Abstract
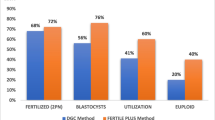
Oocytes play a crucial role in repairing sperm DNA damage, which can affect the next generation; however, certain factors can impair this ability. This study examined whether oocyte vitrification, a widely used method for fertility preservation, negatively affects repair ability. Male DBA/2 mice (n = 28) were injected with 101.60 µmol/100 g body weight of tert-Butyl hydroperoxide (tBHP) for 14 days to induce sperm DNA damage. Histological changes, sperm functions, and DNA fragmentation were assessed using the TUNEL assay. Cumulus-oocyte-complexes (COCs) of superovulated female DBA/2 mice (n = 28) were vitrified using the Cryotop method. Fresh and vitrified oocytes were then fertilized by tBHP-treated and untreated sperms, and subsequent embryonic development was monitored. Additionally, the expression of Mre11a, Rad51, Brca1, and Xrcc4 was assessed in resulting zygotes and blastocysts using real-time PCR. The sperm tBHP treatment reduced differentiated spermatogenic cells in the testicular tissue, sperm concentration, and motility, while increasing DNA fragmentation (P < 0.05). The fertilization rate was decreased in the tBHP-treated sperm–vitrified oocyte group (P < 0.05), and the two-cell rate diminished in tBHP-treated sperm–fresh and vitrified oocyte groups (P < 0.05). The four-cell to blastocyst rate decreased in the untreated sperm–vitrified oocyte and the tBHP-treated sperm–fresh and vitrified oocyte groups (P < 0.05), and the tBHP-treated sperm–vitrified oocyte groups had the lowest blastocyst rate. In zygotes, Brca1 was upregulated in the tBHP-treated sperm–vitrified oocyte group (P < 0.05). Also, in blastocysts, Rad51, Brca1, and Xrcc4 were significantly upregulated in the untreated sperm–vitrified oocytes group (P < 0.05). Damages to the oocyte due to vitrification can disrupt the repair of sperm DNA fragmentation and consequently impair the embryo development.




Similar content being viewed by others
References
Martin JH, Aitken RJ, Bromfield EG, Nixon B. DNA damage and repair in the female germline: contributions to ART. Hum Reprod Update. 2019;25:180–201.
Gao Z, Moorjani P, Sasani TA, Pedersen BS, Quinlan AR, Jorde LB, et al. Overlooked roles of DNA damage and maternal age in generating human germline mutations. Proc Natl Acad Sci U S A. 2019;116:9491–500.
Immler S. The sperm factor: paternal impact beyond genes. Heredity (Edinb). 2018;121:239–47 (Springer US).
Henkel R, Leisegang K. Origins of sperm DNA damage. In: Parekattil S, Esteves S, Agarwal A, (eds.) Male infertility. Springer: Cham; 2020. p. 361–75.
Esteves SC, Santi D, Simoni M. An update on clinical and surgical interventions to reduce sperm DNA fragmentation in infertile men. Andrology England. 2020;8:53–81.
Wright C, Milne S, Leeson H. Sperm DNA damage caused by oxidative stress: modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod Biomed Online Netherlands. 2014;28:684–703.
González-Marín C, Gosálvez J, Roy R. Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int J Mol Sci. 2012;13:14026–52.
Drevet JR, Aitken RJ. Oxidation of sperm nucleus in mammals: a physiological necessity to some extent with adverse impacts on oocyte and offspring. Antioxidants (Basel). 2020;9(2):95. https://doi.org/10.3390/antiox9020095.
Aitken RJ. Role of sperm DNA damage in creating de-novo mutations in human offspring: the ‘post-meiotic oocyte collusion’ hypothesis. Reprod Biomed Online. 2022;45:109–24. https://www.sciencedirect.com/science/article/pii/S1472648322001493.
Stringer JM, Winship A, Zerafa N, Wakefield M, Hutt K. Oocytes can efficiently repair DNA double-strand breaks to restore genetic integrity and protect offspring health. Proc Natl Acad Sci U S A. 2020;117:1–10.
Thompson LH, Schild D. Recombinational DNA repair and human disease. Mutat Res - Fundam Mol Mech Mutagen. 2002;509:49–78.
Mitchell LA, De Iuliis GN, Aitken RJ. The TUNEL assay consistently underestimates DNA damage in human spermatozoa and is influenced by DNA compaction and cell vitality: development of an improved methodology. Int J Androl England. 2011;34:2–13.
Newman H, Catt S, Vining B, Vollenhoven B, Horta F. DNA repair and response to sperm DNA damage in oocytes and embryos, and the potential consequences in ART: a systematic review. Mol Hum Reprod [Internet]. 2022;28:gaab071. https://doi.org/10.1093/molehr/gaab071.
