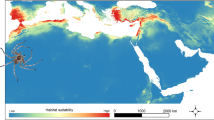
Abstract
Nelsonia is an extant genus of North American woodrats, in which most of the biological information remains unknown. Only N. neotomodon and N. goldmani are recognized inhabiting in similar temperate environments. Recently, a biogeographic hypothesis for the genus pointed out that Nelsonia after diverging from Repomys or Protorepomys in the Californian-Rocky Mountains in USA, the Mexican highlands in Mexico played a significant role in how the two species of Nelsonia became established in two different mountain ranges: N. neotomodon in the Sierra Madre Occidental and N. goldmani in the Transmexican Volcanic Belt. However, no other evidence exists about the biogeographic history of both species that improve the explanation about how past climate events affected their distribution, as well as whether the current environmental space occupied by both species are similar such as previous studies have argued. Therefore, this study aimed to characterize the environmental niche of both species, to test environmental differences, overlap, equivalence, and niche similarity, and lastly, to build their current niche model and paleodistribution for three past scenarios: Pliocene, Last Interglacial, and Last Glacial Maximum. The results revealed significant differences in niche centroid, low niche overlap, and no niche equivalence or similarity, suggesting niche divergence. The current niche suitability showed that the two species have small and restricted potential habitats highlighting the need to reevaluate the current international conservation category of N. neotomodon. The paleodistribution suggested that both species of Nelsonia have contractions and expansions of niche suitability in the past.





Similar content being viewed by others
Availability of data and material
All data generated and analyzed during this study are included in this published article, its supplementary material files, and are available from the corresponding author on reasonable request.
References
Aguirre-Gutiérrez J, Serna-Chavez HM, Villalobos-Arambula AR, Pérez de la Rosa JA, Raes N (2015) Similar but not equivalent: ecological niche comparison across closely–related Mexican white pines. Diversity Distrib 21(3):245–257. https://doi.org/10.1111/ddi.12268
Aiello-Lammens ME, Boria RA, Radosavljevic A, Vilela B, Anderson RP (2015) spThin: an R package for spatial thinning of species occurrence records for use in ecological niche models. Ecography 38(5):541–545. https://doi.org/10.1111/ecog.01132
Areias-Guerreiro J, Mira A, Barbosa AM (2016) How well can models predict changes in species distributions? A 13-year-old otter model revisited. Hystrix 27(1):1–5. https://doi.org/10.4404/hystrix-27.1-11867
Broennimann O, Fitzpatrick MC, Pearman PB, Petitpiette B, Pellissier L, Yoccoz NG, Thuiller W, Fortin MJ, Randin C, Zimmermann NE, Graham CH, Guisan A (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Global Ecol Biogeogr 21(4):481–497. https://doi.org/10.1111/j.1466-8238.2011.00698.x
Bryson RW, Murphy RW, Lathrop A, Lazcano-Villareal D (2011) Evolutionary drivers of phylogeographical diversity in the highlands of Mexico: a case study of the Crotalus triseriatus species group of montane rattlesnakes. J Biogeogr 38(4):697–710. https://doi.org/10.1111/j.1365-2699.2010.02431.x
Carleton MD (1980) Phylogenetic relationships in neotomine-peromyscine rodents (Muroidea) and a reappraisal of the dichotomy within New World Cricetinae. Occas Pap Mus Zool Univ Mich 157:11–46
Corrêa Nogueira TA, Ayala WE, Dayrell JS, Fraga R, Kaefer IL (2019) Scale-dependent estimates of niche overlap and environmental effects on two sister species of Neotropical snakes. Stud Neotrop Fauna E 54(2):121–132. https://doi.org/10.1080/01650521.2019.1616957
Di Cola V, Broennimann O, Petitpierre B, Breiner FT, D’Amen M, Randin C, Engler R, Pottier J, Pio D, Dubuis A, Pellissier L, Mateo RG, Hordijk W, Salamin N, Guisan A (2017) ecospat: an R package to support spatial analyses and modeling of species niches and distributions. Ecography 40(6):774–787. https://doi.org/10.1111/ecog.02671
