Abstract
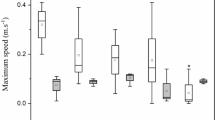
We studied the relationships between a predator fish, Gambusia holbrooki, and its main food prey, within the content of a rice field food web. The influence of some environmental and biological factors on these trophic interactions, in combination with existent quantitative information, allowed us to evaluate the ecological viability of using a non-ionic surfactant, Genapol OXD-080, to control a plague caused by crayfish (Procambarus clarkii) populations in the rice fields. In the Lower Mondego River Valley, Portugal, G. holbrooki is abundant in rice fields. It feeds mainly on copepods, cladocerans and rotifers. Surface insects, such as aphids, collembolans, adult (imago) chironomids and other dipterans, are additional food. Large G. holbrooki consumed greater amounts of cladocerans and adult chironomids than other smaller size groups, while small fish prefered rotifers. Gravid females ate copepods, cladocerans, and adult chironomids and other dipterans in significantly greater amounts than immatures, males, and non-gravid females. Non-gravid females ate collembolans in significantly greater quantities than any other fish group. The population density of copepods, cladocerans, adult chironomids, and other dipterans, the area covered by aquatic vegetation, and water temperature all had significant effects on the total number of prey caught by G. holbrooki. In contrast, a negative correlation was found with rotifers, collembolans, aphids in higher densities, and of increased water volume, dissolved oxygen and pH. G. holbrooki holds a key intermediate position in the rice field food chain, feeding in large amounts of aquatic invertebrates and being eaten, in turn, by piscivores. With regard to the toxicity of Genapol OXD-080 on non-target organisms, LC50 values for G. holbrooki and some of its main prey were several times lower than the concentration necessary to decrease the activity of crayfish populations in the rice fields. Thus, Genapol OXD-080 could potentially cause greater damage to the local populations of non-target species and should not be used without taking precautions not to contaminate other important biological reservoirs, such as the rice field irrigation channels.
Similar content being viewed by others
References
Albuquerque, R. M., 1956. Peixes de Portugal e Ilhas adjacentes. Chaves para a sua determinacyão. Portugalia Acta Biol. 5B, 1164 pp.
Anastácio, P. M. & J. C. Marques, 1995. Population biology and production of the Red Swamp Crayfish Procambarus clarkii (Girard) in the Lower Mondego River Valley, Portugal. J. Crustacean Biol. 15: 156–168.
Anastácio, P. M., S. N. Nielsen, J. C. Marques & S. E. Jørgensen, 1995. Integrated production of crayfish and rice: a management model. Ecol. Eng. 4: 199–210.
American Public Health Association, American Water Works Association, Water Pollution Control Federation, 1980. Standard methods for the examination of water and waste water. 15th edn., Washington, USA, 1134 pp.
Blaustein, L., 1992. Larvivorous fishes fail to control mosquitoes in experimental rice plots. Hydrobiologia 232: 219–232.
Britton, R. H. & M. E. Moser, 1982. Size specific predation by herons and its effect on the sex ratio of natural populations of the mosquitofish Gambusia affinis Baird and Girard. Oecologia 53: 146–151.
Cabral, J. A., P. M. Anastácio, R. Carvalho & J. C. Marques, 1997. A non harmful method of Red Swamp Crayfish (Procambarus clarkii) population control and non target organisms problematics in the Lower Mondego River Valley, Portugal. Proc. Int. Assoc. Astacol. 11th Symp. Freshwater Crayfish 11: 286–292.
Cech, J. J., M. J. Massingill, B. Vondracek & A. L. Linden, 1985. Respiratory metabolism of mosquitofish, Gambusia affinis: effects of temperature, dissolved oxygen, and sex difference. Envir. Biol. Fishes 13: 297–307.
Cech, J. J., R. G. Schwab, W. C. Coles & B. B. Bridges, 1992. Mosquitofish reproduction: effects of photoperiod and nutrition. Aquaculture 101: 361–369.
Colwell, A. E. & C. H. Schaefer, 1983. Effects of an insect growth regulator on plankton and Gambusia affinis. Aquat. Toxicol. 4: 247–269.
Corbet, S. A., G.W. Danahar, V. King, C. L. Chalmers & C. F. Tiley, 1995. Surfactant-enhanced essential oils as mosquito larvicides. Ent. exp. applic. 75: 229–236.
Crivelli, A. J. & V. Boy, 1987. The diet of the mosquitofish Gambusia affinis (Baird & Girard) (Poeciliidae) in Mediterranean France. Rev. Ecol. (Terre Vie) 42: 421–435.
Daniels, G. L. & J. D. Felley, 1992. Life history and foods of Gambusia affinis in two waterways of Southwestern Louisiana. S. West. Nat. 37: 157–165.
Fernandes, M. A. S., I. M. R. Mendonça, J. C. Marques & V. M. C. Madeira, 1994. Seasonal Changes in the biochemical and energy content of Procambarus clarkii (Girard, 1852) in the Lower Mondego river region. J. Crustacean Biol. 14: 736–743.
