Abstract
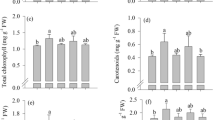
This study aimed to investigate the physiological roles of selenium (Se) in the photosynthetic pigment, antioxidant metabolism, flavonoid biosynthesis, nodulation, and its relationship with soybean production. The treatments consisted of five doses of Se (0 μg kg−1, 7.5 μg kg−1, 15 μg kg−1, 30 μg kg−1, and 45 μg kg−1) applied via soil and in the form of sodium selenate at the V2 phenological stage of soybean. The concentration of chlorophyll b and carotenoids, the activity of antioxidant enzymes (superoxide dismutase, catalase, ascorbate peroxidase), and the concentration of total sugars in soybean leaves increased in response to Se fertilization. In addition, Se increased ureide concentration in leaves and activity of nitrate reductase, resulting in greater bioavailability of amino acids and proteins in the leaves, generating greater stimulation of plant growth and root dry weight, but did not increase yield. The number of nodules and the synthesis of rutin and genistein in the roots were significantly increased by the Se application. Se also stimulated the translocation of amino acids from leaves to the nodules and of ureides from nodules to leaves, increasing efficiency in biological nitrogen fixation. This study presents fundamental new insights regarding Se effect on nitrogen metabolism, flavonoid synthesis, antioxidant metabolism, and nodulation of soybean that can be helpful in increasing the crop tolerance to abiotic stress.







Similar content being viewed by others
Data Availability
The datasets are available from the corresponding author upon request.
References
Agathokleous E (2021) The rise and fall of photosynthesis: hormetic dose response in plants. J For Res 32:889–898. https://doi.org/10.1007/s11676-020-01252-1
Agathokleous E, Feng Z, Peñuelas J (2020) Chlorophyll hormesis: are chlorophylls major components of stress biology in higher plants? Sci Total Environ 726:138637. https://doi.org/10.1016/j.scitotenv.2020.138637
Alexieva V, Sergiev I, Mapelli S, Karanov E (2001) The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ 24:1337–1344. https://doi.org/10.1046/j.1365-3040.2001.00778.x
Azevedo RAD, Alas RM, Smith RJ, Lea PJ (1998) Response of antioxidant enzymes to transfer from elevated carbon dioxide to air and ozone fumigation, in the leaves and roots of wild-type and a catalase-deficient mutant of barley. Physiol Plant 104:280–292. https://doi.org/10.1034/j.1399-3054.1998.1040217.x
Baral B, da Silva JAT, Izaguirre-Mayoral ML (2016) Early signaling, synthesis, transport and metabolism of ureides. J Plant Physiol 193:97–109. https://doi.org/10.1016/j.jplph.2016.01.013
Bosse MA, da Silva MB, de Oliveira NGRM, de Araujo MA, Rodrigues C, de Azevedo JP, Dos Reis AR (2021) Physiological impact of flavonoids on nodulation and ureide metabolism in legume plants. Plant Physiol Biochem 166:512–521. https://doi.org/10.1016/j.plaphy.2021.06.007
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cataldo DA, Maroon M, Schrader LE, Youngs VL (1975) Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun Soil Sci Plant Anal 6:71–80. https://doi.org/10.1080/00103627509366547
Cunha MLO, de Oliveira LCA, Silva VM, Montanha GS, dos Reis AR (2022) Selenium increases photosynthetic capacity, daidzein biosynthesis, nodulation and yield of peanuts plants (Arachis hypogaea L.). Plant Physiol Biochem 190:231–239. https://doi.org/10.1016/j.plaphy.2022.08.006
De Brito Mateus MP, Tavanti RFR, Galindo FS, da Rocha Silva AC, Gouveia GCC, Aparecido CFF, dos Reis AR (2020) Coffea arabica seedlings genotypes are tolerant to high induced selenium stress: evidence from physiological plant responses and antioxidative performance. Ecotoxicol Environ Saf 203:111016. https://doi.org/10.1016/j.ecoenv.2020.111016
Dos Santos SG, da Silva RF, Da Fonseca CS, Pereira W, Santos LA, Reis VM (2017) Development and nitrate reductase activity of sugarcane inoculated with five diazotrophic strains. Arch Microbiol 199:863–873. https://doi.org/10.1007/s00203-017-1357-2
