Abstract
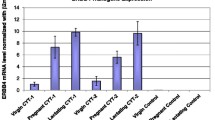
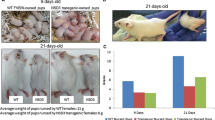
Keratin 8 and 18 are commonly used as tumorigenic markers for various types of carcinomas. They are known to be involved in cell migration, cell invasiveness, plasminogen activity and drug and radiation resistance. To ascertain a potential function for simple epithelium keratins in mammary adenocarcinoma in vivo, keratin-8-deficient mice (mK8) were mated with transgenic mice carrying the middle T oncogene driven by the MMTV promoter. The resulting mK8 knockout and control progeny carrying the middle T transgene developed mammary gland tumours with the same incidence. However, the onset of palpable mammary gland tumours occurred earlier in mK8 mutant than in control mice. This effect was prominent in males where the onset in control animals is delayed overall, because of the lower hormonal inducibility of the MMTV promoter. Metastatic foci were observed in the lungs of all females and of a few males, idependently of the genotype. Histological analysis revealed no morphological differences of the tumorigenic cells in primary tumours nor in metastatic foci. As expected, keratin 8 was absent in the mK8 tumours. Keratin 7 (mK7), keratin 18 (mK18) and keratin 19 (mK19) protein were observed in both primary and metastatic foci. These results constitute the first in vivo analysis of the role of simple epithelium keratins in mammary carcinogenesis. It demonstrates that the latency, but not the incidence nor the morphological features, of PyV middle T-induced mammary gland tumours is affected by keratin 8 deficiency
Similar content being viewed by others
References
Adams, J.M. and Cory, S. (1991) Transgenic models of tumor development. Science 254, 1161–7.
Anderson, J.M., Heindl, L.M., Bauman, P.A., Ludi, C.W., Dalton, W.S. and Cress, A.E. (1996) Cytokeratin expression results in a drug-resistent phenotype to six different chemotherapeutic agents. Clin. Cancer Res. 2, 97–105.
Baribault, H., Price, J., Miyai, K. and Oshima, R.G. (1993) Mid-gestational lethality in mice lacking keratin 8. Genes Dev. 7, 1191–202.
Baribault, H., Penner, J., Iozzo, R. and Wilson-Heiner, M. (1994) Colorectal inflammation and hyperplasia in keratin 8 deficient FVB/N mice. Genes Dev. 8, 2964–73.
Bauman, P.A., Dalton, W.S., Anderson, W.S., Anderson, J.M. and Cress, A.E. (1994) Expression of cytokeratin confers multiple drug resistance. Proc. Natl Acad. Sci. USA 91, 5311–14.
Boller, K., Kemler, R., Baribault, H. and Doetschman, T. (1987) Differential distribution of cytokeratins after microinjection of anti-cytokeratin monoclonal antibodies. Eur. J. Cell Biol. 43, 459–68.
Bonfrer, J.M.G., Groeneveld, E.M., Korse, C.M., Van Dalen, A., Oomen, L.C.J.M. and Ivanyi, D. (1994) Monoclonal antibody M3 used in tissue polypeptide-specific antigen assay for the quantification of tissue polypeptide antigen recognizes keratin 18. Tumor Biol. 15, 210–22.
Brulet, P., Babinet, C., Kemler, R. and Jacob, F. (1980) Monoclonal antibodies against trophectoderm-specific markers during mouse blastocyst formation. Proc. Natl Acad. Sci. USA 77, 4113–7.
Capetanaki, Y., Kuisk, I., Rothblum, K. and Starnes, S. (1990) Mouse vimentin: structural relationship to fos, jun, CREB and tpr. Oncogene 5, 645–55.
Chu, Y.W., Seftor, E.A., Romer, L.H. and Hendrix, M.J.C. (1996) Experimental coexpression of vimentin and keratin intermediate filaments in human melanoma cells augments motility. Am. J. Path. 148, 63–9.
Chu, Y.W., Runyan, R.B., Oshima, R.G. and Hendrix, M.J.C. (1993) Expression of complete keratin filaments in mouse L cells augments cell migration and invasion. Proc. Natl Acad. Sci. USA 90, 4261–5.
Cress, A.E. and Dalton, W.S. (1996) Multiple drug resistance and intermediate filaments. Cancer Metastasis Rev. 15, 499–506.
Einarsson, R. (1995) TPS-A cytokeratin marker for therapy control in breast cancer. Scand. J. Clin. Lab. Invest 55 Suppl 221, 113–5.