Elith J, Kearney M, Phillips S (2010) The art of modelling range-shifting species. Methods Ecol Evol 1(4):330–342. https://doi.org/10.1111/j.2041-210X.2010.00036.x
Engstrom MD, Sánchez-Herrera O, Urbano-Vidales G (1992) Distribution, geographic variation, and systematic relationships within Nelsonia (Rodentia: Sigmodontinae). Proc Biol Soc Wash 105:867–881
García-Mendoza DF, López-González C (2005) The diminutive woodrat (Nelsonia neotomodon) in Chihuahua, México. Southwest Nat 50(4):503–506. https://doi.org/10.1894/0038-4909(2005)050[0503:DWNNIC]2.0.CO;2
González-Cózatl FX, Vallejo RM, Arellano E (2016) First record of Nelsonia goldmani in the state of Morelos, Mexico. Rev Mex Biodivers 87(2):545–547. https://doi.org/10.1016/j.rmb.2015.12.001
Guevara L, Morrone JJ, León-Paniagua L (2018) Spatial variability in species’ potential distributions during the last glacial maximum under different global circulation models: relevance in evolutionary biology. J Zool Syst Evol Res 57(1):113–126. https://doi.org/10.1111/jzs.12238
Guevara L, León-Paniagua L (2019) How to survive a glaciation: the challenge of estimating biologically realistic potential distributions under freezing conditions. Ecography 42(6):1237–1245. https://doi.org/10.1111/ecog.04202
Hijmans RJ, Phillips S, Leathwick J, Elith, J (2017) dismo: species distribution modeling. R package version 1.1–4. https://CRAN.R-project.org/package=dismo. Accessed 27 Mar 2020
Hooper ET (1954) A synopsis of the cricetine rodent genus Nelsonia. Occas Pap Mus Zool Univ Mich 558:1–12
IUCN (2012) IUCN Red list categories and criteria: Version 3.1. Gland, Switzerland and Cambridge, UK.
Karger DN, Conrad O, Böhner J, Kawohl T, Kreft H, Soria-Auza RW, Zimmermann NE, Linder HP, Kessler M (2017) Climatologies at high resolution for the earth’s land surface areas. Sci Data 4:170122. https://doi.org/10.1038/sdata.2017.122
Kozak KH, Wiens JJ (2006) Does niche conservatism promote speciation? A case study in North American salamanders. Evolution 60(12):2604–2621. https://doi.org/10.1554/06-334.1
León-Tapia MA, Cervantes FA (2019a) Noteworthy records and ecological niche modeling of the rare and endangered Goldman´s diminutive woodrat Nelsonia goldmani (Rodentia: Cricetidae) endemic to central Mexican highlands. Mammalia 83(4):330–342. https://doi.org/10.1515/mammalia-2018-0023
León-Tapia MA, Cervantes FA (2019b) First contribution to the description of reproductive structures of Nelsonia goldmani (Rodentia: Cricetidae). Therya 10:155–160. https://doi.org/10.12933/therya-19-747
León-Tapia MA, Cervantes FA (2021) Systematics and the unexpected high mitochondrial genetic divergence of Nelsonia goldmani (Rodentia: Cricetidae) from Mexican highlands. J Mamm Evol. https://doi.org/10.1007/s10914-020-09532-7
Martin RA, Zakrzewski RJ (2019) On the ancestry of woodrats. J Mammal 100(5):1564–1582. https://doi.org/10.1093/jmammal/gyz105
Mastretta-Yanes A, Moreno-Letelier A, Piñero D, Jorgensen TH, Emerson BC (2015) Biodiversity in the Mexican highlands and the interaction of geology, geography, and climate within the Trans-Mexican Volcanic Belt. J Biogeogr 42(9):1586–1600. https://doi.org/10.1111/jbi.12546
McCormack JE, Peterson AT, Bonaccorso E, Smith TB (2008) Speciation in the highlands of Mexico: genetic and phenotypic divergence in the Mexican jay (Aphelocoma ultramarina). Mol Ecol 17(10):2505–2521. https://doi.org/10.1111/j.1365-294x.2008.03776.x
Muscarella R, Galante PJ, Soley-Guardia M, Boria RA, Kass JM, Uriarte M, Anderson RP (2014) ENMeval: an R package for conducting spatially independent evaluations and estimating optimal model complexity for Maxent ecological niche models. Methods Ecol Evol 5(11):1198–1205. https://doi.org/10.1111/2041-210X.12261
Nelsonia neotomodon Merriam (1897) in GBIF Secretariat 2019. GBIF Backbone Taxonomy. https://doi.org/https://doi.org/10.15468/39omei. Accessed 15 Mar 2020
Osorio-Olvera L, Lira-Noriega A, Soberón J, Peterson AT, Falconi M, Contreras-Díaz RG, Martínez-Meyer E, Barve V, Barve N (2020) NTBOX: an r package with graphical user interface for modelling and evaluating multidimensional ecological niches. Methods Ecol Evol 11(10):1199–1206. https://doi.org/10.1111/2041-210X.13452
