Abstract
The sarcomere of striated muscle is a very efficient machine transforming chemical energy into movement. However, a wrong distribution of the generated forces may lead to self-destruction of the engine itself. A well-known example for this is eccentric contraction (elongation of the sarcomere in the activated state), which damages sarcomeric structure and leads to a reduced muscle performance. The goal of this review is to discuss the involvement of different cytoskeletal systems, in particular the M-band filaments, in the mechanisms that provide stability during sarcomeric contraction. The M-band is the transverse structure in the center of the sarcomeric A-band, which is responsible both for the regular packing of thick filaments and for the uniform distribution of the tension over the myosin filament lattice in the activated sarcomere. Although some proteins from the Ig-superfamily, like myomesin and M-protein, are the major candidates for the role of M-band bridges, the exact molecular organisation of the M-band is not clear. However, the protein composition of the M-band seems to modulate the mechanical characteristics of the thick filament lattice, in particular its stiffness, adjusting it to the specific demands in different muscle types. The special M-band design in slow fibers might be part of structural adaptations, favouring sarcomere stability for a continuous contractile activity over a broad working range. In conclusion, we discuss why the interference with M-band structure might have fatal consequences for the integrity of the working sarcomere.
Similar content being viewed by others
References
Agarkova I, Auerbach D, Ehler E and Perriard JC (2000) A novel marker for vertebrate embryonic heart, the EH-myomesin isoform. J Biol Chem 275: 10256–10264.
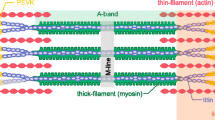
Allen DG (2001) Eccentric muscle damage: mechanisms of early reduction of force. Acta Physiol Scand 171: 311–319. Fig. 6. Consequences of the absence of the M-band for sarcomere stability: for simplicity, the elastic portion of titin is shown as a series of folded domains. After activation, the myosin filaments will accidentally deviate from the central position and stretch some of the titin filaments. This pulling force might cause the denaturation of some parts of the titin molecules. This effect might accumulate and lead to a progressive wearing out of titin in muscles, subjected to prolonged periods of contractile activity. As a result, the degree of thick filament misalignment will progressively increase, as well as the stress that is applied to the Z-discs. According to this model, the following symptoms are to be expected in sarcomeres subjected to multiple contractions: A-band misalignment, Z-disc deformation, and overall sarcomere widening followed by sarcomere disassembly. 200
Allen DL and Leinwand LA (2001) Postnatal myosin heavy chain isoform expression in normal mice and mice null for IIb or IId myosin heavy chains. Dev Biol 229: 383–395.
Almenar-Queralt A, Lee A, Conley CA, Ribas de Pouplana L and Fowler VM (1999) Identification of a novel tropomodulin isoform, skeletal tropomodulin, that caps actin filament pointed ends in fast skeletal muscle. J Biol Chem 274: 28466–28475.
Anastasi G, Cutroneo G, Santoro G and Trimarchi F (1998) The nonjunctional sarcolemmal cytoskeleton: the costameres. Ital J Anat Embryol 103: 1–11.
Auerbach D, Bantle S, Keller S, Hinderling V, Leu M, Ehler E and Perriard JC (1999) Different domains of the M-band protein myomesin are involved in myosin binding and M-band targeting. Mol Biol Cell 10: 1297–1308.
Bagnato P, Barone V, Giacomello E, Rossi D and Sorrentino V (2003) Binding of an ankyrin-1 isoform to obscurin suggests a molecular link between the sarcoplasmic reticulum and myofibrils in striated muscles. J Cell Biol 160: 245–253.
Bantle S, Keller S, Haussmann I, Auerbach D, Perriard E, Muhlebach S and Perriard JC (1996) Tissue-specific isoforms of chicken myomesin are generated by alternative splicing. J Biol Chem 271: 19042–19052.
Biral D, Jakubiec-Puka A, Ciechomska I, Sandri M, Rossini K, Carraro U and Betto R (2000) Loss of dystrophin and some dystrophin-associated proteins with concomitant signs of apoptosis in rat leg muscle overworked in extension. Acta Neuropathol (Berl) 100: 618–626.
