Abstract
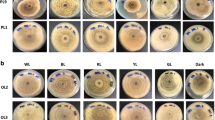
The mycelial biomass of basidiomycetes is a promising source of compounds and represents an alternative for industrial and biotechnological applications. Fungi use light as information and hold photoresponse mechanisms, in which sensors respond to light wavelengths and regulate various biological processes. Therefore, this study aimed to investigate the effects of blue, green, and red lights on the growth, chemical composition, and antioxidant and antimicrobial activity of Lentinus crinitus mycelial biomass. The chemical composition of the mycelial biomass was determined by chromatographic methods, antioxidant activity was analyzed by in vitro assays, and antimicrobial activity was investigated by the microdilution assay. The highest mycelial biomass yield was observed under blue-light cultivation. Many primordia arose under blue or green light, whereas the stroma was formed under red light. The presence of light altered the primary fungal metabolism, increasing the carbohydrate, tocopherol, fatty acid, and soluble sugar contents, mostly mannitol, and reducing the protein and organic acid concentrations. Cultivation under red light increased the phenol concentration. In contrast, cultivation under blue and green lights decreased phenol concentration. Benzoic and gallic acids were the main phenolic acids in the hydroalcoholic extracts, and the latter acids increased in all cultures under light, especially red light. Mycelial biomass cultivated under red light showed the highest antioxidant activity in the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. The ferric reducing antioxidant power (FRAP) method showed that all light wavelengths increased the antioxidant activity of mycelial biomass, with the highest value under red light. Moreover, the β-carotene/linoleic acid co-oxidation (BCLA) assay demonstrated that the antioxidant activity was affected by light cultivation. Mycelial biomass grown under all conditions exhibited antibacterial and antifungal activities. Thus, mycelial biomass cultivation of L. crinitus under light conditions may be a promising strategy for controlling the mycelial chemical composition and biomass yield.
Graphical abstract


Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Shaffique, S., Kang, S. M., Kim, A. Y., Imran, M., Aaqil Khan, M., & Lee, I. J. (2021). Current knowledge of medicinal mushrooms related to antioxidant properties. Sustainability, 13(14), 7948. https://doi.org/10.3390/su13147948
Bertéli, M. B. D., Barros, L., Reis, F. S., Ferreira, I. C., Glamočlija, J., Soković, M., Valle, J. S., Linde, G. A., Ruiz, S. P., & Colauto, N. B. (2021). Antimicrobial activity, chemical composition and cytotoxicity of Lentinus crinitus basidiocarp. Food & Function, 12(15), 6780–6792. https://doi.org/10.1039/d1fo00656h
Bertéli, M. B. D., Lopes, A. D., Colla, I. M., Linde, G. A., & Colauto, N. B. (2016). Agaricus subrufescens: Substratum nitrogen concentration and mycelial extraction method on antitumor activity. Anais da Academia Brasileira de Ciências, 88, 2239–2246. https://doi.org/10.1590/0001-3765201620160161
