Abstract
Purpose
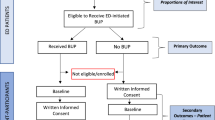
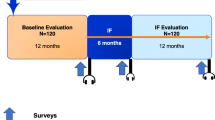
We utilized quality improvement (QI) approaches to increase emergency department (ED) provider engagement with research participant enrollment during the opioid crisis and coronavirus disease (COVID-19) pandemic. The context of this work is the Evaluating Microdosing in the Emergency Department (EMED) study, a randomized trial offering buprenorphine/naloxone to ED patients through randomization to standard or microdosing induction. Engaging providers is crucial for participant recruitment to our study. Anticipating challenges sustaining long-term engagement after a 63% decline in provider referrals four months into enrollments, we applied Plan-Do-Study-Act (PDSA) cycles to develop and implement an engagement strategy to increase and sustain provider engagement by 50% from baseline within 9 months.
Methods
Our engagement strategy was centered on Coffee Carts rounds: 5-min study-related educational presentations for providers on shift; and a secondary initiative, a Suboxone Champions program, to engage interested providers as study-related peer educators. We used provider referrals to our team as a proxy for study engagement and report the percent change in mean weekly referrals across two PDSA cycles relative to our established referral baseline.
Results
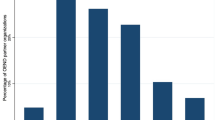
A QI approach afforded real-time review of interventions based on research and provider priorities, increasing engagement via mean weekly provider referrals by 14.5% and 49% across two PDSA cycles relative to baseline, respectively.
Conclusions
Our Coffee Carts and Suboxone Champions program are efficient, low-barrier, educational initiatives to convey study-related information to providers. This work supported our efforts to maximally engage providers, minimize burden, and provide life-saving buprenorphine/naloxone to patients at risk of fatal overdose.
Résumé
But
Nous avons utilisé des approches d’amélioration de la qualité (AQ) pour accroître l’engagement des fournisseurs des services d’urgence (SU) avec l’inscription des participants à la recherche pendant la crise des opioïdes et la pandémie de maladie à coronavirus (COVID-19). Le contexte de ce travail est l’étude Evaluating Microdosing in the Emergency Department (EMED), un essai randomisé offrant de la buprénorphine/naloxone aux patients aux urgences par randomisation à l’induction standard ou au microdosage. L’engagement des fournisseurs est crucial pour le recrutement des participants à notre étude. En anticipant les difficultés à maintenir un engagement à long terme après une baisse de 63 % des recommandations de fournisseurs quatre mois après les inscriptions, nous avons appliqué le Plan-Do-Study-Act (PDSA) cycles d’élaboration et de mise en œuvre d’une stratégie d’engagement visant à accroître et à maintenir l’engagement des fournisseurs de 50 % par rapport au niveau de référence dans les neuf mois.
Méthodes
Notre stratégie de mobilisation était axée sur les tournées de Coffee Carts : des présentations éducatives de cinq minutes sur l’étude pour les fournisseurs sur le quart de travail; et une initiative secondaire, un programme Suboxone Champions, pour mobiliser les fournisseurs intéressés en tant que pairs éducateurs liés à l’étude. Nous avons utilisé les recommandations des fournisseurs à notre équipe comme indicateur de la participation à l’étude et nous avons signalé le pourcentage de changement dans les recommandations hebdomadaires moyennes pour deux cycles PDSA par rapport à notre base de référence établie.
Résultats
Une approche d’AQ a permis d’examiner en temps réel les interventions en fonction des priorités de la recherche et des fournisseurs, ce qui a augmenté l’engagement par l’intermédiaire des recommandations hebdomadaires moyennes des fournisseurs de 14,5 % et de 49 % au cours de deux cycles de PDSA par rapport au niveau de référence, respectivement.
Conclusion
Notre programme Coffee Carts and Suboxone Champions est une initiative éducative efficace et peu contraignante qui permet de transmettre aux fournisseurs des renseignements sur les études. Ce travail a appuyé nos efforts visant à mobiliser au maximum les fournisseurs, à réduire au minimum le fardeau et à fournir de la buprénorphine/naloxone vitale aux patients à risque de surdose mortelle.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Mcrae AD, Perry JJ, Brehaut J, Brown E, Curran J, Emond M, et al. Engaging emergency clinicians in emergency department clinical research. CJEM. 2018;20(3):443–7.