Brockett CL, Morgan DL, Gregory JE and Proske U (2002) Damage to different motor units from active lengthening of the medial gastrocnemius muscle of the cat. J Appl Physiol 92: 1104–1110.
Carlsson E, Fischer C, Sjoberg G, Robson RM, Sejersen T and Thornell LE (2002) Cytoskeletal derangements in hereditary myopathy with a desmin L345P mutation. Acta Neuropathol (Berl) 104: 493–504.
Carlsson E, Grove BK, Wallimann T, Eppenberger HM and Thornell LE (1990) Myofibrillar M-band proteins in rat skeletal muscles during development. Histochemistry 95: 27–35.
Carlsson E and Thornell LE (1987) Diversification of the myofibrillar M-band in rat skeletal muscle during postnatal development. Cell Tissue Res 248: 169–180.
Carlsson L and Thornell LE (2001) Desmin-related myopathies in mice and man. Acta Physiol Scand 171: 341–348.
Carrion-Vazquez M, Oberhauser AF, Fowler SB, Marszalek PE, Broedel SE, Clarke J and Fernandez JM (1999) Mechanical and chemical unfolding of a single protein: a comparison. Proc Natl Acad Sci USA 96: 3694–3699.
Chien KR (1999) Stress pathways and heart failure. Cell 98: 555–558.
Chopard A, Pons F, Charpiot P and Marini JF (2000) Quantitative analysis of relative protein contents by Western blotting: comparison of three members of the dystrophin—glycoprotein complex in slow and fast rat skeletal muscle. Electrophoresis 21: 517–522.
Chopard A, Pons F and Marini JF (2001) Cytoskeletal protein contents before and after hindlimb suspension in a fast and slow rat skeletal muscle. Am J Physiol Regul Integr Comp Physiol 280: R323–R330.
Danowski BA, Imanaka-Yoshida K, Sanger JM and Sanger JW (1992) Costameres are sites of force transmission to the substratum in adult rat cardiomyocytes. J Cell Biol 118: 1411–1420.
Edman AC, Squire JM and Sjostrom M (1988) Fine structure of the Aband in cryo-sections. Diversity of M-band structure in chicken breast muscle. J Ultrastruct Mol Struct Res 100: 1–12.
Ehler E, Rothen BM, Hammerle SP, Komiyama M and Perriard JC (1999) Myofibrillogenesis in the developing chicken heart: assembly of Z-disk, M-line and the thick filaments. J Cell Sci 112: 1529–1539.
Faulkner G, Lanfranchi G and Valle G (2001) Telethonin and other new proteins of the Z-disc of skeletal muscle. IUBMB Life 51: 275–282.
Flick MJ and Konieczny SF (2000) The muscle regulatory and structural protein MLP is a cytoskeletal binding partner of betaIspectrin. J Cell Sci 113: 1553–1564.
Freiburg A, Trombitas K, Hell W, Cazorla O, Fougerousse F, Centner T, Kolmerer B, Witt C, Beckmann JS, Gregorio CC, Granzier H and Labeit S (2000) Series of exon-skipping events in the elastic spring region of titin as the structural basis for myofibrillar elastic diversity. Circ Res 86: 1114–1121.
Friden J and Lieber RL (1998) Segmental muscle fiber lesions after repetitive eccentric contractions. Cell Tissue Res 293: 165–171.
Friden J and Lieber RL (2001) Eccentric exercise-induced injuries to contractile and cytoskeletal muscle fibre components. Acta Physiol Scand 171: 321–326.
Gotthardt M, Hammer RE, Hubner N, Monti J, Witt CC, McNabb M, Richardson JA, Granzier H, Labeit S and Herz J (2003) Conditional expression of mutant M-line titins results in cardiomyopathy with altered sarcomere structure. J Biol Chem 278: 6059–6065.
