Abstract
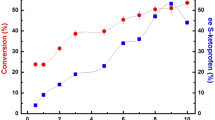
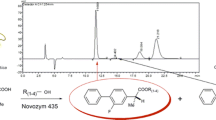
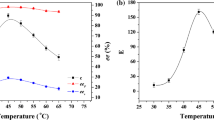
The optimal activity of a Candida rugosa lipase (Lipase OF) for hydrolysis of 2-chloroethyl ester of Ketoprofen [2-(3- benzoyphenyl) propionic acid] was at pH 4.0, while the best enantioselectivity (E) was at pH 2.2 where the enzyme was still 60% active and stable.
Similar content being viewed by others
References
Chen CS, Fujimoto Y, Girdaukas G, Sih CJ (1982) J. Am. Chem. Soc. 104: 7294–7299.
Chen P-Y, Wu S-H, Wang, K-T (1993) Biotechnol. Lett. 15: 181–184.
Cobbs CS, Barton MJ, Peng L, Goswami A, Malick AP, Hamman JP, Calton GJ (1992) US Patent 5: 108–916.
Famaey JP, Paulus, HE (1992) Therapeutic Applications of NSAIDs. New York: Marcel Dekker.
Hernaiz MJ, Sanchez-Montero JM, Sinisterra JV (1995) J. Mol. Catal. 96: 317–327.
Lam KP, Hui AHF, Jones JB (1986) J. Org. Chem. 51: 2047–2050.
Linfield WM, Barauskas RA, Sivieri L, Serota S, Stevesones RW (1984) J. Am. Oil. Chem. Soc. 61: 191–195.
Pronk W, Boswinkel G, Riet K (1992) Enzyme Microb. Technol. 14: 214–220.
Schneider M, Engel N, Honicke P, Heinemann G, Gorisch H (1984) Angew. Chem. Int. Ed. Engl. 23: 67–68.
Sohn HS, Chung SS, Rhee JS (1987) Biotechnol. Lett. 6: 623–633.
Wang YJ, Sheu JY, Wang FF, Shaw JF (1988) Biotechnol. Bioeng. 31: 628–633.
Wu SH, Guo Z-W, Sih C-J (1990) J. Am. Chem. Soc. 112: 1990–1995.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, YY., Xu, JH., Xu, QG. et al. Significant enhancement of lipase enantioselectivity toward (S)- Ketoprofen ester at pH 2. Biotechnology Letters 21, 143–146 (1999). https://doi.org/10.1023/A:1005413100578
Issue Date:
DOI: https://doi.org/10.1023/A:1005413100578