Sasaki H, Hamatani T, Kamijo S, Iwai M, Kobanawa M, Ogawa S, et al. Impact of oxidative stress on age-associated decline in oocyte developmental competence. Front Endocrinol (Lausanne). 2019;10:811.
Leem J, Bai G-Y, Oh JS. The capacity to repair sperm DNA damage in zygotes is enhanced by inhibiting WIP1 activity. Front cell Dev Biol. 2022;10:841327.
Derijck A, van der Heijden G, Giele M, Philippens M, de Boer P. DNA double-strand break repair in parental chromatin of mouse zygotes, the first cell cycle as an origin of de novo mutation. Hum Mol Genet England. 2008;17:1922–37.
Jaroudi S, SenGupta S. DNA repair in mammalian embryos. Mutat Res Netherlands. 2007;635:53–77.
Thompson LH, Schild D. Recombinational DNA repair and human disease. Mutat Res Netherlands. 2002;509:49–78.
Powell MD, Manandhar G, Spate L, Sutovsky M, Zimmerman S, Sachdev SC, et al. Discovery of putative oocyte quality markers by comparative ExacTag proteomics. Proteomics Clin Appl. 2010;4:337–51.
Esbert M, Pacheco A, Soares SR, Amorós D, Florensa M, Ballesteros A, et al. High sperm DNA fragmentation delays human embryo kinetics when oocytes from young and healthy donors are microinjected. Andrology England. 2018;6:697–706.
Horta F, Catt S, Ramachandran P, Vollenhoven B, Temple-Smith P. Female ageing affects the DNA repair capacity of oocytes in IVF using a controlled model of sperm DNA damage in mice. Hum Reprod. 2020;35:529–44.
Horta F, Ravichandran A, Catt S, Vollenhoven B, Temple-Smith P. Ageing and ovarian stimulation modulate the relative levels of transcript abundance of oocyte DNA repair genes during the germinal vesicle-metaphase II transition in mice. J Assist Reprod Genet. 2021;38:55–69.
Martin JH, Bromfiel EG, Aitken RJ, Lord T, Nixon B. Double strand break DNA repair occurs via non-homologous end- joining in mouse MII oocytes. Sci Rep. 2018;8:9685.
López A, Ducolomb Y, Casas E, Retana-Márquez S, Betancourt M, Casillas F. Effects of porcine immature oocyte vitrification on actin microfilament distribution and chromatin integrity during early embryo development in vitro. Front cell Dev Biol. 2021;9:636765.
Turathum B, Roytrakul S, Changsangfa C, Sroyraya M, Tanasawet S, Kitiyanant Y, et al. Missing and overexpressing proteins in domestic cat oocytes following vitrification and in vitro maturation as revealed by proteomic analysis. Biol Res. 2018;51:27.
Rienzi L, Gracia C, Maggiulli R, LaBarbera AR, Kaser DJ, Ubaldi FM, et al. Oocyte, embryo and blastocyst cryopreservation in ART: systematic review and meta-analysis comparing slow-freezing versus vitrification to produce evidence for the development of global guidance. Hum Reprod Update. 2017;23:139–55.
Chang H, Chen H, Zhang L, Wang Y, Xie X, Zhang Y, et al. Effect of oocyte vitrification on DNA damage in metaphase II oocytes and the resulting preimplantation embryos. Mol Reprod Dev United States. 2019;86:1603–14.
Matilla E, Martín-Cano FE, González-Fernández L, Sánchez-Margallo FM, Álvarez IS, MacÍas-García B. N-acetylcysteine addition after vitrification improves oocyte mitochondrial polarization status and the quality of embryos derived from vitrified murine oocytes. BMC Vet Res. 2019;15:31.
Lee HH, Lee HJ, Kim HJ, Lee JH, Ko Y, Kim SM, et al. Effects of antifreeze proteins on the vitrification of mouse oocytes: comparison of three different antifreeze proteins. Hum Reprod. 2015;30:2110–9.
Pereira BC, Ortiz I, Dorado JM, Diaz-Jimenez MA, Consuegra C, Gosalvez J, et al. Effect of permeable cryoprotectant-free vitrification on DNA fragmentation of equine oocyte–cumulus cells. Reprod Domest Anim. 2019;54:53–6.
Zhao X-M, Du W-H, Wang D, Hao H-S, Liu Y, Qin T, et al. Effect of cyclosporine pretreatment on mitochondrial function in vitrified bovine mature oocytes. Fertil Steril. 2011;95:2786–8 United States.