Goulding D, Bullard B and Gautel M (1997) A survey of in situ sarcomere extension in mouse skeletal muscle. J Muscle Res Cell Motil 18: 465–472.
Granzier HL, Akster HA and Ter Keurs HE (1991) Effect of thin filament length on the force-sarcomere length relation of skeletal muscle. Am J Physiol 260: C1060–C1070.
Granzier H and Labeit S (2002) Cardiac titin: an adjustable multifunctional spring. J Physiol 541: 335–342.
Gregorio CC, Granzier H, Sorimachi H and Labeit S (1999) Muscle assembly: a titanic achievement? Curr Opin Cell Biol 11: 18–25.
Gregorio CC, Trombitas K, Centner T, Kolmerer B, Stier G, Kunke K, Suzuki K, Obermayr F, Herrmann B, Granzier H, Sorimachi H and Labeit S (1998) The NH2 terminus of titin spans the Z-disc: its interaction with a novel 19-kD ligand (T-cap) is required for sarcomeric integrity. J Cell Biol 143: 1013–1027.
Gregorio CC, Weber A, Bondad M, Pennise CR and Fowler VM (1995) Requirement of pointed-end capping by tropomodulin to maintain actin filament length in embryonic chick cardiac myocytes. Nature 377: 83–86.
Grove BK, Cerny L, Perriard JC and Eppenberger HM (1985) Myomesin and M-protein: expression of two M-band proteins in pectoral muscle and heart during development. J Cell Biol 101: 1413–1421.
Grove BK, Cerny L, Perriard J-C, Eppenberger HM and Thornell L-E (1989) Fiber type-specific distribution of M-band proteins in chicken muscle. J Histochem Cytochem 37: 447–454.
Hack AA, Lam MY, Cordier L, Shoturma DI, Ly CT, Hadhazy MA, Hadhazy MR, Sweeney HL and McNally EM (2000) Differential requirement for individual sarcoglycans and dystrophin in the assembly and function of the dystrophin-glycoprotein complex. J Cell Sci 113: 2535–2544.
Hanley PJ, Young AA, LeGrice IJ, Edgar SG and Loiselle DS (1999) 3-Dimensional configuration of perimysial collagen fibres in rat cardiac muscle at resting and extended sarcomere lengths. J Physiol 517: 831–837.
Hayes NV, Scott C, Heerkens E, Ohanian V, Maggs AM, Pinder JC, Kordeli E and Baines AJ (2000) Identification of a novel Cterminal variant of beta II spectrin: two isoforms of beta II spectrin have distinct intracellular locations and activities. J Cell Sci 113: 2023–2034.
Ho-Kim MA and Rogers PA (1992) Quantitative analysis of dystrophin in fast-and slow-twitch mammalian skeletal muscle. FEBS Lett 304: 187–191.
Horowits R (1992) Passive force generation and titin isoforms in mammalian skeletal muscle. Biophys J 61: 392–398.
Horowits R and Podolsky RJ (1987) The positional stability of thick filaments in activated skeletal muscle depends on sarcomere length: evidence for the role of titin filaments. J Cell Biol 105: 2217–2223.
Huijing PA (1999) Muscle as a collagen fiber reinforced composite: a review of force transmission in muscle and whole limb. J Biomech 32: 329–345.
Ishijima A, Kojima H, Higuchi H, Harada Y, Funatsu T and Yanagida T (1996) Multiple-and single-molecule analysis of the actomyosin motor by nanometer-piconewton manipulation with a microneedle: unitary steps and forces. Biophys J 70: 383–400.
Keller A, Demeurie J, Merkulova T, Geraud G, Cywiner-Golenzer C, Lucas M and Chatelet FP (2000) Fibre-type distribution and subcellular localisation of alpha and beta enolase in mouse striated muscle. Biol Cell 92: 527–535.
Kellermayer MS, Smith SB, Bustamante C and Granzier HL (2001) Mechanical fatigue in repetitively stretched single molecules of titin. Biophys J 80: 852–863.
