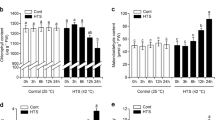
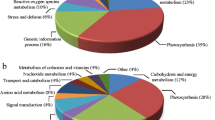
Abstract
Seabuckthorn (Hippophae), is a stress hardy Himalayan shrub adapted to extreme climatic conditions. Still, very little is known about their stress tolerance mechanism. Therefore, aim of the present investigation is to understand the diverse stress hardiness mechanism of Indian seabuckthorn, growing naturally in high-altitude ‘Trans-Himalayan’ (H. rhamnoides, H. tibetana from Lahaul-Spiti, Himachal Pradesh) and lower altitude adapted ‘Sikkim’ (H. salicifolia) populations. Proteome data set generated using the comparative shotgun (nano LC-MS/MS) proteomics approach is the first protein repository of naturally growing Indian seabuckthorn populations. Bioinformatics analysis (KEGG and GO) revealed higher number of proteins associated with cellular, metabolic and biological regulation of physiological processes in Sikkim populations. Conversely, the abundance of proteins associated with redox signaling and response to stimulus indicates better stress-adaptive responses in higher altitude adapted Trans-Himalayan populations to withstand harsh climatic conditions. Interestingly, a positive correlation between proteomic plasticity and corresponding morphological (retarded growth, waxy/silvery-greyish leaf) and biochemical signatures (higher flavonoid and PUFA content) also supported efficient stress adaptive responses in Trans-Himalayan populations to survive harsher alpine climatic conditions. On the contrary, in Sikkim populations ‘H. salicifolia’ proteomic and biochemical (higher phenolics) resilience reflected the underlying physiological adaptive mechanisms to survive sub-alpine climatic conditions at lower elevations. To sum up, these interesting findings showed a clear trade-off between growth and stress tolerance phenomenon in diverse seabuckthorn populations. Trans-Himalayan populations repress their growth and divert energy resources in the direction of better stress responses to survive extreme climatic conditions. In contrast, Sikkim populations at lower elevations may invest in resource allocation or growth-promoting pathways in response to milder stress conditions. To the best of our knowledge, this is the first comprehensive proteome analysis to examine stress acclimation strategies in different naturally growing Indian seabuckthorn populations. The dataset is submitted in PRIDE ‘PRoteomics IDEntifications database’ proteomics data repository with accession number “PXD023184”.






Similar content being viewed by others
References
Basha SM, Mazhar H (2009) Proteomics approach to identify unique xylem sap proteins in pierce’s disease-tolerant Vitis species. Appl Biochem Biotechnol 160:932–944. https://doi.org/10.1007/s12010-009-8620-1
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Bredow M, Walker VK (2017) Ice-binding proteins in plants. Front Plant Sci 8:1–15. https://doi.org/10.3389/fpls.2017.02153
Burcova Z, Kreps F, Schmidt S, Jablonsky M (2017) Composition of fatty acids and tocopherols in peels, seeds and leaves of sea buckthorn. Acta Chim Slovaca 10:29–34. https://doi.org/10.1515/acs-2017-0005
Chaudhary S, Sharma PC (2015) Deep SAGE based differential gene expression analysis under cold and freeze stress in seabuckthorn (Hippophae rhamnoides L.). PLoS ONE. https://doi.org/10.1371/journal.pone.0121982
Cheng Z, Dong K, Ge P et al (2015a) Identification of leaf proteins differentially accumulated between wheat cultivars distinct in their levels of drought tolerance. PLoS ONE. https://doi.org/10.1371/journal.pone.0125302
Cheng Q, Li N, Dong L et al (2015b) Overexpression of soybean isoflavone reductase (GmIFR) enhances resistance to phytophthora sojae in soybean. Front Plant Sci 6:1–11. https://doi.org/10.3389/fpls.2015.01024
Chi YH, Koo SS, Oh HT et al (2019) The physiological functions of universal stress proteins and their molecular mechanism to protect plants from environmental stresses. Front Plant Sci 10:1–13. https://doi.org/10.3389/fpls.2019.00750
