Abstract
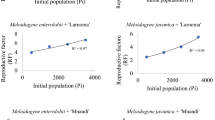
The mungbean, Vigna radiata (L.) R. Wilczek, is one of the most significant grain legume crops grown in east, southeast, and south Asia. Several biotic and abiotic factors affect the growth of a plant. It is susceptible to various diseases and pests, including plant parasitic nematodes and mungbean yellow mosaic virus. Crops suffer severe yield losses due to these. We investigated the effects of the biotic stresses due to the root parasitic Meloidogyne incognita and the Mungbean Yellow Mosaic Virus (MYMV), alone and in combined infections, on the growth parameters of mungbean plant. The root-knot nematode reduced plant growth, the decrease being more as the nematode inoculum level was increased from 1 to 4 J2 cm− 3 soil. The reduction was recorded in terms of shoot length upto 35.7%, root length (34.8%), shoot fresh weight (53.7%), dry weight (49.3%) and leaf surface area by 24.5% at the inoculation level of 4 J2 cm− 3 soil. The MYMV had relatively lesser effect on the and size and biomass of plant, compared to the nematode. It reduced shoot length by 10.3%, root-length (21.7%), shoot fresh weight (14.7%), dry weight (25.1%) at 4 J2 cm− 3 soil. The chlorophyll content was reduced more due to the virus (Chl.-a 49.6, Chl. -b 52.7%) while due to nematode at 4 J2 cm− 3 soil the reduction was Chl.-a 36.5 and Chl.-b 41.0%). Their combined infection caused upto 78.1% reduction in Chlorophyll content. The damage due to the virus was significantly more if plant root was also already infected with the root-knot nematode. With an increase in nematode inoculum level and in the presence of Mungbean yellow mosaic virus disease, the rate of reproduction of nematode was also reduced due to competition and diminished food resources due to viral infection on the foliage. There was a significant positive interaction effect between nematode infection and intensity of MYMV on foliage, rendering the plant more prone to viral infection. The yellow mosaic index increased from 2.8 in control to 3.7, 4.2 and 4.6 at 1, 2 and 4 J per cm3 soil, respectively. Thus, control of nematodes can not only reduce loss due to nematode but also partly offset the crop loss due to the Mungbean Yellow Mosaic Virus.



Similar content being viewed by others
References
Akhtar KP, Kitsanachandee R, Srinives P, Abbas G, Asghar MJ, Shah TM, Sarwar N (2009) Field evaluation of mungbean recombinant inbred lines against mungbean yellow mosaic Disease using new Disease scale in Thailand. Plant Pathol J 25(4):422–428
Alam AM, Somta P, Srinives P (2014) Generation mean and path analyses of reaction to mungbean yellow mosaic virus (MYMV) and yield-related traits in mungbean (Vigna radiata (L.) Wilczek). SABRAO J Breed Genet 46:150–159
Ali MA, Trabulsi IY, Abd Elsamea ME (1981) Antagonistic interaction between Meloidogyne incognita and Rhizobium leguminosarum on cowpea. Plant Dis 65:432–435
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Azhagumurugan C, Rajan MK, Pavaraj M (2014) Effect of root knot nematode, Meloidogyne incognita on the growth characteristics of Horse Gram, Macrotylona uniflorum treated with fruit extracts of Aegle marmelos. World J Zool 9(3):162–165
Binyamin R, Ahmed N, Ashraf W, Li Y, Ghani MU, Zeshan MA, Ali S, Khan AUR, Ahmed R, Ahmed MAA, Aljowaie RM, Alkahtani AM, Vachova P (2022) Prediction of mungbean yellow mosaic virus Disease using multiple regression models. J King Saud Univ – Sci 34:102094. https://doi.org/10.1016/j.jksus.2022.102094
Biswas KK, Malathi VG, &Varma A (2008) Diagnosis of symptomless yellow mosaic begomovirus Infection in pigeonpea by using cloned Mungbean yellow mosaic India virus as probe. J Plant Biochem Biotechnol 17(1):9–14
Byrd DW, Kirkpatrick Jr, Backer KR (1983) An improved technique for cleaning and staining plant tissues for detection of nematodes. J Nematology 15:142–143
Dasgupta DR, Gaur HS (1986) The root-knot nematodes Meloidogyne spp. in India. In: Swarup G, Dasgupta DR (eds) Plant Parasitic nematodes of India: problems and progress. IARI, New Delhi, India, pp 139–171
Davis EL, Mitchum MG (2005) Nematodes. Sophisticated parasites of legumes. Plant Physiol 137(4):1182–1188. https://doi.org/10.1104/pp.104.054973
Eisenback JD, Hirschmann H, Triantaphyllou AC (1980) Morphological comparison of Meloidogyne female head structures, perineal patterns, and stylets. J Nematology 12(4):300–313
Esbenshade PR, Triantaphyllou AC (1985) Use of enzyme phenotypes for identification of Meloidogyne species. J Nematology 17(1):6–20