Mourão, F., Umeo, S. H., Bertéli, M. B. D., Lourenço, E. L., Junior, A. G., Takemura, O. S., Linde, G. A., & Colauto, N. B. (2011). Anti-inflammatory activity of Agaricus blazei in different basidiocarp maturation phases. Food and Agricultural Immunology, 22(4), 325–3333. https://doi.org/10.1080/09540105.2011.581272
Song, X., Gaascht, F., Schmidt-Dannert, C., & Salomon, C. E. (2020). Discovery of antifungal and biofilm preventative compounds from mycelial cultures of a unique North American Hericium sp. fungus. Molecules, 25(4), 963. https://doi.org/10.3390/molecules25040963
Bertéli, M. B. D., Oliveira Filho, O., Freitas, J. D., Bortolucci, W. C., Silva, G. R., Gazim, Z. C., Lívero, F. A. R., Lovato, E. C. W., Valle, J. S., Linde, G. A., Barros, L., Reis, F. S., Ferreira, I. C. F. R., Paccola-Meirelles, L. D., & Colauto, N. B. (2021). Lentinus crinitus basidiocarp stipe and pileus: Chemical composition, cytotoxicity and antioxidant activity. European Food Research and Technology, 247(6), 1355–1366. https://doi.org/10.1007/s00217-021-03713-1
Ogidi, C. O., Ubaru, A. M., Ladi-Lawal, T., Thonda, O. A., Aladejana, O. M., & Malomo, O. (2020). Bioactivity assessment of exopolysaccharides produced by Pleurotus pulmonarius in submerged culture with different agro-waste residues. Heliyon, 6(12), e05685. https://doi.org/10.1016/j.heliyon.2020.e05685
Zhu, H., Tian, L., Zhang, L., Bi, J., Song, Q., Yang, H., & Qiao, J. (2018). Preparation, characterization and antioxidant activity of polysaccharide from spent Lentinus edodes substrate. International Journal of Biological Macromolecules, 112, 976–984. https://doi.org/10.1016/j.ijbiomac.2018.01.196
Lavelli, V., Proserpio, C., Gallotti, F., Laureati, M., & Pagliarini, E. (2018). Circular reuse of bio-resources: The role of Pleurotus spp. in the development of functional foods. Food & Function, 9(3), 1353–1372. https://doi.org/10.1039/c7fo01747b
Taofiq, O., González-Paramás, A. M., Martins, A., Barreiro, M. F., & Ferreira, I. C. (2016). Mushrooms extracts and compounds in cosmetics, cosmeceuticals and nutricosmetics—A review. Industrial Crops and Products, 90, 38–48. https://doi.org/10.1016/j.indcrop.2016.06.012
Martinez-Medina, G. A., Chávez-González, M. L., Verma, D. K., Prado-Barragán, L. A., Martínez-Hernández, J. L., Flores-Gallegos, A. C., Thakur, M., Srivastav, P. P., & Aguilara, C. N. (2021). Bio-functional components in mushrooms, a health opportunity: Ergothionine and huitlacohe as recent trends. Journal of Functional Foods, 77, 104326. https://doi.org/10.1016/j.jff.2020.104326
Wu, Y., Choi, M. H., Li, J., Yang, H., & Shin, H. J. (2016). Mushroom cosmetics: The present and future. Cosmetics, 3(3), 22. https://doi.org/10.3390/cosmetics3030022
Corrochano, L. M. (2019). Light in the fungal world: From photoreception to gene transcription and beyond. Annual Review of Genetics, 53, 149–170. https://doi.org/10.1146/annurev-genet-120417-031415
Dias, L. P., Pedrini, N., Braga, G. U. L., Ferreira, P. C., Pupin, B., Araújo, C. A. S., Corrochano, L. M., & Rangel, D. E. N. (2020). Outcome of blue, green, red, and white light on Metarhizium robertsii during mycelial growth on conidial stress tolerance and gene expression. Fungal Biology, 124, 263–272. https://doi.org/10.1016/j.funbio.2019.04.007
Dias, L. P., Pupin, B., Roberts, D. W., & Rangel, D. E. N. (2022). Low- or high-white light irradiance induces similar conidial stress tolerance in Metarhizium robertsii. Archives of Microbiology, 204, 83. https://doi.org/10.1007/s00203-021-02730-8