Dolkar P, Dolkar D, Angmo S et al (2017) Sexual differences and seasonal variations in total phenolics and antioxidant properties in Hippophae rhamnoides leaves. J Berry Res 7:61–69. https://doi.org/10.3233/JBR-170147
Dolkar P, Dolkar D, Kant A, Chaurasia OP, Stobdan T (2019) Gender differences in phenotypic and adaptive response of Seabuckthorn (Hippophae rhamnoides L.) along an altitudinal gradient in Trans-Himalaya. J Berry Res 9:1–10. https://doi.org/10.3233/JBR-170294
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875. https://doi.org/10.1105/tpc.105.033589
Ghangal R, Chaudhary S, Jain M et al (2013) Optimization of de novo short read assembly of seabuckthorn (Hippophae rhamnoides L.) Transcriptome. PLoS ONE 8:1–7. https://doi.org/10.1371/journal.pone.0072516
Gupta R, Deswal R (2012) Low temperature stress modulated secretome analysis and purification of antifreeze protein from Hippophae rhamnoides, a Himalayan wonder plant. J Proteome Res 11:2684–2696. https://doi.org/10.1021/pr200944z
Gupta R, Deswal R (2014) Refolding of β-stranded class I chitinases of Hippophae rhamnoides enhances the antifreeze activity during cold acclimation. PLoS ONE 9:1–11. https://doi.org/10.1371/journal.pone.0091723
Hanin M, Brini F, Ebel C et al (2011) Versatile proteins for complex mechanisms plant dehydrins and stress tolerance. Plant Signal Behav. https://doi.org/10.4161/psb.6.10.17088
Hashim AM, Alharbi BM, Abdulmajeed AM et al (2020) Oxidative stress responses of some endemic plants to high altitudes by intensifying antioxidants and secondary metabolites content. Plants. https://doi.org/10.3390/plants9070869
He C, Gao G, Zhang J et al (2016) Proteome profiling reveals insights into cold-tolerant growth in sea buckthorn. Proteome Sci 14:1–9. https://doi.org/10.1186/s12953-016-0103-z
He M, Wang Y, Wu J et al (2019) Isolation and characterization of S -Adenosylmethionine synthase gene from cucumber and responsive to abiotic stress. Plant Physiol Biochem 141:431–445. https://doi.org/10.1016/j.plaphy.2019.06.006
Ho S, Tong W, Yu S (2000) Multiple mode regulation of a cysteine proteinase gene expression in rice 1. Plant Physiol 122:57–66
Hon WC, Griffith M, Chong P, Yang D (1994) Extraction and isolation of antifreeze proteins from winter rye (Secale cereale L.) leaves. Plant Physiol 104:971–980. https://doi.org/10.1104/pp.104.3.971
Hong Y, Ni S, Zhang G (2020) Transcriptome and metabolome analysis reveals regulatory networks and key genes controlling barley malting quality in responses to drought stress. Plant Physiol Biochem 152:1–11. https://doi.org/10.1016/j.plaphy.2020.04.029
Jiang T, Zhang X, Wang X, Zhang D (2011) Arabidopsis 3-Ketoacyl-CoA Thiolase-2 (KAT 2), an enzyme of fatty acid β-oxidation, is involved in ABA signal transduction. Plant Cell Physiol 52(3):528–538. https://doi.org/10.1093/pcp/pcr008
Kang CH, Lee YM, Park JH et al (2016) Ribosomal P3 protein AtP3B of Arabidopsis acts as both protein and RNA chaperone to increase tolerance of heat and cold stresses. Plant Cell Environ. https://doi.org/10.1111/pce.12742
Kato S, Tanno Y, Takaichi S, Shinomura T (2019) Low temperature stress alters the expression of phytoene desaturase genes (crtP1 and crtP2) and the z -carotene desaturase gene (crtQ) together with the cellular carotenoid content of Euglena gracilis. Plant Cell Physiol 60:274–284. https://doi.org/10.1093/pcp/pcy208
Korekar G, Dolkar P, Singh H et al (2014) Variability and the genotypic effect on antioxidant activity, total phenolics, carotenoids and ascorbic acid content in seventeen natural population of seabuckthorn from Trans-Himalaya. LWT - Food Sci Technol 55:157–162. https://doi.org/10.1016/j.lwt.2013.09.006
Kosová K, Vítámvás P, Prášil IT, Renaut J (2011) Plant proteome changes under abiotic stress - Contribution of proteomics studies to understanding plant stress response. J Proteomics 74:1301–1322. https://doi.org/10.1016/j.jprot.2011.02.006
Kosová K, Vítámvás P, Urban MO et al (2018) Plant Abiotic stress proteomics : the major factors determining alterations in cellular proteome. Front Plant Sci 9:1–22. https://doi.org/10.1186/s40659-019-0246-3
