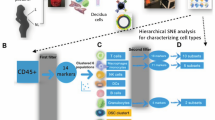
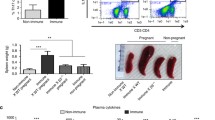
Abstract
The placenta utilizes many mechanisms to protect the haploidentical fetus from recognition by the maternal immune system. However, in cases of villitis of unknown etiology (VUE), maternal lymphocytes gain access into the placenta, causing significant health risks for the fetus. Evidence suggests that VUE is a rejection response between the mother and the haploidentical fetus. Therefore, we profiled human leukocyte antigen (HLA), an important predictor of transplant rejection, in VUE using placental tissue from ten patients with VUE and ten gestational age matched controls. Placentas were stained using novel multiplexed immunofluorescence (MxIF) to investigate morphology and HLA classes I and II. Gene expression was evaluated by microarray, and where available, tissue typing of mother/baby pairs was completed to determine HLA type. MxIF demonstrated strong CD8+ T cell infiltration and HLA class I staining both the distal and stem villi of VUE placentas. Compared to controls, VUE cases had significantly higher expression of HLA class II mRNA and pathway analysis demonstrated that 40% of the differentially expressed genes in VUE are related to tissue rejection. The data suggest that VUE resembles a rejection response between the mother and the fetus. It remains unknown what initiates immune recognition and why some mothers appear to be at higher risk for developing this condition than others. Understanding this etiology will be critical for developing effective interventions or prevention strategies during pregnancy.




Similar content being viewed by others
References
Holtan SG, Creedon DJ, Haluska P, Markovic SN. Cancer and pregnancy: parallels in growth, invasion, and immune modulation and implications for cancer therapeutic agents. Mayo Clin Proc. 2009;84(11):985–1000.
Enninga EA, Holtan SG, Creedon DJ, Dronca RS, Nevala WK, Ognjanovic S, et al. Immunomodulatory effects of sex hormones: requirements for pregnancy and relevance in melanoma. Mayo Clin Proc. 2014;89(4):520–35.
Redman CW, McMichael AJ, Stirrat GM, Sunderland CA, Ting A. Class 1 major histocompatibility complex antigens on human extra-villous trophoblast. Immunology. 1984;52(3):457–68.
Hunt JS, Orr HT. HLA and maternal-fetal recognition. FASEB J. 1992;6(6):2344–8.
Boog G. Chronic villitis of unknown etiology. Eur J Obstet Gynecol Reprod Biol. 2008;136(1):9–15.
Redline RW. Villitis of unknown etiology: noninfectious chronic villitis in the placenta. Hum Pathol. 2007;38(10):1439–46.
Becroft DM, Thompson JM, Mitchell EA. Placental villitis of unknown origin: epidemiologic associations. Am J Obstet Gynecol. 2005;192(1):264–71.
Knox WF, Fox H. Villitis of unknown aetiology: its incidence and significance in placentae from a British population. Placenta. 1984;5(5):395–402.
Salafia CM, Vogel CA, Vintzileos AM, Bantham KF, Pezzullo J, Silberman L. Placental pathologic findings in preterm birth. Am J Obstet Gynecol. 1991;165(4 Pt 1):934–8.
Redline RW, Abramowsky CR. Clinical and pathologic aspects of recurrent placental villitis. Hum Pathol. 1985;16(7):727–31.
Redline RW. Severe fetal placental vascular lesions in term infants with neurologic impairment. Am J Obstet Gynecol. 2005;192(2):452–7.
Garcia AG, Basso NG, Fonseca ME, Zuardi JA, Outanni HN. Enterovirus associated placental morphology: a light, virological, electron microscopic and immunohistologic study. Placenta. 1991;12(5):533–47.
Ernst LM, Crouch J, Rinder H, Howe JG. Bacterial etiology for chronic villitis is not supported by polymerase chain reaction for 16S rRNA DNA. Pediatr Dev Pathol. 2005;8(6):647–53.
Redline RW, Patterson P. Villitis of unknown etiology is associated with major infiltration of fetal tissue by maternal inflammatory cells. Am J Pathol. 1993;143(2):473–9.
Labarrere CA, Catoggio LJ, Mullen EG, Althabe OH. Placental lesions in maternal autoimmune diseases. Am J Reprod Immunol Microbiol. 1986;12(3):78–86.
Magid MS, Kaplan C, Sammaritano LR, Peterson M, Druzin ML, Lockshin MD. Placental pathology in systemic lupus erythematosus: a prospective study. Am J Obstet Gynecol. 1998;179(1):226–34.
Styer AK, Parker HJ, Roberts DJ, Palmer-Toy D, Toth TL, Ecker JL. Placental villitis of unclear etiology during ovum donor in vitro fertilization pregnancy. Am J Obstet Gynecol. 2003;189(4):1184–6.
Schonkeren D, Swings G, Roberts D, Claas F, de Heer E, Scherjon S. Pregnancy close to the edge: an immunosuppressive infiltrate in the chorionic plate of placentas from uncomplicated egg cell donation. PLoS One. 2012;7(3):e32347.
Labarrere C, Althabe O. Chronic villitis of unknown aetiology in recurrent intrauterine fetal growth retardation. Placenta. 1987;8(2):167–73.