Dias, L. P., Souza, R. K. F., Pupin, B., & Rangel, D. E. N. (2021). Conidiation under illumination enhances conidial tolerance of insect-pathogenic fungi to environmental stresses. Fungal Biology, 125, 891–904. https://doi.org/10.1016/j.funbio.2021.06.003
Yu, Z., & Fischer, R. (2019). Light sensing and responses in fungi. Nature Reviews Microbiology, 17(1), 25–36. https://doi.org/10.1038/s41579-018-0109-x
Wu, B., Xu, Z., Knudson, A., Carlson, A., Chen, N., Kovaka, S., LaButti, K., Lipzen, A., Pennachio, C., Riley, R., Schakwitz, W., Umezawa, K., Ohm, R. A., Grigoriev, I. V., Nagy, L. G., Gibbons, J., & Hibbett, D. (2018). Genomics and development of Lentinus tigrinus: A white-rot wood-decaying mushroom with dimorphic fruiting bodies. Genome Biology and Evolution, 10(12), 3250–3261. https://doi.org/10.1093/gbe/evy246
Araújo, N. L., Avelino, K. V., Halabura, M. I. W., Marim, R. A., Kassem, A. S. S., Linde, G. A., Colauto, N. B., & Valle, J. S. (2021). Use of green light to improve the production of lignocellulose-decay enzymes by Pleurotus spp. in liquid cultivation. Enzyme and Microbial Technology, 149, 109860. https://doi.org/10.1016/j.enzmictec.2021.109860
Wang, H., Tong, X., Tian, F., Jia, C., Li, C., & Li, Y. (2020). Transcriptomic profiling sheds light on the blue-light and red-light response of oyster mushroom (Pleurotus ostreatus). AMB Express, 10(1), 1–10. https://doi.org/10.1186/s13568-020-0951-x
Du, F., Zou, Y., Hu, Q., Zhang, H., & Ye, D. (2020). Comparative transcriptomic analysis reveals molecular processes involved in pileus morphogenesis in Pleurotus eryngii under different light conditions. Genomics, 112(2), 1707–1715. https://doi.org/10.1016/j.ygeno.2019.09.014
López-Legarda, X., Arboleda-Echavarría, C., Parra-Saldivar, R., Rostro-Alanis, M., Alzate, J. F., Villa-Pulgarin, J. A., & Segura-Sánchez, F. (2020). Biotechnological production, characterization and in vitro antitumor activity of polysaccharides from a native strain of Lentinus crinitus. International Journal of Biological Macromolecules, 164, 3133–3144. https://doi.org/10.1016/j.ijbiomac.2020.08.191
Meniqueti, A. B., Ruiz, S. P., Faria, M. G. I., Valle, J. S., Gonçalves, A. C., Jr., Dragunski, D. C., Colauto, N. B., & Linde, G. A. (2021). Iron bioaccumulation in Lentinus crinitus mycelia cultivated in agroindustrial byproducts. Waste and Biomass Valorization, 12(9), 4965–4974. https://doi.org/10.1007/s12649-021-01353-w
Faria, M. G. I., Avelino, K. V., Valle, J. S., Silva, G. J., Gonçalves, A. C., Jr., Dragunski, D. C., Colauto, N. B., & Linde, G. A. (2019). Lithium bioaccumulation in Lentinus crinitus mycelial biomass as a potential functional food. Chemosphere, 235, 538–542. https://doi.org/10.1016/j.chemosphere.2019.06.218
Tavares, M. F., Avelino, K. V., Araújo, N. L., Marim, R. A., Linde, G. A., Colauto, N. B., & Valle, J. S. (2020). Decolorization of azo and anthraquinone dyes by crude laccase produced by Lentinus crinitus in solid state cultivation. Brazilian Journal of Microbiology, 51(1), 99–106. https://doi.org/10.1007/s42770-019-00189-w
Silva, G. T., & Gibertoni, T. B. (2006). Aphyllophorales (Basidiomycota) em áreas urbanas da região metropolitana do Recife, PE, Brasil. Hoehnea, 33(4), 533–543.