Gaur HS, Mende N, Von, Perry RN (1996) Differentiation of two groups of species of the genus Meloidogyne by polymerase chain reaction and restriction fragment length polymorphisms of ribosomal DNA. Afro-Asian J Nematology 6:50–54
Goswami BK, Singh S, Verma VS (1974) Interaction of a mosaic virus with root-knot nematode Meloidogyne incognita in Vigna sinensis. Nematologica 20(3):366–367
Hartman KM, Sasser JN (1985) Identification of Meloidogyne species on the basis of differential host test and perineal pattern morphology. In An Advanced Treatise on Meloidogyne, Vol. 2: 69–77
Hisamuddin SS, Azam T (2010) Pathogenicity of root-knot nematode, Meloidogyne incognita on Medicago truncatula (Medik). Archives of Phytopathology and Plant Protection 43:1504–1511. https://doi.org/10.1080/03235400802583537
Husain SI, Khan TA, Jabri MRA (1985) Studies on root-knot, root-rot pea mosaic virus complex of Pisum sativum. Nematologia Mediterranea 13:103–109
Hussain MA, Mukhtar T, Kayani MZ (2011) Assessment of the damage cause by Meloidogyne incognita on okra (Abelmoschus esculentus). J Anim Plant Sci 21:857–861
Jayappa HK, Ramappa, Devamani BD (2017) Management of Mungbean Yellow Mosaic Virus (MYMV) in Mungbean (Vigna radiata L.) Journal of Entomology and Zoology Studies 2017; 5(5): 596–601
Joshi I, Kumar A, Kohli D, Singh AK, Sirohi A, Subramaniam K, Jain PK (2020) Conferring root-knot nematode resistance via host-delivered RNAi-mediated silencing of four Mi-msp genes in Arabidopsis. Plant Sci 298:110592. https://doi.org/10.1016/j.plantsci.2020.110592
Kayani MZ, Mukhtar T, Hussain MA (2017) Effects of southern root knot nematode population densities and plant age on growth and yield parameters of cucumber. Crop Prot 92:207–212. https://doi.org/10.1016/j.cropro.2016.09.007
Kirkpatrick TL, Oosterhuis DM, Wullschleger SD (1991) Interaction of Meloidogyne incognita and water stress in two cotton cultivars. J Nematology 23(4):262–267
Kumar D, Bhatt J, Sharma RL (2018) Effect of different inoculum levels, plant age and multiplication of Meloidogyne incognita on growth of blackgram (Vigna mango L). J Entomol Zool Stud 6:452–455
Malathi VC, John P (2009) Mungbean yellow mosaic viruses. In: Van Regenmortal M, Mahy B (eds) Desk encyclopedia of plant and fungal virology. Academic press, London, pp 217–226
McLaughlin MR, Windham GL (1996) Effects of Peanut Stunt Virus, Meloidogyne incognita, and drought on growth and persistence of white clover. Phytopathology 86:1105–1111
Nariani TK (1960) Yellow mosaic of mung (Phaseolus aureus L). Indian Phytopathol 13:24–29
Ohki ST, Leps WT, Hiruki C (1986) Effects of alfalfa mosaic virus Infection on factors associated with symbiotic N2 fixation in alfalfa. Can J Plant Pathol 8(3):277–281
Olmo R, Cabrera J, Diaz-Manzano FE, Ruiz-Ferrer V, Barcala M, Ishida T, Garcia A, Andres MF, Ruiz-Lara S, Verdugo I, Pernas M, Fukaki H, Pozo JCD, Moreno-Risueno MA, Kyndit T, Gheysen G, Fenoll C, Sawa S, Escobar C (2020) Root-knot nematodes induce gall formation by recruiting developmental pathways to post-embryonic organogenesis and regeneration to promote transient pluripotency. New Phytol 227(1):200–215. https://doi.org/10.1111/nph.16521
Patel KA, Patel BN (1995) Effect of Tobacco mosaic virus and root-knot nematode Infection on chemical constituents of bidi Tobacco. Indian J Mycol Plant Pathol 25(3):228–230
Pathak AK, Jhamaria SL (2004) Evaluation of mungbean (Vigna radiata L.) varieties to yellow mosaic virus. J Mycol Plant Pathol 34(1):64–65
Patil BL, Fauquet CM (2021) Ecology of Plant Infecting Viruses, With Special Reference To Geminiviruses. Studies in Viral Ecology, 183–229
Patil J, Miller AJ, Gaur HS (2013) Effect of nitrogen supply form on the invasion of rice roots by the root-knot nematode, Meloidogyne Graminicola. Nematology 15(4):483–492
Pazarlar S, Gumus M, Oztekin GB (2013) The effects of Tobacco mosaic virus Infection on growth and physiological parameters in some pepper varieties (Capsicum annuum L). Notulae Botanicae Horti Agrobotanici Cluj-Napoca 41(2):427–433. https://doi.org/10.15835/nbha4129008
Pratap A, Douglas C, Prajapati U, Kumari G, War AR, Tomar R, Pandey AK, Dubey S (2020) Breeding progress and future challenges: biotic stresses. In The mungbean genome Springer, Cham, pp. 55–80. https://doi.org/10.1007/978-3-030-20008-4_5
Salam SA, Patil MS, Dyadgi AS (2011) Status of mungbean yellow mosaic virus Disease incidence on green gram. Karnataka J Agricultural Sci 24(2):247–248