Khong TY, Mooney EE, Ariel I, et al. Sampling and definitions of placental lesions: Amsterdam placental workshop group consensus statement. Arch Pathol Lab Med. 2016;140(7):698–713.
Gerdes MJ, Sevinsky CJ, Sood A, Adak S, Bello MO, Bordwell A, et al. Highly multiplexed single-cell analysis of formalin-fixed, paraffin-embedded cancer tissue. Proc Natl Acad Sci U S A. 2013;110(29):11982–7.
Derricott H, Jones RL, Greenwood SL, Batra G, Evans MJ, Heazell AE. Characterizing Villitis of unknown etiology and inflammation in stillbirth. Am J Pathol. 2016;186(4):952–61.
Kim JS, Romero R, Kim MR, Kim YM, Friel L, Espinoza J, et al. Involvement of Hofbauer cells and maternal T cells in villitis of unknown aetiology. Histopathology. 2008;52(4):457–64.
Reyes L, Golos TG. Hofbauer cells: their role in healthy and complicated pregnancy. Front Immunol. 2018;9:2628.
Myerson D, Parkin RK, Benirschke K, Tschetter CN, Hyde SR. The pathogenesis of villitis of unknown etiology: analysis with a new conjoint immunohistochemistry-in situ hybridization procedure to identify specific maternal and fetal cells. Pediatr Dev Pathol. 2006;9(4):257–65.
Kim MJ, Romero R, Kim CJ, et al. Villitis of unknown etiology is associated with a distinct pattern of chemokine up-regulation in the feto-maternal and placental compartments: implications for conjoint maternal allograft rejection and maternal anti-fetal graft-versus-host disease. J Immunol. 2009;182(6):3919–27.
Yusuf K, Kliman HJ. The fetus, not the mother, elicits maternal immunologic rejection: lessons from discordant dizygotic twin placentas. J Perinat Med. 2008;36(4):291–6.
Lee J, Romero R, Xu Y, Kim JS, Topping V, Yoo W, et al. A signature of maternal anti-fetal rejection in spontaneous preterm birth: chronic chorioamnionitis, anti-human leukocyte antigen antibodies, and C4d. PLoS One. 2011;6(2):e16806.
Rudzinski E, Gilroy M, Newbill C, Morgan T. Positive C4d immunostaining of placental villous syncytiotrophoblasts supports host-versus-graft rejection in villitis of unknown etiology. Pediatr Dev Pathol. 2013;16(1):7–13.
AL K, Kim YW, Shim JY, et al. Distinct patterns of C4d immunoreactivity in placentas with villitis of unknown etiology, cytomegaloviral placentitis, and infarct. Placenta. 2013;34(5):432–5.
Tamblyn JA, Lissauer DM, Powell R, Cox P, Kilby MD. The immunological basis of villitis of unknown etiology - review. Placenta. 2013;34(10):846–55.
Lannaman K, Romero R, Chaiworapongsa T, Kim YM, Korzeniewski SJ, Maymon E, et al. Fetal death: an extreme manifestation of maternal anti-fetal rejection. J Perinat Med. 2017;45(7):851–68.
Derricott H, Jones RL, Heazell AE. Investigating the association of villitis of unknown etiology with stillbirth and fetal growth restriction - a systematic review. Placenta. 2013;34(10):856–62.
Ptacek I, Sebire NJ, Man JA, Brownbill P, Heazell AE. Systematic review of placental pathology reported in association with stillbirth. Placenta. 2014;35(8):552–62.
Dufour JH, Dziejman M, Liu MT, Leung JH, Lane TE, Luster AD. IFN-gamma-inducible protein 10 (IP-10; CXCL10)-deficient mice reveal a role for IP-10 in effector T cell generation and trafficking. J Immunol. 2002;168(7):3195–204.
Xiao J, Garcia-Lloret M, Winkler-Lowen B, Miller R, Simpson K, Guilbert LJ. ICAM-1-mediated adhesion of peripheral blood monocytes to the maternal surface of placental syncytiotrophoblasts: implications for placental villitis. Am J Pathol. 1997;150(5):1845–60.
Derricott H, Jones RL, Heazell AEP, Greenwood SL. Co-culture of placental explants with isolated CD4 and CD8 T cells: a functional model to define the consequences of placental inflammation. 2015.
Derricott H, Heazell AEP, Greenwood SL, Jones RL. A novel in vitro model of villitis of unknown etiology demonstrates altered placental hormone and cytokine profile. Am J Reprod Immunol. 2017;78(5).
Grigsby PL. Animal models to study placental development and function throughout Normal and dysfunctional human pregnancy. Semin Reprod Med. 2016;34(1):11–6.
Acknowledgments
The authors would like to acknowledge Wendy Nevala, MS, for her excellent training in the MxIF technique.
Funding
Financial support for this project was provided by Mayo Clinic Division of Anatomic Pathology (SEK) and NICHD K12 HD065987 (EALE).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Enninga, E.A.L., Leontovich, A.A., Fedyshyn, B. et al. Upregulation of HLA-Class I and II in Placentas Diagnosed with Villitis of Unknown Etiology. Reprod. Sci. 27, 1129–1138 (2020). https://doi.org/10.1007/s43032-019-00101-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-019-00101-9