Vargas-Isla, R., Ishikawa, N. K., & Py-Daniel, V. (2013). Contribuições etnomicológicas dos povos indígenas da Amazônia. Biota Amazônia, 3(1), 58–65. https://doi.org/10.18561/2179-5746/biotaamazonia.v3n1p58-65
Marim, R. A., Avelino, K. V., Halabura, M. I. W., Araujo, N. L., Santana, T. T., Linde, G. A., Colauto, N. B., & Valle, J. S. (2020). Lentinus crinitus response to blue light on carbohydrate-active enzymes. Bioscience Journal, 36(3), 924–931. https://doi.org/10.14393/BJ-v36n3a2020-49986
Zaghi Junior, L. L., Bertéli, M. B. D., Freitas, J. D. S., Oliveira Filho, O. B. Q., Lopes, A. D., Ruiz, S. P., Valle, J. S., Linde, G. A., & Colauto, N. B. (2020). Five-year cryopreservation at −80 °C of edible and medicinal basidiomycetes by wheat grain technique. Journal of Microbiological Methods, 176, 106030. https://doi.org/10.1016/j.mimet.2020.106030
AOAC-Association of Official Analytical Chemists. (2016). Official methods of analysis of AOAC international. AOAC International.
Ćirić, A., Kruljević, I., Stojković, D., Fernandes, Â., Barros, L., Calhelha, R. C., Ferreira, I. C. F. R., Soković, M., & Glamočlija, J. (2019). Comparative investigation on edible mushrooms Macrolepiota mastoidea, M. rhacodes and M. procera: functional foods with diverse biological activities. Food & Function, 10(12), 7678–7686. https://doi.org/10.1039/c9fo01900f
Barros, L., Pereira, C., & Ferreira, I. C. F. R. (2013). Optimized analysis of organic acids in edible mushrooms from Portugal by ultra fast liquid chromatography and photodiode array detection. Food Analytical Methods, 6(1), 309–316. https://doi.org/10.1007/s12161-012-9443-1
Spréa, R. M., Fernandes, Â., Calhelha, R. C., Pereira, C., Pires, T. C. S. P., Alves, M. J., Canan, C., Barros, L., Amaral, J. S., & Ferreira, I. C. F. R. (2020). Chemical and bioactive characterization of the aromatic plant Levisticum officinale W.D.J. Koch: A comprehensive study. Food & Function, 11, 1292–1303. https://doi.org/10.1039/C9FO02841B
Saltarelli, R., Ceccaroli, P., Iotti, M., Zambonelli, A., Buffalini, M., Casadei, L., Vallorani, L., & Stocchi, V. (2009). Biochemical characterisation and antioxidant activity of mycelium of Ganoderma lucidum from Central Italy. Food Chemistry, 116(1), 143–151. https://doi.org/10.1016/j.foodchem.2009.02.023
Palacios, I., Lozano, M., Moro, C., D’arrigo, M., Rostagno, M. A., Martínez, J. A., & Villares, A. (2011). Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chemistry, 128(3), 674–678. https://doi.org/10.1016/j.foodchem.2011.03.085
Fernandes, A., Barros, L., Antonio, A. L., Barreira, J. C. M., Oliveira, M. B. P. P., Martins, A., & Ferreira, I. C. F. R. (2014). Using gamma irradiation to attenuate the effects caused by drying or freezing in Macrolepiota procera organic acids and phenolic compounds. Food and Bioprocess Technology, 7, 3012–3021. https://doi.org/10.1007/s11947-013-1248-8
Singleton, V. L., Orthofer, R., & Lamuela-Raventós, R. M. (1999). Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In L. Packer (Ed.), Methods in enzymology (1st ed., pp. 152–178). Academic Press. https://doi.org/10.1016/S0076-6879(99)99017-1
Rufino, M. D. S., Alves, R. E., Brito, E. S., Morais, S. M., Sampaio, C. D. G., Pérez-Jimenez, J., & Saura-Calixto, F. D. (2007). Metodologia científica: Determinação da atividade antioxidante total em frutas pela captura do radical livre DPPH. Comunicado Técnico, Embrapa, 127, 1–4.