Kellermayer MS, Smith SB, Granzier HL and Bustamante C (1997) Folding-unfolding transitions in single titin molecules characterized with laser tweezers. Science 276: 1112–1116.
Kemp TJ, Sadusky TJ, Simon M, Brown R, Eastwood M, Sassoon DA and Coulton GR (2001) Identification of a novel stretch-responsive skeletal muscle gene (Smpx). Genomics 72: 260–271.
Kenny PA, Liston EM and Higgins DG (1999) Molecular evolution of immunoglobulin and fibronectin domains in titin and related muscle proteins. Gene 232: 11–23.
Knappeis GG and Carlsen F (1968) The ultrastructure of the M-line in skeletal muscle. J Cell Biol 38: 202–211.
Knupp C, Luther PK and Squire JM (2002) Titin organisation and the 3D architecture of the vertebrate-striated muscle I-band. J Mol Biol 322: 731–739.
Kolmerer B, Olivieri N, Witt CC, Herrmann BG and Labeit S (1996) Genomic organization of M-line titin and its tissue-specific expression in two distinct isoforms. J Mol Biol 256: 556–563.
Kruger M, Wright J and Wang K (1991) Nebulin as a length regulator of thin filaments of vertebrate skeletal muscles: correlation of thin filament length, nebulin size, and epitope profile. J Cell Biol 115: 97–107.
Labeit S and Kolmerer B (1995) Titins: giant proteins in charge of muscle ultrastructure and elasticity. Science 270: 293–296.
Lange S, Auerbach D, McLoughlin P, Perriard E, Schafer BW, Perriard JC and Ehler E (2002) Subcellular targeting of metabolic enzymes to titin in heart muscle may be mediated by DRAL/FHL-2. J Cell Sci 115: 4925–4936.
Li Z, Colucci-Guyon E, Pincon-Raymond M, Mericskay M, Pournin S, Paulin D and Babinet C (1996) Cardiovascular lesions and skeletal myopathy in mice lacking desmin. Dev Biol 175: 362–366.
Li H, Linke WA, Oberhauser AF, Carrion-Vazquez M, Kerkvliet JG, Lu H, Marszalek PE and Fernandez JM (2002) Reverse engineering of the giant muscle protein titin. Nature 418: 998–1002.
Li Z, Mericskay M, Agbulut O, Butler-Browne G, Carlsson L, Thornell LE, Babinet C and Paulin D (1997) Desmin is essential for the tensile strength and integrity of myofibrils but not for myogenic commitment, differentiation, and fusion of skeletal muscle. J Cell Biol 139: 129–144.
Lieber RL, Thornell LE and Friden J (1996) Muscle cytoskeletal disruption occurs within the first 15 min of cyclic eccentric contraction. J Appl Physiol 80: 278–284.
Linke WA and Granzier H (1998) A spring tale: new facts on titin elasticity. Biophys J 75: 2613–2614.
Linke WA, Rudy DE, Centner T, Gautel M, Witt C, Labeit S and Gregorio CC (1999) I-band titin in cardiac muscle is a threeelement molecular spring and is critical for maintaining thin filament structure. J Cell Biol 146: 631–644.
Littlefield R and Fowler VM (2002) Measurement of thin filament lengths by distributed deconvolution analysis of fluorescence images. Biophys J 82: 2548–2564.
Liversage AD, Holmes D, Knight PJ, Tskhovrebova L and Trinick J (2001) Titin and the sarcomere symmetry paradox. J Mol Biol 305: 401–409.
Luther PK, Munro PM and Squire JM (1981) Three-dimensional structure of the vertebrate muscle A-band. III. M-region structure and myosin filament symmetry. J Mol Biol 151: 703–730.
Luther P and Squire J (1978) Three-dimensional structure of the vertebrate muscle M-region. J Mol Biol 125: 313–324.
Macpherson PC, Dennis RG and Faulkner JA (1997) Sarcomere dynamics and contraction-induced injury to maximally activated single muscle fibres from soleus muscles of rats. J Physiol 500: 523–533.