Laskowski MJ, Dreher KA, Gehring MA et al (2002) FQR1, a novel primary auxin-response gene, encodes a flavin mononucleotide-binding quinone reductase. Plant Physiol 128:578–590. https://doi.org/10.1104/pp.010581.Abel
Li C, Xu G, Zang R et al (2007) Sex-related differences in leaf morphological and physiological responses in Hippophae rhamnoides along an altitudinal gradient. Tree Physiol 27:399–406. https://doi.org/10.1093/treephys/27.3.399
Li X, Yang Y, Ma L et al (2014) Comparative proteomics analyses of Kobresia pygmaea adaptation to environment along an elevational gradient on the central tibetan plateau. PLoS ONE 9(6):e98410. https://doi.org/10.1371/journal.pone.0098410
Ma L, Sun X, Kong X et al (2015) Physiological, biochemical and proteomics analysis reveals the adaptation strategies of the alpine plant Potentilla saundersiana at altitude gradient of the North western Tibetan Plateau. J Proteomics 112:63–82. https://doi.org/10.1016/j.jprot.2014.08.009
Maheshwari DT, Yogendra Kumar MS, Verma SK, Singh VK, Singh SN (2011) Antioxidant and hepatoprotective activities of phenolic rich fraction of sea buckthorn (Hippophae rhamnoides L.) leaves. Food Chem Toxic 49:2422–2428. https://doi.org/10.1016/j.fct.2011.06.061
Osakabe Y, Yamaguchi-shinozaki K, Shinozaki K, Tran LP (2013) Sensing the environment: key roles of membrane-localized kinases in plant perception and response to abiotic stress. J Exp Bot 64:445–458. https://doi.org/10.1093/jxb/ers354
Raina SN, Jain S, Sehgal D et al (2012) Diversity and relationships of multipurpose seabuckthorn (Hippophae L.) germplasm from the Indian Himalayas as assessed by AFLP and SAMPL markers. Genet Resour Crop Evol 59:1033–1053. https://doi.org/10.1007/s10722-011-9742-1
Ruan C, Rumpunen K, Nybom H et al (2013) Advances in improvement of quality and resistance in a multipurpose crop: sea buckthorn advances in improvement of quality and resistance in a multipurpose crop: sea buckthorn. Crit Rev Biotechnol 33:126–144. https://doi.org/10.3109/07388551.2012.676024
Sasaki Y, Nagano Y (2004) Plant acetyl-CoA carboxylase: structure, biosynthesis, regulation, and gene manipulation for plant breeding. Biosci Biotechnol Biochem 68:1175–1184. https://doi.org/10.1271/bbb.68.1175
Schmitz J, Rossoni AW, Maurino VG (2018) Dissecting the physiological function of plant glyoxalase i and glyoxalase i-like proteins. Front Plant Sci 9:1–7. https://doi.org/10.3389/fpls.2018.01618
Sharma B, Arora S, Sahoo D, Deswal R (2019b) Comparative fatty acid profiling of Indian seabuckthorn showed altitudinal gradient dependent species-specific variations. Physiol Mol Biol Plants 26:1–9. https://doi.org/10.1007/s12298-019-00720-1
Sharma B, Deswal R (2020) Ecophysiolomic analysis of stress tolerant Himalayan shrub Hipppophae rhamnoides shows multifactorial acclimation strategies induced by diverse environmental conditions. Physiol Plant 68:58–76. https://doi.org/10.1111/ppl.12942
Sharma B, Gupta R, Deswal R (2016) Mining the protein repertoire of a Himalayan shrub, Hippophae rhamnoides for antifreeze proteins. J Proteins Proteomics 7:199–211
Sharma B, Gupta R, Sahoo D, Deswal R (2019a) Purification of dual-functioning chitinases with hydrolytic and antifreeze activities from Hippophae rhamnoides seedlings. J Proteins Proteomics 10:69–81. https://doi.org/10.1007/s42485-019-00007-9
Sharma B, Sahoo D, Deswal R (2018) Single-step purification and characterization of antifreeze proteins from leaf and berry of a freeze-tolerant shrub seabuckthorn (Hippophae rhamnoides). J Sep Sci 41:3938–3945. https://doi.org/10.1002/jssc.201800553
Shayan S, Norouzi M, Moghaddam Vahed M et al (2020) Leaf proteome pattern of two bread wheat varieties under water deficit stress conditions. Curr Plant Biol 23:100146. https://doi.org/10.1016/j.cpb.2020.100146
Singh V, Kallio H, Yang B (2003) Sea buckthorn (Hippophae L.): a multipurpose wonder plant in biochemistry and pharmacology. In: Singh V (ed), 2nd edn. Indus Publishing Company, India, pp 151–158
Singh TA, Sarangi PK, Singh NJ (2015) Sea buckthorn (Hippophae Sp.): highly potential underutilised plant in Sikkim: a review. Bioscan 10:1413–1417
