Abstract
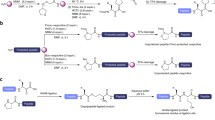
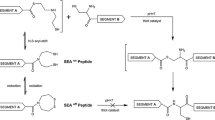
We describe, and compare, two methods which enable theassembly of proteins from peptide thioester fragmentsprepared by Fmoc chemistry mediated solid phase synthesis. The first, which utilizes iso-thiouronium salts,allows formation of thiophenyl esters directly frompartially protected peptides, either in solution or onresin. The second uses ‘sulfonamide’ based safety-catchresins. Data on yields, generality and potential for side-reactions are provided.
Similar content being viewed by others
References
Muir, T.W., Dawson, P.E. and Kent, S.B.H., Methods Enzymol., 289 (1997) 266.
Tam, J.P. and Zhang, L., In Epton, R. (Ed.), Innovation and Perspectives in Solid Phase Synthesis & Combinatorial Libraries, 1998, Collected Papers, Fifth International Symposium, London, Mayflower, Birmingham, UK, 1999, pp. 1-6.
Ingenito, R., Bianchi, E., Fattori, D. and Pessi, A., J. Am. Chem. Soc., 121 (1999) 11369.
Shin, Y., Winans, K.A., Backes, B.J., Kent, S.B.H., Ellman, J.A. and Bertozzii, C.R., J. Am. Chem. Soc., 121 (1999) 11684.
Alsina, J., Yokum, T.S., Albericio, F. and Barany, G., J. Org. Chem., 64 (1999), 8761.
Backes, B.J. and Ellman, J.A., J. Org. Chem., 64 (1999) 2322.
Kenner, G.W., McDermott, J.R. and Sheppard, R.C., J. Chem. Soc., (1971) 636.
Eilingsfeld, H., Neubauer, G., Seefelder, M. and Weidinger, H., Chem. Ber., 97 (1964) 1232.
Garner, P., Anderson, J.T., Dey, S., Youngs, W.J. and Galat, K., J. Org. Chem., 63 (1998) 5732.
Bailen, M.A., Chinchilla, R., Dodsworth, D.A. and Najera, C., J. Org. Chem., 64 (1999) 8936.
Gaube, A. and Wieland, T., Annalen, (1977) 859.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Biancalana, S., Hudson, D., Songster, M.F. et al. Fmoc chemistry compatible thio-ligation assembly of proteins. Letters in Peptide Science 7, 291–297 (2000). https://doi.org/10.1023/A:1011849912229
Issue Date:
DOI: https://doi.org/10.1023/A:1011849912229