Wu Z, Pan B, Qazi IH, Yang H, Guo S, Yang J, Zhang Y, Zeng C, Zhang M, Han H, Meng Q, Zhou G. Melatonin improves in vitro development of vitrified-warmed mouse germinal vesicle oocytes potentially via modulation of spindle assembly checkpoint-related genes. Cells. 2019;8(9):1009. https://doi.org/10.3390/cells8091009.
Monzo C, Haouzi D, Roman K, Assou S, Dechaud H, Hamamah S. Slow freezing and vitrification differentially modify the gene expression profile of human metaphase II oocytes. Hum Reprod. 2012;27:2160–8.
Khodavandpour Z, Zavareh S, Farrokh P, Nasiri M. Assessment of DNA repair gene expressions in vitrified mouse preantral follicles. Cell J. 2020;22:81–8.
Somfai T, Haraguchi S, Dang-Nguyen TQ, Kaneko H, Kikuchi K. Vitrification of porcine immature oocytes and zygotes results in different levels of DNA damage which reflects developmental competence to the blastocyst stage. PLoS One. 2023;18:e0282959.
Baniasadi F, Hajiaghalou S, Shahverdi A, Pirhajati V, Fathi R. Static magnetic field halves cryoinjuries of vitrified mouse COCs, improves their functions and modulates pluripotency of derived blastocysts. Theriogenology. 2021;163:31–42.
Baniasadi F, Hajiaghalou S, Shahverdi A, et al. The beneficial effects of static magnetic field and iron oxide nanoparticles on the vitrification of mature mice oocytes. Reproductive Sciences. 2023;30(7):2122–2136. https://doi.org/10.1007/s43032-022-01144-1.
Fatemi N, Sanati MH, Jamali Zavarehei M, Ayat H, Esmaeili V, Golkar-Narenji A, et al. Effect of tertiary-butyl hydroperoxide (TBHP)-induced oxidative stress on mice sperm quality and testis histopathology. Andrologia. 2013;45:232–9.
Fatemi N, Sanati MH, Shamsara M, Moayer F, Zavarehei MJ, Pouya A, et al. TBHP-induced oxidative stress alters microRNAs expression in mouse testis. J Assist Reprod Genet. 2014;31:1287–93.
Malatesta M. Histological and histochemical methods - theory and practice. Eur J Histochem. 2016;60. Available from: https://www.ejh.it/index.php/ejh/article/view/2639.
Stringer JM, Winship A, Liew SH, Hutt K. The capacity of oocytes for DNA repair. Cell Mol Life Sci. 2018;75:2777–92. https://doi.org/10.1007/s00018-018-2833-9. Springer International Publishing.
Ješeta M, Myšková M, Žáková J, Crha I, Crha K, Chmelikova E, et al. Can oocytes repair fragmented DNA of spermatozoa? Med J Cell Biol. 2020;8:73–7.
Ribas-Maynou J, Benet J. Single and double strand sperm DNA damage: Different reproductive effects on male fertility. Genes (Basel). 2019;10(2):105. https://doi.org/10.3390/genes10020105.
Winship AL, Stringer JM, Liew SH, Hutt KJ. The importance of DNA repair for maintaining oocyte quality in response to anti-cancer treatments, environmental toxins and maternal ageing. Hum Reprod Update. 2018;24:119–34.
Sies H. Glutathione and its role in cellular functions. Free Radic Biol Med. 1999;27:916–21.
Chetty CS, Vemuri MC, Campbell K, Suresh C. Lead-induced cell death of human neuroblastoma cells involves GSH deprivation. Cell Mol Biol Lett. 2005;10:413–23.
Kaur P, Kaur G, Bansal MP. Upregulation of AP1 by tertiary butyl hydroperoxide induced oxidative stress and subsequent effect on spermatogenesis in mice testis. Mol Cell Biochem. 2008;308:177–81.
Circu ML, Aw TY. Glutathione and modulation of cell apoptosis. Biochim Biophys Acta. 2012;1823:1767–77.
Seli E, Gardner DK, Schoolcraft WB, Moffatt O, Sakkas D. Extent of nuclear DNA damage in ejaculated spermatozoa impacts on blastocyst development after in vitro fertilization. Fertil Steril. 2004;82:378–83.
Li Z, Wang L, Cai J, Huang H. Correlation of sperm DNA damage with IVF and ICSI outcomes: a systematic review and meta-analysis. J Assist Reprod Genet. 2006;23:367–76.
Lewis SEM, Aitken RJ, Conner SJ, De Iuliis G, Evenson DP, Henkel R, et al. The impact of sperm DNA damage in assisted conception and beyond: recent advances in diagnosis and treatment. Reprod Biomed Online. 2013;27:325–37.