Macpherson PC, Schork MA and Faulkner JA (1996) Contractioninduced injury to single fiber segments from fast and slow muscles of rats by single stretches. Am J Physiol 271: C1438–C1446.
McElhinny AS, Kakinuma K, Sorimachi H, Labeit S and Gregorio CC (2002) Muscle-specific RING finger-1 interacts with titin to regulate sarcomeric M-line and thick filament structure and may have nuclear functions via its interaction with glucocorticoid modulatory element binding protein-1. J Cell Biol 157: 125–136.
McElhinny AS, Kolmerer B, Fowler VM, Labeit S and Gregorio CC (2001) The N-terminal end of nebulin interacts with tropomodulin at the pointed ends of the thin filaments. J Biol Chem 276: 583–592.
McLoughlin P, Ehler E, Carlile G, Licht JD and Schafer BW (2002) The LIM-only protein DRAL/FHL2 interacts with and is a corepressor for the promyelocytic leukemia zinc finger protein. J Biol Chem 277: 37045–37053.
Meyer PL (1966) Introductory Probability and Statistical Applications. Addison-Wesley, Reading, MA.
Millman BM (1998) The filament lattice of striated muscle. Physiol Rev 78: 359–391.
Milner DJ, Taffet GE, Wang X, Pham T, Tamura T, Hartley C, Gerdes AM and Capetanaki Y (1999) The absence of desmin leads to cardiomyocyte hypertrophy and cardiac dilation with compromised systolic function. J Mol Cell Cardiol 31: 2063–2076.
Minajeva A, Kulke M, Fernandez JM and Linke WA (2001) Unfolding of titin domains explains the viscoelastic behavior of skeletal myofibrils. Biophys J 80: 1442–1451.
Monti RJ, Roy RR, Hodgson JA and Edgerton VR (1999) Transmission of forces within mammalian skeletal muscles. J Biomech 32: 371–380.
Morgan DL (1990) New insights into the behavior of muscle during active lengthening. Biophys J 57: 209–221.
Muller JM, Isele U, Metzger E, Rempel A, Moser M, Pscherer A, Breyer T, Holubarsch C, Buettner R and Schule R (2000) FHL2, a novel tissue-specific coactivator of the androgen receptor. Embo J 19: 359–369.
Mutungi G and Ranatunga KW (1996) The viscous, viscoelastic and elastic characteristics of resting fast and slow mammalian (rat) muscle fibres. J Physiol 496: 827–836.
Nave R, Furst DO and Weber K (1989) Visualization of the polarity of isolated titin molecules: a single globular head on a long thin rod as the M band anchoring domain? J Cell Biol 109: 2177–2187.
Nishino I and Ozawa E (2002) Muscular dystrophies. Curr Opin Neurol 15: 539–544.
Obermann WMJ, Gautel M, Steiner F, Vanderven PFM, Weber K and Furst DO (1996) The structure of the sarcomeric M band: localization of defined domains of myomesin, M protein, and the 250 Kd carboxy terminal region of titin by immunoelectron microscopy. J Cell Biol 134: 1441–1453.
Obermann WMJ, Gautel M, Weber K and Furst DO (1997) Molecular structure of the sarcomeric M-band: mapping of titin and myosin binding domains in myomesin and the identification of a potential regulatory phosphorylation site in myomesin. Embo J 16: 211–220.
Obermann WMJ, van der Ven PFM, Steiner F, Weber K and Fuürst DO (1998) Mapping of a myosin-binding domain and a regulatory phosphorylation site in M-protein, a structural protein of the sarcomeric M band. Mol Biol Cell 9: 829–840.
Ogilvie RW, Armstrong RB, Baird KE and Bottoms CL (1988) Lesions in the rat soleus muscle following eccentrically biased exercise. Am J Anat 182: 335–346.
Page SG and Huxley HE (1963) Filament lengths in striated muscle. J Cell Biol 19: 369–390.