Rufino, M. D. S., Alves, R. E., Brito, E. S., Morais, S. M., Sampaio, C. D. G., Pérez-Jimenez, J., & Saura-Calixto, F. D. (2006). Metodologia científica: Determinação da atividade antioxidante total em frutas pelo método de redução do ferro (FRAP). Comunicado Técnico, Embrapa, 125, 1–4.
Rufino, M. D. S., Alves, R. E., Brito, E. S., Mancini Filho, J., & Moreira, A. V. B. (2006). Metodologia científica: Determinação da atividade antioxidante total em frutas no sistema beta-caroteno/ácido linoleico. Comunicado Técnico, Embrapa, 126, 1–4.
Kostić, M., Smiljković, M., Petrović, J., Glamočlija, J., Barros, L., Ferreira, I. C. F. R., Ćirić, A., & Soković, M. (2017). Chemical, nutritive composition and a wide range of bioactive properties of honey mushroom Armillaria mellea (Vahl: Fr.) Kummer. Food & Function, 8(9), 3239–3249. https://doi.org/10.1039/c7fo00887b
Soković, M., Glamočlija, J., Marin, P. D., Brkić, D., & van Griensven, L. J. L. D. (2010). Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules, 15, 7532–7546. https://doi.org/10.3390/molecules15117532
Tisch, D., & Schmoll, M. (2010). Light regulation of metabolic pathways in fungi. Applied Microbiology and Biotechnology, 85, 1259–1277. https://doi.org/10.1007/s00253-009-2320-1
Bayram, Ö., Feussner, K., Dumkow, M., Herrfurth, C., Feussner, I., & Braus, G. H. (2016). Changes of global gene expression and secondary metabolite accumulation during light-dependent Aspergillus nidulans development. Fungal Genetics and Biology, 87, 30–53. https://doi.org/10.1016/j.fgb.2016.01.004
Lavín, J. L., Ramírez, L., Pisabarro, A. G., & Oguiza, J. A. (2015). Genomewide analysis of phytochrome proteins in the phylum Basidiomycota. Journal of Basic Microbiology, 55(9), 1141–1147. https://doi.org/10.1002/jobm.201500078
Dasgupta, A., Fuller, K. K., Dunlap, J. C., & Loros, J. J. (2016). Seeing the world differently: Variability in the photosensory mechanisms of two model fungi. Environmental Microbiology, 18(1), 5–20. https://doi.org/10.1111/1462-2920.13055
Wang, Z., Wang, J., Li, N., Li, J., Trail, F., Dunlap, J. C., & Townsend, J. P. (2018). Light sensing by opsins and fungal ecology: NOP-1 modulates entry into sexual reproduction in response to environmental cues. Molecular Ecology, 27(1), 216–232. https://doi.org/10.1111/mec.14425
Arjona, D., Aragón, C., Aguilera, J. A., Ramírez, L., & Pisabarro, A. G. (2009). Reproducible and controllable light induction of in vitro fruiting of the white-rot basidiomycete Pleurotus ostreatus. Mycological Research, 113(5), 552–558. https://doi.org/10.1016/j.mycres.2008.12.006
Damaso, E. J., Jr., Dulay, R. M. R., Kalaw, S. P., & Reyes, R. G. (2018). Effects of color light emitting diode (led) on the mycelial growth, fruiting body production, and antioxidant activity of Lentinus tigrinus. International Journal of Science and Technology, 3(2), 9–16. https://doi.org/10.22137/ijst.2018.v3n2.02
Corrochano, L. M. (2007). Fungal photoreceptors: Sensory molecules for fungal development and behavior. Photochemical & Photobiological Sciences, 6(7), 725–736. https://doi.org/10.1039/B702155K
Kamada, T., Sano, H., Nakazawa, T., & Nakahori, K. (2010). Regulation of fruiting body photomorphogenesis in Coprinopsis cinerea. Fungal Genetics and Biology, 47(11), 917–921. https://doi.org/10.1016/j.fgb.2010.05.003