Dubois M, Gilles KA, Hamilton JK, Rebers PT, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Ferrari S, Marsala L, Luis Oliveira Cunha M (2021b) Effects of low doses of glyphosate on agronomic traits of upland rice. Gesun Pflan 73:533–539. https://doi.org/10.1007/s10343-021-00573-3
Ferrari S, Marsala L, Oliveira Cunha ML, dos Santos Cordeiro LF, Tropaldi L, de Mattos Barretto VC, Alves de Oliveira LC (2021a) Can the application of low doses of glyphosate induce the hormesis effect in upland rice? J Environ Sci Health B 56:814–820. https://doi.org/10.1080/03601234.2021.1957372
Ferrari S, do Valle Polycarpo G, Vargas PF, Fernandes AM, Luís Oliveira Cunha M, Pagliari P (2022) Mix of trinexapac-ethyl and nitrogen application to reduce upland rice plant height and increase yield. Plant Growth Regul 96:209–219. https://doi.org/10.1007/s10725-021-00770-0
Fordyce FM (2013) Selenium deficiency and toxicity in the environment. In: Essentials of medical geology. Springer, Dordrecht, pp 375–416. https://doi.org/10.1007/978-94-007-4375-5_16
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gouveia GCC, Galindo FS, Lanza MGDB, da Rocha Silva AC, de Brito Mateus MP, da Silva MS, Dos Reis AR (2020) Selenium toxicity stress-induced phenotypical, biochemical and physiological responses in rice plants: characterization of symptoms and plant metabolic adjustment. Ecotoxicol Environ Saf 202:110916. https://doi.org/10.1016/j.ecoenv.2020.110916
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Ikram M, Raja NI, Javed B, Hussain M, Hussain M, Ehsan M, Akram A (2020) Foliar applications of bio-fabricated selenium nanoparticles to improve the growth of wheat plants under drought stress. Green Process Synth 9:706–714. https://doi.org/10.1515/gps-2020-0067
Karwat H, Sparke MA, Rasche F, Arango J, Nuñez J, Rao I, Cadisch G (2019) Nitrate reductase activity in leaves as a plant physiological indicator of in vivo biological nitrification inhibition by Brachiaria humidicola. Plant Physiol Biochem 137:113–120. https://doi.org/10.1016/j.plaphy.2019.02.002
Kaur S, Nayyar H (2015) Selenium fertilization to salt-stressed mungbean (Vigna radiate L. Wilczek) plants reduces sodium uptake, improves reproductive function, pod set and seed yield. Sci Hortic-Amsterdam 197:304–317. https://doi.org/10.1016/j.scienta.2015.09.048
Khalofah A, Migdadi H, El-Harty E (2021) Antioxidant enzymatic activities and growth response of quinoa (Chenopodium quinoa Willd) to exogenous selenium application. Plants 10:719. https://doi.org/10.3390/plants10040719
Kitajim K, Hogan KP (2003) Increases of chlorophyll a/b ratios during acclimation of tropical woody seedlings to nitrogen limitation and high light. Plant Cell Environ 26:857–865. https://doi.org/10.1046/j.1365-3040.2003.01017.x
Klimov VV (2003) Discovery of pheophytin function in the photosynthetic energy conversion as the primary electron acceptor of photosystem II. Photosynth Res 76:247–253. https://doi.org/10.1023/A:1024990408747
Lanza MGDB, dos Reis AR (2021) Roles of selenium in mineral plant nutrition: ROS scavenging responses against abiotic stresses. Plant Physiol Biochem 164:27–43. https://doi.org/10.1016/j.plaphy.2021.04.026
Lanza MGDB, Silva VM, Montanha GS, Lavres J, de Carvalho HWP, Dos Reis AR (2021) Assessment of selenium spatial distribution using μ-XFR in cowpea (Vigna unguiculata (L.) Walp.) plants: integration of physiological and biochemical responses. Ecotoxicol Environ Saf 207:111216. https://doi.org/10.1016/j.ecoenv.2020.111216
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Marchica A, Ascrizzi R, Flamini G, Cotrozzi L, Tonelli M, Lorenzini G, Pellegrini E (2021) Ozone as eustress for enhancing secondary metabolites and bioactive properties in Salvia officinalis. Ind Crop Prod 170:113730. https://doi.org/10.1016/j.indcrop.2021.113730
Marsala L, Oliveira Cunha ML, do Nascimento V, Pereira Prado E, da Silva Viana R, Ferrari S (2022) Can 2,4-D promote the hormesis effect in upland rice? J Environ Sci Health Part B 57:680–685. https://doi.org/10.1080/03601234.2022.2099687
McCullough H (1967) The determination of ammonia in whole blood by a direct colorimetric method. Clin Chim Acta 2:297–304. https://doi.org/10.1016/0009-8981(67)90133-7