Brown SG, Isbister GK. Clinical research is a priority for emergency medicine but how do we make it happen, and do it well? Emerg Med Australas. 2014;26(1):14–8.
Limkakeng AT Jr, de Oliveira LLH, Moreira T, Phadtare A, Rodrigues CG, Hocker MB, et al. Systematic review and metasummary of attitudes toward research in emergency medical conditions. J Med Ethics. 2014;40:401–8.
Gualano M, Sinigaglia T, Lo Moro G, Rousset S, Cremona A, Bert F, et al. The burden of burnout among healthcare professionals of intensive care units and emergency departments during the COVID-19 pandemic: as systematic review. Int J Environ Res Public Health. 2021;18(15):1–17.
BC Centre for Disease Control. Overdose response indictator report, March 2022. Provincial Health Services Authority. 2022. Accessed 27 Nov 2022.
Ehrlich H, McKenney M, Elkbuli A. The impact of COVID-19 pandemic on conducting emergency medicine clinical research. Am J Emerg Med. 2021;1(46):783–4.
Select Standing Committee on Health. Closing gaps, reducing barriers: Expanding the response to the toxic drug and overdose crisis Victoria: Legislative Assembly of British Columbia. 2022.
Moe J, Badke K, Pratt M, Cho RY, Azar P, Flemming H, et al. Microdosing and standard-dosing take-home buprenorphine from the emergency department: a feasibility study. J Am Coll Emerg Phys Open. 2020;1(6):1712–22.
Ogrinc G, Davies L, Goodman D, Batalden PB, Davidoff F, Stevens D. SQUIRE 2.0 (Standards for quality improvement reporting excellence): revised publication guidelines from a detailed consensus process. BMJ Qual Saf. 2016;25:986–92.
Leis JA, Shojania KG. A primer on PDSA: executing plan-do-study-act cycles in practice, not just in name. BMJ Qual Saf. 2017;26(7):572–7.
Reed JE, Card AJ. The problem with plan-do-study-act cycles. BMJ Qual Saf. 2016;25(3):147–52.
Dearing JW, Cox JG. Diffusion of innovations theory, principles, and practice. Health Aff. 2018;37(2):183–90.
Cullen L, Hanrahan K, Farrington M, Anderson R, Dimmer E, Miner R, et al. Evidence-based practice change champion program improves quality care. J Nurs Adm. 2020;50(3):128–34.
Provost LP, Muray SK. The health care data guide: learning from data for improvement, 2nd edition. 2022.
American Addictions Centers. How does suboxone work [web streaming video]. YouTube; December 21, 2018. [April 2, 2023]. Video: 2:24 min. Available from: https://www.youtube.com/watch?v=4hwtsd5RikQ
Snihur A, Mullin A, Haller A, Wiley R, Clifford P, Roposa K, et al. Fostering clinical research in the community hospital: opportunities and best practices. Healthc Q. 2020;23(2):30–6.
Moe J, Chong M, Scheuermeyer FX, Purssell R, Slaunwhite A. Death after emergency department visits for opioid overdose in British Columbia: a retrospective cohort analysis. hcq. 2021;9(1):E242–51.
Acknowledgements
This work would not be possible without the dedicated support of our Nursing Educators, ED Research Nurses, Clinical Pharmacy and Physician Leads, Suboxone Champions, ED Physicians, Addictions Medicine colleagues, and our EMED study team members. Specifically, we would like to thank Lisa Welch, Dr. Jessica Hann, Dr. Asha Olmstead, and Cindy Liu.
Funding
Health Canada/Substance Use and Addictions Program, 2021-HQ-000012, Jessica Moe, Canadian Institutes of Health Research, PJT-178119, Jessica Moe.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hussey, A., Pozsgay, K., Crawford, C.M.L. et al. Using quality improvement approaches to increase emergency department provider engagement in research participant enrollment during COVID-19 and opioid overdose public health emergencies. Can J Emerg Med 26, 349–358 (2024). https://doi.org/10.1007/s43678-024-00691-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43678-024-00691-7
Keywords
- Emergency medicine
- Healthcare research
- Opioid crisis
- COVID-19 pandemic
- Stakeholder engagement
- Quality improvement