Yi, Z. L., Huang, W. F., Ren, Y., Onac, E., Zhou, G. F., Peng, S., Wang, X. J., & Li, H. H. (2014). LED lights increase bioactive substances at low energy costs in culturing fruiting bodies of Cordyceps militaris. Scientia Horticulturae, 175, 139–143. https://doi.org/10.1016/j.scienta.2014.06
Xie, C., Gong, W., Zhu, Z., Yan, L., Hu, Z., & Peng, Y. (2018). Comparative transcriptomics of Pleurotus eryngii reveals blue-light regulation of carbohydrate-active enzymes (CAZymes) expression at primordium differentiated into fruiting body stage. Genomics, 110(3), 201–209. https://doi.org/10.1016/j.ygeno.2017.09.012
Wu, J. Y., Chen, H. B., Chen, M. J., Kan, S. C., Shieh, C. J., & Liu, Y. C. (2013). Quantitative analysis of LED effects on edible mushroom Pleurotus eryngii in solid and submerged cultures. Journal of Chemical Technology & Biotechnology, 88(10), 1841–1846. https://doi.org/10.1002/jctb.4038
Barros, L., Cruz, T., Baptista, P., Estevinho, L. M., & Ferreira, I. C. F. R. (2008). Wild and commercial mushrooms as source of nutrients and nutraceuticals. Food and Chemical Toxicology, 46(8), 2742–2747. https://doi.org/10.1016/j.fct.2008.04.030
Câmara, J. S., Albuquerque, B. R., Aguiar, J., Corrêa, R. C., Gonçalves, J. L., Granato, D., Pereira, J. A. M., Barros, L., & Ferreira, I. C. F. R. (2020). Food bioactive compounds and emerging techniques for their extraction: Polyphenols as a case study. Foods, 10(1), 37. https://doi.org/10.3390/foods10010037
Dávila, G. L. R., Murillo, A. W., Zambrano, F. C. J., Suárez, M. H., & Méndez, A. J. J. (2020). Evaluation of nutritional values of wild mushrooms and spent substrate of Lentinus crinitus (L.) Fr. Heliyon, 6(3), e03502. https://doi.org/10.1016/j.heliyon.2020.e03502
Pawlik, A., Mazur, A., Wielbo, J., Koper, P., Zebracki, K., Kubik-Komar, A., & Janusz, G. (2019). RNA sequencing reveals differential gene expression of Cerrena unicolor in response to variable lighting conditions. International Journal of Molecular Sciences, 20(2), 290. https://doi.org/10.3390/ijms20020290
Corrêa, R. C. G., Souza, A. H. P., Calhelha, R. C., Barros, L., Glamočlija, J., Soković, M., Peralta, R. M., Bracht, A., & Ferreira, I. C. F. R. (2015). Bioactive formulations prepared from fruiting bodies and submerged culture mycelia of the Brazilian edible mushroom Pleurotus ostreatoroseus Singer. Food & Function, 6(7), 2155–2164. https://doi.org/10.1039/c5fo00465a
Meena, M., Prasad, V., Zehra, A., Gupta, V. K., & Upadhyay, R. S. (2015). Mannitol metabolism during pathogenic fungal–host interactions under stressed conditions. Frontiers in Microbiology, 6, 1019. https://doi.org/10.3389/fmicb.2015.01019
Stoop, J. M., & Mooibroek, H. (1998). Cloning and characterization of NADP-mannitol dehydrogenase cDNA from the button mushroom, Agaricus bisporus, and its expression in response to NaCl stress. Applied and Environmental Microbiology, 64(12), 4689–4696. https://doi.org/10.1128/AEM.64.12.4689-4696.1998
Zhao, X., Yu, C., Zhao, Y., Liu, S., Wang, H., Wang, C., Guo, L., & Chen, M. (2019). Changes in mannitol content, regulation of genes involved in mannitol metabolism, and the protective effect of mannitol on Volvariella volvacea at low temperature. BioMed Research International. https://doi.org/10.1155/2019/1493721
Casas-Flores, S., & Herrera-Estrella, A. (2016). The bright and dark sides of fungal life. In I. S. Druzhinina & C. P. Kubicek (Eds.), The mycota: Environmental and microbial relationships (3rd ed., pp. 41–77). Springer. https://doi.org/10.1007/978-3-319-29532-9_3