Moldes CA, Medici LO, Abrahao OS, Tsai SM, Azevedo RA (2008) Biochemical responses of glyphosate resistant and susceptible soybean plants exposed to glyphosate. Acta Physiol Plant 30:469–479. https://doi.org/10.1007/s11738-008-0144-8
Raso MJ, Muñoz A, Pineda M, Piedras P (2007) Biochemical characterisation of an allantoate-degrading enzyme from French bean (Phaseolus vulgaris): the requirement of phenylhydrazine. Planta 226:1333–1342. https://doi.org/10.1007/s00425-007-0570-7
Reis AR, Favarin JL, Gallo LA, Malavolta E, Moraes MF, Lavres Junior J (2009) Nitrate reductase and glutamine synthetase activity in coffee leaves during fruit development. Revista Brasileira de Ciência do Solo 33:315–324
Reis HPG, de Queiroz Barcelos JP, Junior EF, Santos EF, Silva VM, Moraes MF, dos Reis AR (2018) Agronomic biofortification of upland rice with selenium and nitrogen and its relation to grain quality. J Cereal Sci 79:508–515. https://doi.org/10.1016/j.jcs.2018.01.004
Ríos JJ, Blasco B, Cervilla LM, Rubio-Wilhelmi MM, Rosales MA, Sanchez-Rodriguez E, Ruiz JM (2010) Nitrogen-use efficiency in relation to different forms and application rates of Se in lettuce plants. J Plant Growth Regul 29:164–170. https://doi.org/10.1007/s00344-009-9130-7
Rios JJ, Blasco B, Rosales MA, Sanchez-Rodriguez E, Leyva R, Cervilla LM, Ruiz JM (2010) Response of nitrogen metabolism in lettuce plants subjected to different doses and forms of selenium. J Sci Food Agric 90:1914–1919. https://doi.org/10.1002/jsfa.4032
Sachdev S, Ansari SA, Ansari MI, Fujita M, Hasanuzzaman M (2021) Abiotic stress and reactive oxygen species: generation, signaling, and defense mechanisms. Antioxidants 10:277. https://doi.org/10.3390/antiox10020277
Silva VM, Boleta EHM, Lanza MGDB, Lavres J, Martins JT, Santos EF, dos Reis AR (2018) Physiological, biochemical, and ultrastructural characterization of selenium toxicity in cowpea plants. Environ Exp Bot 150:172–182. https://doi.org/10.1016/j.envexpbot.2018.03.020
Silva VM, Boleta EH, Martins JT, Dos Santos FL, da Rocha Silva AC, Alcock TD, Dos Reis AR (2019) Agronomic biofortification of cowpea with selenium: effects of selenate and selenite applications on selenium and phytate concentrations in seeds. J Sci Food Agric 99:5969–5983. https://doi.org/10.1002/jsfa.9872
Silva VM, Tavanti RFR, Gratão PL, Alcock TD, Dos Reis AR (2020) Selenate and selenite affect photosynthetic pigments and ROS scavenging through distinct mechanisms in cowpea (Vigna unguiculata (L.) Walp) plants. Ecotoxicol Environ Saf 201:110777. https://doi.org/10.1016/j.ecoenv.2020.110777
Stirbet A, Lazár D, Kromdijk J (2018) Chlorophyll a fluorescence induction: can just a one-second measurement be used to quantify abiotic stress responses? Photosynthetica 56:86–104. https://doi.org/10.1007/s11099-018-0770-3
Tibiletti T, Hernández-Prieto MA, Matthijs HC, Niyogi KK, Funk C (2016) Deletion of the gene family of small chlorophyll-binding proteins (ScpABCDE) offsets C/N homeostasis in Synechocystis PCC 6803. Biochim Biophys Acta 1857:396–407. https://doi.org/10.1016/j.bbabio.2015.11.011
Vogels GD, Van Der Drift C (1970) Differential analyses of glyoxylate derivatives. Anal Biochem 33:143–157. https://doi.org/10.1016/0003-2697(70)90448-3
Yao X, Jianzhou C, Xueli H, Binbin L, Jingmin L, Zhaowei Y (2013) Effects of selenium on agronomical characters of winter wheat exposed to enhanced ultraviolet-B. Ecotox Environ Safe 92:320–326. https://doi.org/10.1016/j.ecoenv.2013.03.024
Yemm EW, Cocking EC, Ricketts RE (1955) The determination of amino-acids with ninhydrin. Analyst 80:209–214. https://doi.org/10.1039/AN9558000209
Funding
This research was supported by FAPESP (2020/10053-8).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1:
Figures S1-S4 (DOCX 801 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cunha, M.L.O., de Oliveira, L.C.A., Mendes, N.A.C. et al. Selenium Increases Photosynthetic Pigments, Flavonoid Biosynthesis, Nodulation, and Growth of Soybean Plants (Glycine max L.). J Soil Sci Plant Nutr 23, 1397–1407 (2023). https://doi.org/10.1007/s42729-023-01131-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-023-01131-8