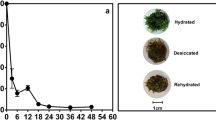
Abstract
A simple ion leakage assay was used to investigate the effect ofabscisic acid (ABA) pretreatment on desiccation tolerance in the mossAtrichum androgynum. Results from experiments involvingtheapplication of the protein synthesis inhibitor cyclohexamide during desiccationor rehydration suggested that pretreatment with ABA does not facilitate theproduction of “protection” or “repair” proteins duringdesiccation or rehydration. Rather, ABA induces the synthesis of these proteinsduring pretreatment. The ABA-induced increase in tolerance was much less ifplants were pretreated in the dark. Exposure to red light could not substitutefor white light, suggesting that ABA action does not require phytochrome.Desiccating intact long stem segments or long segments separated into apicalandbasal parts had no effect on the desiccation tolerance of either plant part.This suggests that no movement of signals or protective molecules occurs fromthe stem bases to the apices during desiccation. Basal stem segments were muchmore sensitive to desiccation than apical regions, suggesting during senescencea breakdown in mechanisms that protect mosses from injury occurs. Theimplications of these findings for the water relations of A.androgynum are discussed.
Similar content being viewed by others
References
Beckett R.P. 1999. Partial dehydration and ABA induce tolerance to desiccation-induced ion leakage in the moss Atrichum androgynum. S. Afr. J. Bot. 65: 212–279.
Beckett R.P. and Hoddinott N. 1997. Seasonal variations in tolerance to ion leakage following desiccation in the moss Atrichum androgynum from a KwaZulu-Natal afromontane forest. S. Afr. J. Bot. 63: 276–279.
Brown D.H. and Buck G.W. 1979. Desiccation effects and cation distribution in bryophytes. New Phytol. 82: 115–125.
Close T.J. 1996. Dehydrins: Emergence of a biochemical role of a family of plant dehydration proteins. Physiol. Plant 97: 795–803.
Esch H. and Lamparter T. 1998. Light regulation of phytochrome content in wild-type and aphototropic mutants of the moss Ceratodon purpureus. Photochem. Photobiol. 67: 450–455.
Gaff D.F. 1997. Mechanisms of desiccation tolerance in resurrection vascular plants. In: Basra A.S. and Basra R.K. (eds), Mechanisms of Environmental Stress Resistance in Plants. Harwood Academic Publishers, Netherlands, pp. 43–58.
Khurana J.P., Kochhar A. and Tyagi A.K. 1998. Photosensory perception and signal transduction in higher plants ? molecular genetic analysis. Crit. Rev. Pl. Sci. 17: 465–539.
Oliver M.J. and Bewley J.D. 1997. Desiccation-tolerance of plant tissues: a mechanistic overview. Hort. Rev. 18: 171–213.
Oliver M.J., Mishler B.D. and Quisenberry J.E. 1993. Comparative measures of desiccation tolerance in the Tortula ruralis complex. Variation in damage control and repair. Am. J. Bot. 80: 127–217.
Oliver M.J., Wood A.J. and O'Mahony P. 1997. How some plants recover from vegetative desiccation: a repair based strategy. Acta Physiol. Plant 19: 419–425.
Oliver M.J., Wood A.J. and O'Mahony P. 1998. 'To dryness and beyond': preparation for the dried state and rehydration in vegetative desiccation-tolerant plants. Pl. Gr. Reg. 24: 193–201.
Weatherwax S.C., Williams S.A., Tingay S. and Tobin E.M. 1998. The phytochrome response of the Lemna gibba NRP; gene is mediated primarily through changes in abscisic acid levels. Plant Physiol. 116: 1299–1305.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Beckett, R. ABA-induced tolerance to ion leakage during rehydration following desiccation in the moss Atrichum androgynum . Plant Growth Regulation 35, 131–135 (2001). https://doi.org/10.1023/A:1014477603983
Issue Date:
DOI: https://doi.org/10.1023/A:1014477603983