Otto-Bliesner BL, Marshall SJ, Overpeck JT, Miller GH, Hu A, CAPE Last Interglacial Project members (2006) Simulating arctic climate warmth and icefield retreat in the last interglaciation. Science 311(5768):1751–1753. https://doi.org/10.1126/science.1120808
Phillips SJ, Anderson RP, Dudík M, Schapire RE, Blair ME (2017) Opening the black box: an open-source release of Maxent. Ecography 40(7):887–893. https://doi.org/10.1111/ecog.03049
R Development Core Team (2017) R: a language and environment for statistical computing. R foundation for statistical computing. Vienna, Austria. https://www.r-project.org/. Accessed 03 May 2019
Schoener TW (1968) The Anolis lizards of Bimini: resource partitioning in a complex fauna. Ecology 49(4):704–726. https://doi.org/10.2307/1935534
Urbina-Cardona JN, Flores-Villela O (2010) Ecological-niche modeling and prioritization of conservation-area networks for Mexican herpetofauna. Conserv Biol 24(4):1031–1041. https://doi.org/10.1111/j.1523-1739.2009.01432.x
Varela S, Lima-Ribeiro MS, Terribile LC (2015) A short guide to the climatic variables of the last glacial maximum for biogeographers. PLoS ONE 10(6):e0129037. https://doi.org/10.1371/journal.pone.0129037
Warren DL, Glor RE, Turelli M (2008) Environmental niche equivalency versus conservatism: quantitative approaches to niche evolution. Evolution 62(11):2868–2883. https://doi.org/10.1111/j.1558-5646.2008.00482.x
Yousefkhani SSH, Rastegar-Pouyani E, Aliabadian M (2016) Ecological niche differentiation and taxonomic distinction between Eremias strauchi strauchi and Eremias strauchi kopetdaghica (Squamata: Lacertidae) on the Iranian Plateau based on ecological niche modeling. Ital J Zool 83(3):408–416. https://doi.org/10.1080/11250003.2016.1209581
Acknowledgements
I thank the anonymous reviewers who improved the manuscript with their suggestions and valuable comments.
Funding
This work was not funded.
Author information
Authors and Affiliations
Contributions
The author contributed to the conceptualization, design, analyses, and writing of this study.
Corresponding author
Ethics declarations
Conflicts of interest/competing interests
The author declares that he has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling editor: Sabine Begall.
Supplementary Information
Below is the link to the electronic supplementary material.
42991_2021_130_MOESM1_ESM.xlsx
Supplementary material 1: geographic information from published and museum specimens of Nelsonia neotomodon and N. goldmani used as occurrences for the environmental analyses. The museum acronyms are: American Museum of Natural History (AMNH), California Academy of Sciences (CAS), Colección de Mamíferos del Centro de Investigación en Biodiversidad y Conservación (CMC), Canadian Museum of Nature (CMN), Colección Nacional de Mamíferos (CNMA), Colección de Mamíferos del Centro Interdisciplinario de Investigación para el Desarrollo Integral Regional, Unidad Durango (CRD), Escuela Nacional de Ciencias Biológicas IPN (ENCB), Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias (INIFAP), Museum of Natural History University of Kansas (KU), Natural History Museum of Los Angeles County (LACM), Lousiana State University Museum of Zoology (LSUMZ), Michigan State University Museum (MSU), Museum of Vertebrate Zoology (MVZ), University of Arizona Museum of Natural History (UAZ), Florida Museum of Natural History (UFMNH), University of Michigan Museum of Zoology (UMMZ), and National Museum of Natural History (USNM).
42991_2021_130_MOESM2_ESM.tif
Supplementary information 2: multivariate environmental similarity surface (MESS; left) and most dissimilar variable (MoD; right) of three past scenarios projected. From top to bottom: Last Glacial Maximum (A and B), Last Interglacial (C and D), and Pliocene (E and F). Maps show the analog and non-analog areas, and the environmental conditions outside the rage in the calibration variables.
Rights and permissions
About this article
Cite this article
León-Tapia, M.Á. Environmental niche differentiation and paleodistribution of the rare montane woodrats of the genus Nelsonia (Rodentia: Cricetidae). Mamm Biol 101, 521–530 (2021). https://doi.org/10.1007/s42991-021-00130-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-021-00130-5