Paul C, Murray AA, Spears N, Saunders PTK. A single, mild, transient scrotal heat stress causes DNA damage, subfertility and impairs formation of blastocysts in mice. Reproduction. 2008;136:73–84.
Fatehi AN, Bevers MM, Schoevers E, Roelen BAJ, Colenbrander B, Gadella BM. DNA damage in bovine sperm does not block fertilization and early embryonic development but induces apoptosis after the first cleavages. J Androl. 2006;27:176–88.
Sun MH, Yang M, Xie FY, Wang W, Zhang L, Shen W, Yin S, Ma JY. DNA Double-strand breaks induce the nuclear actin filaments formation in cumulus-enclosed oocytes but not in denuded oocytes. PLoS One. 2017;12(1):e0170308. https://doi.org/10.1371/journal.pone.0170308.
De Gheselle S, De Sutter P, Tilleman K. In-vitro development of embryos derived from vitrified-warmed oocytes is delayed compared with embryos derived from fresh oocytes: a time-lapse sibling oocyte study. Reprod Biomed Online. 2020;40:82–90.
Sudiman J, Lee A, Ong KL, Yuan WZ, Jansen S, Temple-Smith P, et al. Tolerance of lamb and mouse oocytes to cryoprotectants during vitrification. Zygote England. 2019;27:36–45.
Wang N, Hao H-S, Li C-Y, Zhao Y-H, Wang H-Y, Yan C-L, et al. Calcium ion regulation by BAPTA-AM and ruthenium red improved the fertilisation capacity and developmental ability of vitrified bovine oocytes. Sci Rep. 2017;7:10652.
Somfai T, Ozawa M, Noguchi J, Kaneko H, Kuriani Karja NW, Farhudin M, et al. Developmental competence of in vitro-fertilized porcine oocytes after in vitro maturation and solid surface vitrification: effect of cryopreservation on oocyte antioxidative system and cell cycle stage. Cryobiology. 2007;55:115–26.
Khokhlova EV, Fesenko ZS, Sopova JV, Leonova EI. Features of DNA repair in the early stages of mammalian embryonic development. Genes (Basel). 2020;11(10):1138. https://doi.org/10.3390/genes11101138.
Musson R, Gąsior Ł, Bisogno S, Ptak GE. DNA damage in preimplantation embryos and gametes: specification, clinical relevance and repair strategies. Hum Reprod Update. 2022;28(3):376–99. https://doi.org/10.1093/humupd/dmab046.
Jaroudi S, Kakourou G, Cawood S, Doshi A, Ranieri DM, Serhal P, et al. Expression profiling of DNA repair genes in human oocytes and blastocysts using microarrays. Hum Reprod [Internet]. 2009;24:2649–55. https://doi.org/10.1093/humrep/dep224.
Stigliani S, Moretti S, Anserini P, Casciano I, Venturini PL, Scaruffi P. Storage time does not modify the gene expression profile of cryopreserved human metaphase II oocytes. Hum Reprod England. 2015;30:2519–26.
Yan CT, Kaushal D, Murphy M, Zhang Y, Datta A, Chen C, et al. XRCC4 suppresses medulloblastomas with recurrent translocations in p53-deficient mice. Proc Natl Acad Sci U S A. 2006;103:7378–83 (United States).
Shirazi A, Naderi MM, Hassanpour H, Heidari M, Borjian S, Sarvari A, et al. The effect of ovine oocyte vitrification on expression of subset of genes involved in epigenetic modifications during oocyte maturation and early embryo development. Theriogenology. 2016;86:2136–46 (United States).
Acknowledgements
The authors thank all colleagues in the embryology lab, sperm biology lab, animal science lab, molecular lab, and histology lab of Royan Institute.
Funding
This project (96000285) was funded by Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine.
Author information
Authors and Affiliations
Contributions
N.K. conducted all of the experimental work and drafted the manuscript. R.F. designed and supervised the study and interpreted results, mentioned fundamental advice to develop the experiment, and reviewed the manuscript. V.A. ran a statistical analysis, mentioned fundamental advice to develop the experiment, and reviewed the manuscript. H.G. proposed the hypothesis, designed and supervised the study and interpreted results, mentioned fundamental advice to develop the experiment, and reviewed the manuscript. All authors contributed to the report.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khajedehi, N., Fathi, R., Akbarinejad, V. et al. Oocyte Vitrification Reduces its Capability to Repair Sperm DNA Fragmentation and Impairs Embryonic Development. Reprod. Sci. (2023). https://doi.org/10.1007/s43032-023-01419-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43032-023-01419-1