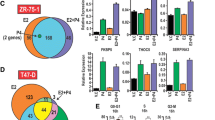
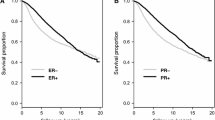
Abstract
Even the first expression profiling studies of breast cancers have generated new insights. They suggest for example, that information about tumor aggressiveness, prognosis, metastatic potential, or treatment outcome is encoded in, and can be deduced from, the primary tumor. On the other hand no clinical genomic array data have yet been published that deal with hormonal aspects of breast tumorigenesis, tumor progression, or therapeutics. Rather, studies have focused on experimental model systems. We review below the currently published data on array profiling in clinical breast cancer, then describe our studies in breast cancer cell lines and xenograft models dealing with progesterone receptors (PRs) and the role of progesterone. We demonstrate that the two PR isoforms, PR-A and PR-B, have mostly nonoverlapping molecular signatures when liganded by progesterone, with PR-B the more active form. Additionally, we document the surprising finding that unliganded PRs can regulate gene transcription, with PR-A the more active form. In ovariectomized mice supplemented with estradiol but lacking measurable progesterone, PR-B-expressing tumors grow to twice the size of PR-A-expressing ones. We conclude that in breast cancers, PRs are more than simple markers of estrogen receptor function. Rather, presence of PRs and the ratio of the two isoforms directly influence tumor phenotype, even in the absence of ligand.
Similar content being viewed by others
REFERENCES
M. Schena, D. Shalon, R. W. Davis, and P. O. Brown (1995). Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science 270:467–470.
A. C. Pease, D. Solas, E. J. Sullivan, M. T. Cronin, C. P. Holmes, and S. P. Fodor (1994). Light-generated oligonucleotide arrays for rapid DNA sequence analysis. Proc. Natl. Acad. Sci. U.S.A. 91:5022–5026.
H. C. King and A. A. Sinha (2001). Gene expression profile analysis by DNA microarrays: Promise and pitfalls. JAMA 286:2280–2288.
T. R. Golub, D. K. Slonim, P. Tamayo, C. Huard, M. Gaasenbeek, and J. P. Mesirov, et al. (1999). Molecular classification of cancer: Class discovery and class prediction by gene expression monitoring. Science 286:531–537.
A. A. Alizadeh, D. T. Ross, C. M. Perou, and M. van de Rijn (2001). Towards a novel classification of human malignancies based on gene expression patterns. J. Pathol. 195:41–52.
M. Bittner, P. Meltzer, Y. Chen, Y. Jiang, E. Seftor, M. Hendrix, et al. (2000). Molecular classification of cutaneous malignant melanoma by gene expression profiling. Nature 406:536–540.
E. A. Clark, T. R. Golub, E. S. Lander, and R. O. Hynes (2000). Genomic analysis of metastasis reveals an essential role for RhoC. Nature 406:532–535.
U. Alon, N. Barkai, D. A. Notterman, K. Gish, S. Ybarra, D. Mack, et al. (1999). Broad patterns of gene expression revealed by clustering analysis of tumor and normal colon tissues probed by oligonucleotide arrays. Proc. Natl. Acad. Sci. U.S.A. 96:6745–6750.
L. Bubendorf, M. Kolmer, J. Kononen, P. Koivisto, S. Mousses, Y. Chen, et al. (1999). Hormone therapy failure in human prostate cancer: Analysis by complementary DNA and tissue microarrays. J. Natl. Cancer. Inst. 91:1758–1764.
S. Ramaswamy and T. R. Golub (2002). DNA microarrays in clinical oncology. J. Clin. Oncol. 20:1932–1941.
S. Ramaswamy, P. Tamayo, R. Rifkin, S. Mukherjee, C. H. Yeang, M. Angelo, et al. (2001). Multiclass cancer diagnosis using tumor gene expression signatures. Proc. Natl. Acad. Sci. U.S.A. 98:15149–15154.