Liu, J. Y., Chang, M. C., Meng, J. L., Feng, C. P., Zhao, H., & Zhang, M. L. (2017). Comparative proteome reveals metabolic changes during the fruiting process in Flammulina velutipes. Journal of Agricultural and Food Chemistry, 65(24), 5091–5100. https://doi.org/10.1021/acs.jafc.7b01120
Kojima, M., Kimura, N., & Miura, R. (2015). Regulation of primary metabolic pathways in oyster mushroom mycelia induced by blue light stimulation: Accumulation of shikimic acid. Scientific Reports, 5(1), 1–7. https://doi.org/10.1155/2015/290161
Szarka, A., Tomasskovics, B., & Bánhegyi, G. (2012). The ascorbate-glutathione-α-tocopherol triad in abiotic stress response. International Journal of Molecular Sciences, 13(4), 4458–4483. https://doi.org/10.3390/ijms13044458
Petrović, J., Stojković, D., Reis, F. S., Barros, L., Glamočlija, J., Ćirić, A., Ferreira, I. C. F. R., & Soković, M. (2014). Study on chemical, bioactive and food preserving properties of Laetiporus sulphureus (Bull.: Fr.) Murr. Food & Function, 5(7), 1441–1451.
Kalač, P. (2013). A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. Journal of the Science of Food and Agriculture, 93(2), 209–218. https://doi.org/10.1002/jsfa.5960
Pinto, S., Barros, L., Sousa, M. J., & Ferreira, I. C. F. R. (2013). Chemical characterization and antioxidant properties of Lepista nuda fruiting bodies and mycelia obtained by in vitro culture: Effects of collection habitat and culture media. Food Research International, 51(2), 496–502. https://doi.org/10.1016/j.foodres.2013.01.009
Chen, C. H., Ringelberg, C. S., Gross, R. H., Dunlap, J. C., & Loros, J. J. (2009). Genome-wide analysis of light-inducible responses reveals hierarchical light signalling in Neurospora. EMBO Journal, 28(8), 1029–1042. https://doi.org/10.1038/emboj.2009.54
Zheng, W., Zhang, M., Zhao, Y., Miao, K., & Jiang, H. (2009). NMR-based metabolomic analysis on effect of light on production of antioxidant phenolic compounds in submerged cultures of Inonotus obliquus. Bioresource Technology, 100(19), 4481–4487. https://doi.org/10.1016/j.biortech.2009.04.027
Gornostai, T. Y. G., Borovskii, G. G., Kashchenko, N. I., & Olennikov, D. N. (2018). Phenolic compounds of Inonotus rheades (Agaricomycetes) mycelium: RP-UPLC-DAD-ESI/MS profile and effect of light wavelength on styrylpyrone content. International Journal of Medicinal Mushrooms, 20(7), 637–645. https://doi.org/10.1615/intjmedmushrooms.2018026595
Balasundram, N., Sundram, K., & Samman, S. (2006). Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chemistry, 99(1), 191–203. https://doi.org/10.1016/j.foodchem.2005.07.042
Heleno, S. A., Barros, L., Martins, A., Queiroz, M. J. R., Santos-Buelga, C., & Ferreira, I. C. F. R. (2012). Fruiting body, spores and in vitro produced mycelium of Ganoderma lucidum from Northeast Portugal: A comparative study of the antioxidant potential of phenolic and polysaccharidic extracts. Food Research International, 46(1), 135–140. https://doi.org/10.1016/j.foodres.2011.12.009
Carvajal, A. E. S., Koehnlein, E. A., Soares, A. A., Eler, G. J., Nakashima, A. T., Bracht, A., & Peralta, R. M. (2011). Bioactives of fruiting bodies and submerged culture mycelia of Agaricus brasiliensis (A. blazei) and their antioxidant properties. LWT-Food Science and Technology, 46(2), 493–499. https://doi.org/10.1016/j.lwt.2011.11.018