Fernandes, M. A. S., M. I. R. Mendonça, J. C. Marques & V. M. C. Madeira, 1995. Procambarus clarkii energy and nutrient transfer capacities in the Lower Mondego river valley (Portugal). Proc. Int. Assoc. of Astacol. 10th Symp. Freshwater Crayfish 10: 98–104.
Fonseca, J. C., J. C. Marques & V. M. C. Madeira, 1997. Oxygen uptake inhibition in Procambarus clarkii, Louisiana crayfish, by biodegradable surfactants: An ecotechnological approach for population control in rice fields. Proc. Int. Assoc. Astacol. 11th Symposium, Freshwater Crayfish 11: 235–242.
Forés, E. & F. A. Comín, 1992. Ricefields, a limnological perspective. Limnetica 8: 101–109.
Harrington, R. W. & E. S. Harrington, 1961. Food selection among fishes invading a high subtropical saltmarsh: from onset of flooding through the progress of a mosquito brood. Ecology 42: 646–666.
Haynes, J. L. & R. C. Cashner, 1995. Life history and population dynamics of the western mosquitofish: a comparison of natural and introduced populations. J. Fish Biol. 46: 1026–1041.
Holmstrup, M. & P. H. Krogh, 1996. Effects of an anionic surfactant, linear alkylbenzene sulfonate, on survival, reproduction and growth of the soil-living Collembolans Folsomia fimetaria. Environ. Toxicol. Chem. 15(10): 1745–1748.
Homski, D., M. Goren & A. Gasith, 1994. Comparative evaluation of the larvivorous fish Gambusia affinis and Aphanius dispar as mosquito control agents. Hydrobiologia 284: 137–146.
Hoy, J. B., 1985. Experimental mass-rearing of the mosquitofish, Gambusia affinis. J. Am. Mosq. Control Assoc. 1: 295–298.
Hurlbert, S. H.& M. S. Mulla, 1981. Impacts ofmosquitofish (Gambusia affinis) predation on plankton communities. Hydrobiologia 83: 125–151.
Hurlbert, S. H., J. Zedler & D. Fairbanks, 1972. Ecosystem alteration by mosquitofish (Gambusia affinis) predation. Science 175: 639–641.
Imai, T., S. Tsuchiya, K. Morita & T. Fujimori, 1994. Surface tension-dependent surfactant toxicity on the Green Peach Aphid, Myzus persicae (Sulzer) (Hemiptera: Aphididae). Appl. Entomol. Zool. 29: 389–393.
Ivlev, V. S., 1961. Experimental Ecology of the Feeding of Fishes. New Haven: Yale Univ. press, 302 pp.
Jørgensen, S. E., J. C. Marques & P. M. Anastácio, 1997. Modelling the fate of surfactants and pesticides in a rice field. Ecol. Model. 104: 205–213.
Linden, A. L. & J. J. Cech, 1990. Prey selection by mosquitofish (Gambusia affinis) in California rice fields: effect of vegetation and prey species. J. Am. Mosq. Control Assoc. 6: 115–120.
Lydeard, C. & M. C. Belk, 1993. Management of indigenous fish species impacted by introduced mosquitofish: an experimental approach. S. West. Nat. 38: 370–373.
Meffe, G. K., 1987. Embryo size variation in mosquitofish: optimality vs. plasticity in propagule size. Copeia 1987: 762–768.
Nesbit, D. H. & G. K. Meffe, 1993. Cannibalism frequencies in wild populations of the eastern mosquitofish (Gambusia holbrooki: Poeciliidae) in South Carolina. Copeia 1993: 867–870.
Reddy, S. R., 1975. Effect of water temperature on the predatory efficiency of Gambusia affinis. Experimentia 31: 801–802.
Rupp, H. R., 1996. Adverse assessments of Gambusia affinis: an alternative view for mosquito control practitioners. J. Am. Mosq. Control Assoc. 12: 155–166.
Schaefer, J. F., S. T. Heulett & T. M. Farrell, 1994. Interactions between two poeciliid fishes (Gambusia holbrooki and Heterandria formosa) and their prey in a Florida marsh. Copeia 1994: 516–520.
Walters, L. L. & E. F. Legner, 1980. Impact of the Desert Pupfish, Cyprinodon macularius, and Gambusia affinis affinis on fauna in pond ecosystems. Hilgardia 48: 1–18.
Wurtsbaugh, W., J. J. Cech & J. Compton, 1980. Effect of fish size on prey size selection in Gambusia affinis. Proceedings and papers of the forty-eighth annual Conference of the California Mosquito and Vector Control Association, Inc., January 20–23, pp. 48–51.
Zar, J. H., 1984. Biostatistical Analysis (2nd edn). Prentice-Hall International, Inc., Englewood Cliffs, New Jersey, USA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cabral, J.A., Mieiro, C.L. & Marques, J.C. Environmental and biological factors influence the relationship between a predator fish, Gambusia holbrooki, and its main prey in rice fields of the Lower Mondego River Valley (Portugal). Hydrobiologia 382, 41–51 (1998). https://doi.org/10.1023/A:1003480920168
Issue Date:
DOI: https://doi.org/10.1023/A:1003480920168