Palmer S, Groves N, Schindeler A, Yeoh T, Biben C, Wang CC, Sparrow DB, Barnett L, Jenkins NA, Copeland NG, Koentgen F, Mohun T and Harvey RP (2001) The small muscle-specific protein Cs1 modifies cell shape and promotes myocyte fusion in an insulinlike growth factor 1-dependent manner. J Cell Biol 153: 985–998.
Pardo JV, Siliciano JD and Craig SW (1983) A vinculin-containing cortical lattice in skeletal muscle: transverse lattice elements (‘costameres’) mark sites of attachment between myofibrils and sarcolemma. Proc Natl Acad Sci USA 80: 1008–1012.
Pask HT, Jones KL, Luther PK and Squire JM (1994) M-band structure, M-bridge interactions and contraction speed in vertebrate cardiac muscles. J Muscle Res Cell Motil 15: 633–645.
Patel TJ and Lieber RL (1998) Force transmission in skeletal muscle: from actomyosin to external tendons. Exerc Sport Sci Rev 25: 321–363.
Pierobon-Bormioli S (1981) Transverse sarcomere filamentous systems: ‘Z-and M-cables’. J Muscle Res Cell Motil 2: 401–413.
Pizon V, Iakovenko A, Van Der Ven PF, Kelly R, Fatu C, Furst DO, Karsenti E and Gautel M (2002) Transient association of titin and myosin with microtubules in nascent myofibrils directed by the MURF2 RING-finger protein. J Cell Sci 115: 4469–4482.
Price MG and Gomer RH (1993) Skelemin, a cytoskeletal M-disc periphery protein, contains motifs of adhesion/recognition and intermediate filament proteins. J Biol Chem 268: 21800–21810.
Proske U and Morgan DL (2001) Muscle damage from eccentric exercise: mechanism, mechanical signs, adaptation and clinical applications. J Physiol 537: 333–345.
Rief M, Gautel M, Schemmel A and Gaub HE (1998) The mechanical stability of immunoglobulin and fibronectin III domains in the muscle protein titin measured by atomic force microscopy. Biophys J 75: 3008–3014.
Robinson TF and Winegrad S (1979) The measurement and dynamic implications of thin filament lengths in heart muscle. J Physiol 286: 607–619.
Sjostrom M and Squire JM (1977) Fine structure of the A-band in cryo-sections. The structure of the A-band of human skeletal muscle fibres from ultra-thin cryo-sections negatively stained. J Mol Biol 109: 49–68.
Smolich JJ (1995) Ultrastructural and functional features of the developing mammalian heart: a brief overview. Reprod Fertil Dev 7: 451–461.
Sorimachi H, Freiburg A, Kolmerer B, Ishiura S, Stier G, Gregorio CC, Labeit D, Linke WA, Suzuki K and Labeit S (1997) Tissue-specific expression and alpha-actinin binding properties of the Zdisc titin: implications for the nature of vertebrate Z-discs. J Mol Biol 270: 688–695.
Squire JM (1997) Architecture and function in the muscle sarcomere. Curr Opin Struct Biol 7: 247–257.
Steiner F, Weber K and Furst D (1999) M-band proteins myomesin and skelemin are incoded by the same gene: analysis of its organisation and expression. Genomics 56: 78–89.
Stolz M, Kraft T and Wallimann T (1998) The isoenzyme-diagnostic regions of muscle-type creatine kinase, the M-260 and M-300 box, are not responsible for its binding to the myofibrillar M-band. Eur J Cell Biol 77: 1–9.
Street SF (1983) Lateral transmission of tension in frog myofibers: a myofibrillar network and transverse cytoskeletal connections are possible transmitters. J Cell Physiol 114: 346–364.
Thornell L, Carlsson L, Li Z, Mericskay M and Paulin D (1997) Null mutation in the desmin gene gives rise to a cardiomyopathy. J Mol Cell Cardiol 29: 2107–2124.
Towbin JA and Bowles NE (2000) Genetic abnormalities responsible for dilated cardiomyopathy. Curr Cardiol Rep 2: 475–480.