Reis, F. S., Barros, L., Martins, A., & Ferreira, I. C. F. R. (2012). Chemical composition and nutritional value of the most widely appreciated cultivated mushrooms: An inter-species comparative study. Food and Chemical Toxicology, 50(2), 191–197. https://doi.org/10.1016/j.fct.2011.10.056
Brand-Williams, W., Cuvelier, M. E., & Berset, C. L. W. T. (1995). Use of a free radical method to evaluate antioxidant activity. LWT-Food Science and Technology, 28(1), 25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Badhani, B., Sharma, N., & Kakkar, R. (2015). Gallic acid: A versatile antioxidant with promising therapeutic and industrial applications. RSC Advances, 5(35), 27540–27557. https://doi.org/10.1039/C5RA01911G
González-Palma, I., Escalona-Buendía, H. B., Ponce-Alquicira, E., Téllez-Téllez, M., Gupta, V. K., Díaz-Godínez, G., & Soriano-Santos, J. (2016). Evaluation of the antioxidant activity of aqueous and methanol extracts of Pleurotus ostreatus in different growth stages. Frontiers in Microbiology, 7, 1099. https://doi.org/10.3389/fmicb.2016.01099
Dundar, A., Okumus, V., Ozdemir, S., & Yildiz, A. (2013). Antioxidant properties of cultured mycelia from four Pleurotus species produced in submerged medium. International Journal of Food Properties, 16(5), 1105–1116. https://doi.org/10.1080/10942912.2011.576793
Bertéli, M. B. D., Souza, M. M. M., Barros, L., Ferreira, I. C. F. R., Glamočlija, J., Soković, M., Dragunski, D. C., Valle, J. S., Ferreira, E. S., Pinto, L. C., Souza, C. O., Ruiz, S. P., Linde, G. A., & Colauto, N. B. (2022). Basidiocarp structures of Lentinus crinitus: An antimicrobial source against foodborne pathogens and food spoilage microorganisms. World Journal of Microbiology and Biotechnology, 38(5), 74. https://doi.org/10.1007/s11274-022-03257-w
Heleno, S. A., Ferreira, I. C. F. R., Esteves, A. P., Ćirić, A., Glamočlija, J., Martins, A., Soković, M., & Queiroz, M. J. R. P. (2013). Antimicrobial and demelanizing activity of Ganoderma lucidum extract, p-hydroxybenzoic and cinnamic acids and their synthetic acetylated glucuronide methyl esters. Food and Chemical Toxicology, 58, 95–100. https://doi.org/10.1016/j.fct.2013.04.02
Acknowledgements
The authors thank the Universidade Paranaense (36900/2020), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brazil (CAPES), Fundação Araucária, and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, 307953/2017-3) for financial support and fellowships. The authors are also grateful to the Foundation for Science and Technology (FCT, Portugal) for financial support from the national funds FCT/MCTES to CIMO (UIDB/00690/2020) and P.I. for the institutional scientific employment program contract for L.B. and A.F. This research was also funded by the Serbian Ministry of Education, Science, and Technological Development (Contract No. 451-03-68/2022-14/200007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Halabura, M.I.W., Avelino, K.V., Araújo, N.L. et al. Light conditions affect the growth, chemical composition, antioxidant and antimicrobial activities of the white-rot fungus Lentinus crinitus mycelial biomass. Photochem Photobiol Sci 22, 669–686 (2023). https://doi.org/10.1007/s43630-022-00344-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-022-00344-7