D. T. Ross, U. Scherf, M. B. Eisen, C. M. Perou, C. Rees, P. Spellman, et al. (2000). Systematic variation in gene expression patterns in human cancer cell lines. Nat. Genet. 24:227–235.
K. J. Martin, E. Graner, Y. Li, L. M. Price, B. M. Kritzman, M. V. Fournier, et al. (2001). High-sensitivity array analysis of gene expression for the early detection of disseminated breast tumor cells in peripheral blood. Proc. Natl. Acad. Sci. U.S.A. 98:2646–2651.
I. Hedenfalk, D. Duggan, Y. Chen, M. Radmacher, M. Bittner, R. Simon, et al. (2001). Gene-expression profiles in hereditary breast cancer. N. Engl. J. Med. 344:539–548.
D. P. Harkin (2000). Uncovering functionally relevant signaling pathways using microarray-based expression profiling. Oncologist 5:501–507.
L. J. van't Veer, H. Dai, M. J. van de Vijver, Y. D. He, A. A. Hart, M. Mao, et al. (2002). Gene expression profiling predicts clinical outcome of breast cancer. Nature 415:530–536.
C. M. Perou, S. S. Jeffrey, M. van de Rijn, C. A. Rees, M. B. Eisen, D. T. Ross, et al. (1999). Distinctive gene expression patterns in human mammary epithelial cells and breast cancers. Proc. Natl. Acad. Sci. U.S.A. 96:9212–9217.
M. A. Unger, M. Rishi, V. B. Clemmer, J. L. Hartman, E. A. Keiper, J. D. Greshock, et al. (2001). Characterization of adjacent breast tumors using oligonucleotide microarrays. Breast Cancer Res. 3:336–341.
C. M. Perou, T. Sorlie, M. B. Eisen, M. van de Rijn, S. S. Jeffrey, C. A. Rees, et al. (2000). Molecular portraits of human breast tumours. Nature 406:747–752.
A. Ahr, T. Karn, C. Solbach, T. Seiter, K. Strebhardt, U. Holtrich, et al. (2002). Identification of high risk breast-cancer patients by gene expression profiling. Lancet 359:131–132.
S. Ramaswamy, K. N. Ross, E. S. Lander, and T. R. Golub (2003). A molecular signature of metastasis in primary solid tumors. Nat. Genet. 33:49–54.
L. A. Liotta and E. C. Kohn (2003). Cancer's deadly signature. Nat. Genet. 33:10–11.
M. J. van de Vijver, Y. D. He, L. J. van't Veer, H. Dai, A. A. Hart, D. W. Voskuil, et al. (2002). A gene-expression signature as a predictor of survival in breast cancer. N. Engl. J. Med. 347:1999–2009.
A. Adeyinka, E. Emberley, Y. Niu, L. Snell, L. C. Murphy, H. Sowter, et al. (2002). Analysis of gene expression in ductal carcinoma in situ of the breast. Clin. Cancer Res. 8:3788–3795.
V. Luzzi, V. Holtschlag, and M. A. Watson (2001). Expression profiling of ductal carcinoma in situ by laser capture microdissection and high-density oligonucleotide arrays. Am. J. Pathol. 158:2005–2010.
S. K. Gruvberger, M. Ringner, P. Eden, A. Borg, M. Ferno, C. Peterson, et al. (2002). Expression profiling to predict outcome in breast cancer: The influence of sample selection. Breast Cancer Res. 5:23–26.
L. J. van't Veer, H. Dai, M. J. van de Vijver, Y. D. He, A. A. Hart, R. Bernards, et al. (2002). Expression profiling predicts outcome in breast cancer. Breast Cancer Res. 5:57–58.
T. A. Ince and R. A. Weinberg (2002). Functional genomics and the breast cancer problem. Cancer Cell 1:15–17.
A. Kallioniemi (2002). Molecular signatures of breast cancer—predicting the future. N. Engl. J. Med. 347:2067–2068.
T. Sorlie, C. M. Perou, R. Tibshirani, T. Aas, S. Geisler, H. Johnsen, et al. (2001). Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. U.S.A. 98:10869–10874.