Traeger L and Goldstein MA (1983) Thin filaments are not of uniform length in rat skeletal muscle. J Cell Biol 96: 100–103.
Trombitas K, Freiburg A, Centner T, Labeit S and Granzier H (1999) Molecular dissection of N2B cardiac titin’s extensibility. Biophys J 77: 3189–3196.
Tskhovrebova L and Trinick J (2002) Role of titin in vertebrate striated muscle. Philos Trans R Soc Lond B Biol Sci 357: 199–206.
Tskhovrebova L, Trinick J, Sleep JA and Simmons RM (1997) Elasticity and unfolding of single molecules of the giant muscle protein titin. Nature 387: 308–312.
Turner DC, Maier V and Eppenberger HM (1974) Creatine kinase and aldolase isoenzyme transitions in cultures of chick skeletal muscle cells. Dev Biol 37: 63–89.
van der Ven PF, Bartsch JW, Gautel M, Jockusch H and Furst DO (2000) A functional knock-out of titin results in defective myofibril assembly. J Cell Sci 113: 1405–1414.
Vendelin M, Bovendeerd PH, Engelbrecht J and Arts T (2002) Optimizing ventricular fibers: uniform strain or stress, but not ATP consumption, leads to high efficiency. Am J Physiol Heart Circ Physiol 283: H1072–H1081.
Vijayan K, Thompson JL, Norenberg KM, Fitts RH and Riley DA (2001) Fiber-type susceptibility to eccentric contraction-induced damage of hindlimb-unloaded rat AL muscles. J Appl Physiol 90: 770–776.
Walmsley B, Hodgson JA and Burke RE (1978) Forces produced by medial gastrocnemius and soleus muscles during locomotion in freely moving cats. J Neurophysiol 41: 1203–1216.
Wang K and Ramirez-Mitchell R (1983) A network of transverse and longitudinal intermediate filaments is associated with sarcomeres of adult vertebrate skeletal muscle. J Cell Biol 96: 562–570.
Warren GL, Ingalls CP, Lowe DA and Armstrong RB (2001) Excitation—contraction uncoupling: major role in contractioninduced muscle injury. Exerc Sport Sci Rev 29: 82–87.
Watanabe K, Muhle-Goll C, Kellermayer MS, Labeit S and Granzier H (2002) Different molecular mechanics displayed by titin’s constitutively and differentially expressed tandem Ig segments. J Struct Biol 137: 248–258.
Widrick JJ, Romatowski JG, Karhanek M and Fitts RH (1997) Contractile properties of rat, rhesus monkey, and human type I muscle fibers. Am J Physiol 272: R34–R42.
Williams MW and Bloch RJ (1999) Differential distribution of dystrophin and beta-spectrin at the sarcolemma of fast twitch skeletal muscle fibers. J Muscle Res Cell Motil 20: 383–393.
Xu X, Meiler SE, Zhong TP, Mohideen M, Crossley DA, Burggren WW and Fishman MC (2002) Cardiomyopathy in zebrafish due to mutation in an alternatively spliced exon of titin. Nat Genet 30: 205–209.
Yamaguchi M, Izumimoto M, Robson RM and Stromer MH (1985) Fine structure of wide and narrow vertebrate muscle Z-lines. A proposed model and computer simulation of Z-line architecture. J Mol Biol 184: 621–643.
Yeung EW, Balnave CD, Ballard HJ, Bourreau JP and Allen DG (2002) Development of T-tubular vacuoles in eccentrically damaged mouse muscle fibres. J Physiol 540: 581–592.
Young P, Ehler E and Gautel M (2001) Obscurin, a giant sarcomeric Rho guanine nucleotide exchange factor protein involved in sarcomere assembly. J Cell Biol 154: 123–136.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agarkova, I., Ehler, E., Lange, S. et al. M-band: a safeguard for sarcomere stability?. J Muscle Res Cell Motil 24, 191–203 (2003). https://doi.org/10.1023/A:1026094924677
Issue Date:
DOI: https://doi.org/10.1023/A:1026094924677