Fuchs, E. and Weber, K. (1994) Intermediate filaments: structure, dynamics function and disease. Annu. Rev. Biochem. 63, 345–82.
Guy, C., Cardiff, R.D. and Muller, W.J. (1992) Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol. Cell. Biol. 12, 954–61.
Hembrough, T.A., Vasudevan, J., Allietta, M.M., Glass, W.F., II and Gonias, S.L. (1995) A cytokeratin 8-like protein with plasminogen-binding activity is present on the external surfaces of hepatocytes, HepG2 cells and breast carcinoma cell lines. J. Cell Sci. 108, 1071–82.
Hembrough, T.A., Kralovich, K.R., Li, L. and Gonias, S.L. (1996a) Cytokeratin 8 released by breast carcinoma cells in vitro binds plasminogen and tissue-type plasminogen activator and promotes plasminogen activation. Biochem. J. 317, 763–9.
Hembrough, T.A., Li, L. and Gonias, S.L. (1996b) Cell-surface cytokeratin 8 is the major plasminogen receptor on breast cancer cells and is required for the accelerated activation of cell-associated plasminogen by tissue-type plasminogen activator. J. Biol. Chem. 271, 25684–91.
Hendrix, M.J.C., Seftor, E.A., Chu, Y.W., Seftor, R.E.B., Nagle, R.B., McDaniel, K.M., Leong, S.P.L., Yohem, K.H., Leibovitz, A.M., Meyskens, F.L.J., Conaway, D.H., Welch, D.R., Liotta, L.A. and Stetler-Stevenson, W. (1992) Coexpression of vimentin and keratins by human melanoma tumor cells: correlation with invasive and metastatic potential. J. Natl. Cancer. Inst. 84, 165–74.
Klymkowsky, M.W., Shook, D.R. and Maynell, L.A. (1992) Evidence that the deep keratin filament systems of the Xenopus embryo act to ensure normal gastrulation. Proc. Natl Acad. Sci. USA 89, 8736–40.
Lane, E.B. and Alexander, C.M. (1990) Use of keratin antibodies in tumor diagnosis. Seminars Cancer Biol. 1, 165–79.
Liao, J. and Omary, M.B. (1996) 14–3–3 proteins associate with phosphorylated simple epithelial keratins during cell cycle progression and act as a solubility cofactor. J. Cell. Biol. 133, 345–57.
Moll, R. (1994) Cytokeratins in the histological diagnosis of malignant tumors. Int. J. Biol. Markers. 9, 63–9.
Moll, R., Franke, W.W., Schiller, D.L., Geiger, B. and Krepler, R. (1982) The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells. Cell 31, 11–24.
Omary, M.B., Baxter, G.T., Chou, C.F., Riopel, C.L., Lin, W.Y. and Strulovici, B. (1992) PKC epsilon-related kinase associates with and phosphorylates cytokeratin 8 and 18. J. Cell. Biol. 117, 583–93.
Oshima, R.G., Baribault, H. and Caulin, C. (1996) Oncogenic regulation and function of keratius 8 and 18. Cancer Metastasis Rev. 15, 445–71.
Parekh, H.K. and Simpkins, H. (1995) The differential expression of cytokeratin 18 in cisplatin-sensitive and-resistant human ovarian adenocarcinoma cells and its association with drug sensitivity. Cancer Res. 55, 5203–6.
Ramaekers, F., Huijsmans, A., Schaart, G., Moesker, O and Voojis, P. (1987) Tissue-distribution of keratin 7 as monitored by a monoclonal antibody. Exp. Cell Res. 179, 235–49.
Steinert, P.M. and Roop, D.R. (1988) Molecular and cellular biology of intermediate filaments. Ann. Rev. Bioch. 57, 593–625.
Svitkina, T.M., Verkhovsky, A.B. and Borisy, G.G. (1996) Plectin sidearms mediate interaction of intermediate filaments with microtubules and other components of the cytoskeleton. J. Cell. Biol. 135, 991–1007.
Torpey, N., Wylie, C.C. and Heasman, J. (1992) Function of maternal cytokeratin in Xenopus development. Nature 357, 413–5.
Wiche et al., (1993) Cited in text.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BARIBAULT, H., WILSON-HEINER, M., MULLER, W. et al. Functional analysis of mouse keratin 8 in polyoma middle T-induced mammary gland tumours. Transgenic Res 6, 359–367 (1997). https://doi.org/10.1023/A:1018427215923
Issue Date:
DOI: https://doi.org/10.1023/A:1018427215923