F. Bertucci, V. Nasser, S. Granjeaud, F. Eisinger, J. Adelaide, R. Tagett, et al. (2002). Gene expression profiles of poor-prognosis primary breast cancer correlate with survival. Hum. Mol. Genet. 11:863–872.
K. B. Horwitz, W. L. McGuire, O. H. Pearson, and A. Segaloff (1975). Predicting response to endocrine therapy in human breast cancer. A hypothesis. Science 189:726–727.
D. C. Sgroi, S. Teng, G. Robinson, R. LeVangie, J. R. Hudson, and A. G. Elkahloun (1999). In vivo gene expression profile analysis of human breast cancer progression. Cancer Res. 59:5656–5661.
C. Sotiriou, T. J. Powles, M. Dowsett, A. A. Jazaeri, A. L. Feldman, L. Assersohn, et al. (2002). Gene expression profiles derived from fine needle aspiration correlate with response to systemic chemotherapy in breast cancer. Breast Cancer Res. 4:R3.
U. Scherf, D. T. Ross, M. Waltham, L. H. Smith, J. K. Lee, L. Tanabe, et al. (2000). A gene expression database for the molecular pharmacology of cancer. Nat. Genet. 24:236–244.
J. K. Richer, B. M. Jacobsen, N. G. Manning, M. G. Abel, D. M. Wolf, and K. B. Horwitz (2002). Differential gene regulation by the two progesterone receptor isoforms in human breast cancer cells. J. Biol. Chem. 277:5209–5218.
B. M. Jacobsen, J. K. Richer, S. A. Schittone, and K. B. Horwitz (2002). New human breast cancer cells to study progesterone receptor isoform ratio effects and ligand-independent gene regulation. J. Biol. Chem. 277:27793–27800.
A. Inoue, N. Yoshida, Y. Omoto, S. Oguchi, T. Yamori, R. Kiyama, et al. (2002). Development of cDNA microarray for expression profiling of estrogen-responsive genes. J. Mol. Endocrinol. 29:175–192.
M. Soulez and M. G. Parker (2001). Identification of novel oestrogen receptor target genes in human ZR75–1 breast cancer cells by expression profiling. J. Mol. Endocrinol. 27:259–274.
A. S. Levenson, I. L. Kliakhandler, K. M. Svoboda, K. M. Pease, S. A. Kaiser, J. E. Ward III, et al. (2002). Molecular classification of selective oestrogen receptor modulators on the basis of gene expression profiles of breast cancer cells expressing oestrogen receptor alpha. Br. J. Cancer 87:449–456.
S. G. Hilsenbeck, W. E. Friedrichs, R. Schiff, P. O'Connell, R. K. Hansen, C. K. Osborne, et al. (1999). Statistical analysis of array expression data as applied to the problem of tamoxifen resistance. J. Natl. Cancer. Inst. 91:453–459.
K. B. Horwitz and W. L. McGuire (1978). Estrogen control of progesterone receptor in human breast cancer. Correlation with nuclear processing of estrogen receptor. J. Biol. Chem. 253:2223–2228.
W. L. McGuire (1978). Hormone receptors: Their role in predicting prognosis and response to endocrine therapy. Semin. Oncol. 5:428–433.
K. B. Horwitz, L. L. Wei, S. M. Sedlacek, and C. N. d'Arville (1985). Progestin action and progesterone receptor structure in human breast cancer: A review. Recent Prog. Horm. Res. 41:249–316.
S. D. Costa, S. Lange, K. Klinga, E. Merkle, and M. Kaufmann (2002). Factors influencing the prognostic role of oestrogen and progesterone receptor levels in breast cancer—Results of the analysis of 670 patients with 11 years of follow-up. Eur. J. Cancer 38:1329–1334.
Writing Group for the Women's Health Initiative Investigators: Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results from the Women's Health Initiative randomized controlled trial. JAMA 288:321–333.
