Abstract
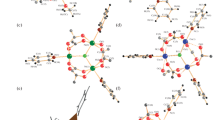
Transformations of polymeric trimethylacetate complexes [M(OH) n (OOCCMe3)2 – n ] m (M = Ni (I) and Co (II)) and clusters Ni9(μ4-OH)3(μ3-OH)3(μ-O,O-OOCCMe3)(μ-O,O"-OOCCMe3)7(μ3-O,O,O"-OOCCMe3)3(μ4-O,O,O",O"-OOCCMe3)(HOOCCMe3)4(III) and Co6(μ3-OH)2(μ-OOCCMe3)10(HOOCCMe3)4(VIII), which are formed from Iand IIupon their recrystallization from nonpolar solvents, were studied. It was shown that the action of N-phenyl-o-phenylenediamine (L) on Ior IIIresults, depending on the solvent, in different tetranuclear clusters with the hydroxo bridges. For example, in benzene, the L2Ni4(μ3-OH)2(HOOCCMe3)4(μ-OOCCMe3)6complex (IX) is formed; its L molecules are coordinated in a monodentate way, whereas in acetonitrile, they chelate to give the {[o-C6H4(NH2)(NHPh)]2Ni4(μ3-OH)2(MeCN)2(OOCCMe3)2(μ-OOCCMe3)4} compound (X). Heating of Xin the presence of atmospheric oxygen yields IX, the mononuclear bissemiquinonediimine [o-C6H4(NH)(NPh)]2Ni complex (XI), and water. It was noted that the use of aniline in these reactions affords, independent of the nature of the solvent, only one (NH2C6H5)2Ni4(μ3-OH)2(HOOCCMe3)4(μ-OOCCMe3)6cluster (VI); in acetonitrile, this cluster is formed as the solvate VI· 2HOOCCMe3(VIa). When treated with ethanol, Iand IIIgive the Ni4(EtOH)6(μ3-OH)2(μ2-OOCCMe3)4(OOCCMe3)2cluster (V), which is structurally close to the known cobalt-containing analog IV. Thermolysis of IVin decalin at 170°С causes its dimerization, giving the octanuclear Co8(μ4-O)2(μ n -OOCCMe3)12complex (VII) with the tetradentate oxo bridges.
Similar content being viewed by others
REFERENCES
Pasynskii, A.A., Torubaev, Yu.V., Drukovskii, A.V., et al., Zh. Neorg. Khim., 1997, vol. 42, no. 6, p. 957.
Holm, R.H., Kennepohl, P., and Solomon, E.I., Chem.Rev., 1996, vol. 96, p. 2239.
Pearson, M.A., Schaller, R.A., Michel, L.O., et al., Biochemistry, 1998, vol. 37, p. 6214.
Lippard, S.J., Science, 1995, vol. 268, p. 996.
Maslennikova, G.N., Mamaladze, R.A., Midzuta, S., and Koumoto, K., Keramicheskie materialy (Ceramic Materials), Moscow: Stroiizdat, 1991, p. 120.
Eremenko, I.L., Nefedov, S.E., Sidorov, A.A., et al., Inorg. Chem., 1999, vol. 38, p. 3764.
Eremenko, I.L., Nefedov, S.E., Sidorov, A.A., and Moiseev, I.I., Izv. Ross. Akad. Nauk, Ser. Khim., 1999, vol. 48, no. 3, p. 409.
Golubnichaya, M.A., Sidorov, A.A., Fomina, I.G., et al., Izv. Ross. Akad. Nauk, Ser. Khim., 1999, vol. 48, no. 9, p. 1773.
Eremenko, I.L., Golubnichaya, M.A., Nefedov, S.E., et al., Izv. Ross. Akad. Nauk, Ser. Khim., 1998, vol. 47, no. 4, p. 725.
Sidorov, A.A., Danilov, P.V., Nefedov, S.E., et al., Zh. Neorg. Khim., 1998, vol. 43, no. 6, p. 930.
Sidorov, A.A., Deomidov, S.M., Nefedov, S.E., et al., Zh. Neorg. Khim., 1999, vol. 44, no. 3, p. 396.
Novotortsev, V.M., Cand. Sci. (Chem.) Dissertation, Moscow: Inst. of General and Inorganic Chemistry, USSR Academy of Sciences, 1974.
SMART (control) and SAINT (integration) Software, Version 5.0, Bruker AXS Inc. Madison, 1997.
Sheldrick, G.M., SADABS: Program for Scaling and Correction of Area Detector Data, Göttingen: Univ. of Göttingen, 1997.
Sheldrick, G.M., SHELXS97: Program for the Solution of Crystal Structures, Göttingen: Univ. of Göttingen, 1997.
Gifeisman, G.Sh., Dvorkin, A.A., Timko, G.A., et al., Izv. Akad. Nauk Mold. SSR, Ser. Fiz.-Tekh. Mat. Nauk, 1985, no. 2, p. 22.
Golubnichaya, M.A., Sidorov, A.A., Fomina, I.G., et al., Zh. Neorg. Khim., 1999, vol. 44, no. 9, p. 1479.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sidorov, A.A., Fomina, I.G., Talismanov, S.S. et al. Formation and Transformations of Polynuclear Hydroxo- and Oxotrimethylacetate Complexes of Ni(II) and Co(II). Russian Journal of Coordination Chemistry 27, 548–559 (2001). https://doi.org/10.1023/A:1011349727749
Issue Date:
DOI: https://doi.org/10.1023/A:1011349727749