Sasanelli N, D’addabbo T, Lišková M (2006) Influence of the root-knot nematode Meloidogyne incognita r. 1 on growth of grapevine. Helminthologia 43(3):168–170
Sasser JN (1980) Root-knot nematodes: A Global Menace to Crop Production. Plant Dis 64(1):36–41
Schindler A (1961) A simple substitute for a Baermann funnel. Plant Disease Reporter 45(9):747–749
Schomaker CH, Been TH (2006) Plant growth and population dynamics. Plant Nematology, pp 275–301
Seinhorst JW (1966) The relationships between Population increase and Population Density in Plant Parasitic nematodes. Nematologica 12(1):157–169
Selvi R, Muthiah AR, Manivannan N, Raveendran TS, Manickam A, Samiyappan R (2006) Tagging of RAPD marker for MYMV resistance in mungbean (Vigna radiata (L.) Wilczek). Asian J Plant Sci 5:277–280
Sharma SB, Sharma HK, Pankaj (2000) Nematode problems in India. In: Crop Pest Management – Challenges for the Millennium. Eds. D Prasad & S.N Puri. Jyoti Publ., New Delhi: 267–275 pp
Siengchi K, Ruanpanun P, Somta P (2020) Root-knot nematode (Meloidogyne Incognita Chitwood) population density on plant growth parameters related to plant age og mung bean (Vigna radiata (L. Wilczek J ISSAAS 26(1):111–122
Sikora RA, Coyne D, Hallmann J, Timper P (eds) (2018) Plant Parasitic nematodes in Subtropical and Topical Agriculture. CABI, UK, p 492
Sikora RA, Desaeger J, Molendijk L (eds) (2022) Integrated nematode management: state-of-the-art and visions for the future. CABI, UK, p 498
Singh DB, Reddy PP (1981) Influence of M. incognita infestation on rhizobium nodule formation in French bean. Nematologia Mediterranea 9:1–5
Singh JS, Raghubanshi AS, Singh RS, Srivastava SC (1989) Microbial biomass acts as a source of plant nutrients in dry tropical forest and savanna. Nature 338(6215):499–500
Soriano I, Reversat G (2003) Management of Meloidogyne graminicola and yield of upland rice in South-Luzon, Philippines. Nematology 5(6):879–884
Tabreiz AK, Iram TA, Shweta S (2012) Comparative studies on the pathogenic potential of Meloidogyne spp. on mung bean (Vigna radiata L). Afr J Microbiol Res 6:7134–7138. https://doi.org/10.5897/AJMR11.1053
Taylor AL, Sasser JN (1978) Biology, identification and control of root-knot nematodes. North Carolina State University Graphics, USA
Thongmeearkom P, Kittipakorn K, Surin P (1981) Outbreak of mungbean yellow mosaic Disease in Thailand. Thai J Agricultural Sci 14(2):201–206
Varshney S, Pandey RC, Panday RK, Dwivedi BK, Singh V (2005) Interaction between mungbean, yellow mosaic virus and root knot nematode on growth of mungbean plants. Pakistan J Nematology 23(1):93–98
Velie EM, Schairer C, Flood A, He JP, Khattree R, Schatzkin A (2005) Empirically derived dietary patterns and risk of postmenopausal Breast cancer in a large prospective cohort study. Am J Clin Nutr 82(6):1308–1319
Waghmare C, Singh P, Paul S, Sharma HK (2022) Influence of root-knot nematode, Meloidogyne incognita (Kofoid & White) Chitwood Infection on different plant growth parameters in Mungbean, Vigna radiata (L.) Wilczek. Indian J Exp Biol 60(05):351–359
Walker GE, Wallace HR (1975) The influence of root-knot nematode (Meloidogyne Jav Anica) and Tobacco ringspot virus on the growth and mineral content of Frenchbeans (Phaseolus vulgaris). Nematologica 21(4):455–462
Williamson VM, Hussey RS (1996) Nematode pathogenesis and resistance in plants. Plant Cell 8(10):1735–1745
Zijlstra C (2000) Identification of Meloidogyne chitwoodi, M. fallax and M. hapla based on SCAR-PCR: a powerful way of enabling reliable identification of populations or individuals that share common traits. European Journal of Plant Pathology, 106(3): 283–290
Acknowledgements
The authors are grateful to Dr. Shalini Gaur Rudra of ICAR-IARI, New Delhi for her help in improvement of the manuscript, to Dr. Anil Sirohi of the Division of Nematology, ICAR-IARI, New Delhi for the starting culture of M. incognita, and to Division of Genetics, ICAR-IARI, New Delhi for seed of mungbean cv. Pusa Vishal. The authors are also grateful to the Sharda University, Greater Noida for the financial assistance for these investigations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Srivastava, P., Gaur, H.S. Damage potential of root-knot nematode and mungbean yellow mosaic virus alone and together on the growth characteristics of mungbean plant. Vegetos (2023). https://doi.org/10.1007/s42535-023-00758-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42535-023-00758-3