L. Bergkvist, H-O. Adami, I. Persson, R. Hoover, and C. Schairer (1989). The risk of breast cancer after estrogen and estrogen-progestin replacement. N. Engl. J. Med. 321:293–297.
I. Persson, E. Weiderpass, L. Bergkvist, R. Bergstrom, and C. Schairer (1999). Risks of breast and endometrial cancer after estrogen and estrogen-progestin replacement. Cancer Causes Control 10:253–260.
C. Schairer, J. Lubin, R. Troisi, S. Sturgeon, L. Brinton, and R. Hoover (2000). Menopausal estrogen and estrogen-progestin replacement therapy and breast cancer risk. JAMA 283:485–491.
G. A. Greendale, B. A. Reboussin, S. Slone, C. Wasilauskas, M. C. Pike, and G. Ursin (2003). Postmenopausal hormone therapy and change in mammographic density. J. Natl. Cancer Inst. 95:30–37.
C. L. Clarke and R. L. Sutherland (1990). Progestin regulation of cellular proliferation. Endocr. Rev. 11:266–301.
C. A. Sartorius, M. Y. Melville, A. R. Hovland, L. Tung, G. S. Takimoto, and K. B. Horwitz (1994). A third transactivation function (AF3) of human progesterone receptors located in the unique N-terminal segment of the B isoform. Mol. Endocrinol. 8:1347–1360.
M. E. Meyer, C. Quirin-Stricker, T. Lerouge, M. T. Bocquel, and H. Gronemeyer (1992). A limiting factor mediates the differential activation of promoters by the human progesterone receptor isoforms. J. Biol. Chem. 267:10882–10887.
E. Vegeto, M. M. Shabaz, D. X. Wen, M. E. Goldman, B. W. O'Malley, and D. P. McDonnell (1993). Human progesterone receptor A form is a cell-and promoter-specific repressor of human progesterone receptor B function. Mol. Endocrinol. 7:1244–1255.
C. A. Sartorius, L. Tung, G. S. Takimoto, and K. B. Horwitz (1993). Antagonist-occupied human progesterone receptors bound to DNA are functionally switched to transcriptional agonists by cAMP. J. Biol. Chem. 268:9262–9266.
L. Tung, M. K. Mohamed, J. P. Hoeffler, G. S. Takimoto, and K. B. Horwitz (1993). Antagonist-occupied human progesterone B-receptors activate transcription without binding to progesterone response elements and are dominantly inhibited by A-receptors. Mol. Endocrinol. 7:1256–1265.
D. P. McDonnell and M. E. Goldman (1994). RU486 exerts antiestrogenic activities through a novel progesterone receptor A form-mediated mechanism. J. Biol. Chem. 269:11945–11949.
A. R. Hovland, R. L. Powell, G. S. Takimoto, L. Tung, and K. B. Horwitz (1998). An N-terminal inhibitory function, IF, suppresses transcription by the A-isoform but not the B-isoform of human progesterone receptors. J. Biol. Chem. 273:5455–5460.
P. A. Mote, R. L. Balleine, E. M. McGowan, and C. L. Clarke (1999). Colocalization of progesterone receptors A and B by dual immunofluorescent histochemistry in human endometrium during the menstrual cycle. J. Clin. Endocrinol. Metab. 84:2963–2971.
P. A. Mote, R. L. Balleine, E. M. McGowan, and C. L. Clarke (2000). Heterogeneity of progesterone receptors A and B expression in human endometrial glands and stroma. Hum. Reprod. 15 (Suppl. 3):48–56.
C. J. Farr, D. J. Easty, J. Ragoussis, J. Collignon, R. Lovell-Badge, and P. N. Goodfellow (1993). Characterization and mapping of the human SOX 4 gene. Mamm. Genome 4:577–584.
