X-ray diffraction (XRD), differential thermal analysis coupled with thermogravimetric analysis (DTA–TGA) and transmission electron microscope (TEM) analysis. Homogeneous precipitation produced an amorphous trihydrate precipitate; the autoclave treatment converted this to crystalline boehmite (monohydrate). The zirconia was soluble in the transition alumina but was insoluble in α-Al2O3 so that phase transformation to α-Al2O3 was accompanied by a phase separation to form an alumina-zirconia nanocomposite. The thermal stability of the transition phases was increased both by the dopant and by the autoclave treatment. A combination of both parameters yielded the most stable transition alumina, which withstood 1 h at 1200°C without transformation to α-Al2O3.
Similar content being viewed by others
References
H. C. STUMPF, A. S. RUSSELL, J. W. NEWSOME and C. M. TUCKER, Ind. Engng. Chem. 42 (1950) 1398.
G. ERVIN, Jr, Acta Crystallogr. 5 (1952) 103.
H. SAALFELD, Clay Min. Bull. 3 (1985) 249.
A. J. LEONARD, F. VAN CAUWELAERT and J. J. FRIPIAT, J. Phys. Chem. 71 (1967) 695.
P. A. BADKAR and J. E. BAILEY, J. Mater. Sci. 11 (1976) 1794.
S. J. WILSON, Proc. Brit. Ceram. Soc. 28 (1979) 281.
S. J. WILSON and J. D. C. McCONNELL, J. Solid State Chem. 34 (1980) 315.
H. S. SANTOS, P. K. KIYOHARA and P. S. SANTOS, Ceram. Int. 20 (1994) 175.
T. FUKUI and M. HORI, J. Mater. Sci. Lett. 13 (1994) 413.
A. UENO, in Proceeding of the 11th Symposium on Catalytic Combustion, Tokyo, May 1991, (1991) p. 1–8.
Y. MIZUSHIMA and M. HORI, in Proceedings of the Eurogel ’91 Conference, edited by S. Vilminot, R. Nass and H. Schmidt, Saarbrucken, Germany, 1991 (Elsevier, Amsterdam, 1992) p. 195–201.
P. BURTIN, J. P. BRUNELLE, M. PIJOLAT and M. SOUSTELLE, Appl. Catal. 34 (1987) 225.
Idem, ibid. 34 (1987) 239.
R. K. ILER, J. Amer. Ceram. Soc. 47 (1964) 339.
B. BEGUIN, M. E. GARBOWSKI and M. PRIMET, J. Catal. 127 (1991) 595.
M. MACHIDA, K. EGUCHI and H. ARAI, ibid. 103 (1987) 385.
G. GROPPI, C. CRISTIANI, P. FORZATTI and M. BELLOTTO, J. Mater. Sci. 29 (1994) 3441.
R. BRACE and E. MATIJEVIC, J. Inorg. Nucl. Chem. 35 (1973) 3691.
M. D. SACKS, T.-Y. TSENG and S. Y. LEE, Ceram. Bull. 63 (1984) 301.
B. C. LIPPENS and J. H. DE BOER, Acta Crystallogr. 17 (1964) 1312.
D. S. TUCKER, J. Amer. Ceram. Soc. 68 (1985) C163.
M. L. BALMER, F. F. LANGE and G. C. LEVI, ibid. 77 (1994) 2069.
O. YAMAGUCHI, M. SHIRAI and M. YOSHINAKA, ibid. 71 (1988) C510.
E. A. PUGAR and E. D. MORGAN, ibid. 69 (1986) C120.
M. R. GALLAS and G. P. PIERMARINI, ibid. 77 (1994) 2917.
J. R. WYNNYCKYJ and C. G. MORRIS, Metall. Trans. B 16 (1985) 345.
D. S. TUCKER, E. J. JENKINS and J. J. HREN, J. Electron Microsc. Technol. 2 (1985) 29.
F. W. DYNYS and J. W. HALLORAN, J. Amer. Ceram. Soc. 65 (1982) 442.
F. OUDET, P. COURTINE and A. VEJUX, J. Catal. 114 (1988) 112.
B. DJURIČIČ, S. PICKERING, D. McGARRY, P. GLAUDE, P. TAMBUYSER and K. SCHUSTER, Ceram. Int. 21 (1995) 195.
F. R. CHEN, G. COUDURIER, J. F. JOLY and J. C. VEDRIN, J. Catalysis 143 (1993) 616.
J. LIVAGE, K. DOI and C. MAZIERES, J. Amer. Ceram. Soc. 51 (1968) 349.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DJURICIC, B., PICKERING, S., GLAUDE, P. et al. Thermal stability of transition phases in zirconia-doped alumina. Journal of Materials Science 32, 589–601 (1997). https://doi.org/10.1023/A:1018567230733
Issue Date:
DOI: https://doi.org/10.1023/A:1018567230733