J. Fujimoto, S. Ichigo, R. Hirose, H. Sakaguchi, and T. Tamaya (1997). Clinical implication of expression of progesterone receptor form A and B mRNAs in secondary spreading of gynecologic cancers. J. Steroid Biochem. Mol. Biol. 62:449–454.
B. Mulac-Jericevic, R. A. Mullinax, F. J. DeMayo, J. P. Lydon, and O. M. Conneely (2000). Subgroup of reproductive functions of progesterone mediated by progesterone receptor-B isoform. Science 289:1751–1754.
G. Shyamala, X. Yang, G. Silberstein, M. H. Barcellos-Hoff, and E. Dale (1998). Transgenic mice carrying an imbalance in the native ratio of A to B forms of progesterone receptor exhibit developmental abnormalities in mammary glands. Proc. Natl. Acad. Sci. U.S.A. 95:696–701.
G. Shyamala, X. Yang, R. D. Cardiff, and E. Dale (2000). Impact of progesterone receptor on cell-fate decisions during mammary gland development. Proc. Natl. Acad. Sci. U.S.A. 97:3044–3049.
J. D. Graham, C. Yeates, R. L. Balleine, S. S. Harvey, J. S. Milliken, A. M. Bilous, et al. (1995). Characterization of progesterone receptor A and B expression in human breast cancer. Cancer Res. 55:5063–5068.
E. M. McGowan and C. L. Clarke (1999). Effect of overexpression of progesterone receptor A on endogenous progestin-sensitive endpoints in breast cancer cells. Mol. Endocrinol. 13:1657–1671.
A. Bamberger, K. Milde-Langosch, H. Schulte, and T. Loning (2000). Progesterone receptor isoforms, pr-b and pr-a, in breast cancer: Correlations with clinicopathologic tumor parameters and expression of ap-1 factors. Horm. Res. 54:32–37.
P. A. Mote, S. Bartow, N. Tran, and C. L. Clarke (2002). Loss of co-ordinate expression of progesterone receptors A and B is an early event in breast carcinogenesis. Breast Cancer Res. Treat. 72:163–172.
I. D. Guerreiro Da Silva, Y. F. Hu, I. H. Russo, X. Ao, A. M. Salicioni, X. Yang, et al. (2000). S100P calcium-binding protein overexpression is associated with immortalization of human breast epithelial cells in vitro and early stages of breast cancer development in vivo. Int. J. Oncol. 16:231–240.
B. A. Lwaleed, M. Chisholm, and J. L. Francis (1999). Urinary tissue factor levels in patients with breast and colorectal cancer. J. Pathol. 187:291–294.
T. Ueno, M. Toi, M. Koike, S. Nakamura, T. Tominaga, B. A. Lwaleed, et al. (2000). Tissue factor expression in breast cancer tissues: Its correlation with prognosis and plasma concentration. Br. J. Cancer 83:164–170.
S. Goruppi, C. Chiaruttini, M. E. Ruaro, B. Varnum, and C. Schneider (2001). Gas6 induces growth, beta-Catenin stabilization, and T-cell factor transcriptional activation in contact-inhibited C57 mammary cells. Mol. Cell. Biol. 21:902–915.
M. Nacht, A. T. Ferguson, W. Zhang, J. M. Petroziello, B. P. Cook, Y. H. Gao, et al. (1999). Combining serial analysis of gene expression and array technologies to identify genes differentially expressed in breast cancer. Cancer Res. 59:5464–5470.
F. Arcuri, C. Monder, C. J. Lockwood, and F. Schatz (1996). Expression of 11 beta-hydroxysteroid dehydrogenase during decidualization of human endometrial stromal cells. Endocrinology 137:595–600.
A. D. Darnel, T. K. Archer, and K. Yang (1999). Regulation of 11beta-hydroxysteroid dehydrogenase type 2 by steroid hormones and epidermal growth factor in the Ishikawa human endometrial cell line. J. Steroid Biochem. Mol. Biol. 70:203–210.
H. Watari, E. J. Blanchette-Mackie, N. K. Dwyer, M. Watari, C. G. Burd, S. Patel, et al. (2000). Determinants of NPC1 expression and action: Key promoter regions, posttranscriptional control, and the importance of a "cysteine-rich" loop. Exp. Cell. Res. 259:247–256.
H. A. Kester, B. M. van der Leede, P. T. van der Saag, and B. van der Burg (1997). Novel progesterone target genes identified by an improved differential display technique suggest that progestin-induced growth inhibition of breast cancer cells coincides with enhancement of differentiation. J. Biol. Chem. 272:16637–16643.
X. Liu, G. W. Robinson, K. U. Wagner, L. Garrett, A. Wynshaw-Boris, and L. Hennighausen (1997). Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev. 11:179–186.
G. W. Robinson, P. F. Johnson, L. Hennighausen, and E. Sterneck (1998). The C/EBPbeta transcription factor regulates epithelial cell proliferation and differentiation in the mammary gland. Genes Dev. 12:1907–1916.
T. N. Seagroves, S. Krnacik, B. Raught, J. Gay, B. Burgess-Beusse, G. J. Darlington, et al. (1998). C/EBPbeta, but not C/EBPalpha, is essential for ductal morphogenesis, lobuloalveolar proliferation, and functional differentiation in the mouse mammary gland. Genes Dev. 12:1917–1928.
Y. Friedmann and C. W. Daniel (1996). Regulated expression of homeobox genes Msx-1 and Msx-2 in mouse mammary gland development suggests a role in hormone action and epithelial-stromal interactions. Dev. Biol. 177:347–355.
S. van der Flier, A. Brinkman, M. P. Look, E. M. Kok, M. E. Meijer-van Gelder, J. G. Klijn, et al. (2000). Bcar1/p130Cas protein and primary breast cancer: Prognosis and response to tamoxifen treatment. J. Natl. Cancer Inst. 92:120–127.
S. van der Flier, C. M. Chan, A. Brinkman, M. Smid, S. R. Johnston, L. C. Dorssers, et al. (2000). BCAR1/p130Cas expression in untreated and acquired tamoxifen-resistant human breast carcinomas. Int. J. Cancer 89:465–468.
Y. P. Cheon, Q. Li, X. Xu, F. J. DeMayo, I. C. Bagchi, and M. K. Bagchi (2002). A genomic approach to identify novel progesterone receptor regulated pathways in the uterus during implantation. Mol. Endocrinol. 16:2853–2871.
L. Tung, T. Shen, M. G. Abel, R. L. Powell, G. S. Takimoto, C. A. Sartorius, et al. (2001). Mapping the unique activation function 3 in the progesterone B-receptor upstream segment. Two LXXLL motifs and a tryptophan residue are required for activity. J. Biol. Chem. 276:39843–39851.
C. Atalay, M. Kanlioz, and M. Altinok (2002). Menstrual cycle and hormone receptor status in breast cancer patients. Neoplasma 49:278.
D. No, T. P. Yao, and R. M. Evans (1996). Ecdysone-inducible gene expression in mammalian cells and transgenic mice. Proc. Natl. Acad. Sci. U.S.A. 93:3346–3351.
C. A. Sartorius, T. J. Shen, and K. B. Horwitz (in press). Progesterone receptors A and B differentially affect the growth of estrogen-dependent human breast tumor xenografts. Breast Cancer Res. Treat.
T. K. Jenssen, W. P. Kuo, T. Stokke, and E. Hovig (2002). Associations between gene expressions in breast cancer and patient survival. Hum. Genet. 111:411–420.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jacobsen, B.M., Richer, J.K., Sartorius, C.A. et al. Expression Profiling of Human Breast Cancers and Gene Regulation by Progesterone Receptors. J Mammary Gland Biol Neoplasia 8, 257–268 (2003). https://doi.org/10.1023/B:JOMG.0000010028.48159.84
Issue Date:
DOI: https://doi.org/10.1023/B:JOMG.0000010028.48159.84