Soto G, Stritzler M, Lisi C, Alleva K, Pagano ME, Ardila F et al (2011) Acetoacetyl-CoA thiolase regulates the mevalonate pathway during abiotic stress adaptation. J Exp Bot 62:5699–5711. https://doi.org/10.1093/jxb/err287
Stone SL (2014) The role of ubiquitin and the 26S proteasome in plant abiotic stress signaling. Front Plant Sci 5:135. https://doi.org/10.3389/fpls.2014.00135
Sunkar R, Bartels D, Kirch HH (2003) Overexpression of a stress-inducible aldehyde dehydrogenase gene from Arabidopsis thaliana in transgenic plants improves stress tolerance. Plant J 35:452–64
Suryakumar G, Gupta A (2011) Medicinal and therapeutic potential of Seabuckthorn (Hippophae rhamnoides L.). J Ethnopharmacol 138:268–278. https://doi.org/10.1016/j.jep.2011.09.024
Thomashow MF (1998) Role of cold-responsive genes in plant freezing tolerance. Plant Physiol 118:1–7. https://doi.org/10.1104/pp.118.1.1
Ugarte MR, Escudero A, Gavilán RG (2019) Metabolic and physiological responses of Mediterranean high-mountain and alpine plants to combined abiotic stresses. Physiol Plant 165:403–412. https://doi.org/10.1111/ppl.12898
Wang W, Vinocur B, Shoseyov O, Altman A (2004) Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 9:13–15. https://doi.org/10.1016/j.tplants.2004.03.006
Wang X, Yen J, Peter Kaiser LH (2011) Regulation of the 26S proteasome complex during oxidative stress. Sci Signal 3:1–17. https://doi.org/10.1126/scisignal.2001232.Regulation
Willick IR, Takahashi D, Fowler DB et al (2018) Tissue-specific changes in apoplastic proteins and cell wall structure during cold acclimation of winter wheat crowns. J Exp Bot 69:1221–1234. https://doi.org/10.1093/jxb/erx450
Xia Q, Ponnaiah M (2018) Integrating proteomics and enzymatic profiling to decipher seed metabolism affected by temperature in seed dormancy and germination. Plant Sci 269:118–125. https://doi.org/10.1016/j.plantsci.2018.01.014
Xu G, Li C, Yao Y (2009) Proteomics analysis of drought stress-responsive proteins in Hippophae rhamnoides L. Plant Mol Biol Report 27:153–161. https://doi.org/10.1007/s11105-008-0067-y
Ye G, Ma Y, Feng Z, Zhang X (2018) Transcriptomic analysis of drought stress responses of sea buckthorn (Hippophae rhamnoides subsp. sinensis) by RNA-Seq. PLoS ONE. https://doi.org/10.1371/journal.pone.0202213
Ye W, Hu S, Wu L et al (2016) White stripe leaf 12 (WSL12), encoding a nucleoside diphosphate kinase 2 (OsNDPK2), regulates chloroplast development and abiotic stress response in rice (Oryza sativa L.). Mol Breed 36:1–15. https://doi.org/10.1007/s11032-016-0479-6
Zenda T, Liu S, Wang X et al (2019) Key maize drought-responsive genes and pathways revealed by comparative transcriptome and physiological analyses of contrasting inbred lines. Int J Mol Sci 20:1–30. https://doi.org/10.3390/ijms20061268
Zeng XuL, Singh A et al (2015) Involvement of calmodulin and calmodulin-like proteins in plant responses to abiotic stresses. Front Plant Sci 6:1–12. https://doi.org/10.3389/fpls.2015.00600
Zheng W, Komatsu S, Zhu W et al (2016) Response and defense mechanisms of taxus chinensis leaves under UV-A radiation are revealed using comparative proteomics and metabolomics analyses. Plant Cell Physiol 57:1839–1853. https://doi.org/10.1093/pcp/pcw106
Zhou L, Cheung M, Li M et al (2010) Rice hypersensitive induced reaction protein 1 (OsHIR1) associates with plasma membrane and triggers hypersensitive cell death. BMC Plant Biol. https://doi.org/10.1186/1471-2229/10/290
Acknowledgements
This work was supported by financial assistance from Department of Biotechnology (IBSD/A1/P(PH-2)/4) to RD. BS is thankful to DBT for providing fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
42485_2021_63_MOESM1_ESM.xlsx
Supplementary file1 Dataset containing complete list of common and differentially accumulated proteins in Trans-Himalayan and Sikkim seabuckthorn leaves (XLSX 29 kb)
42485_2021_63_MOESM2_ESM.xlsx
Supplementary file2 List of unique proteins identified in leaves of H.rhamnoides, H.tibetana and H.salicifolia (XLSX 29 kb)
42485_2021_63_MOESM4_ESM.xlsx
Supplementary file4 Comparative proteome profiling in Trans-Himalayan and Sikkim Himalayan seabuckthorn leaves hints towards differential stress adaptive responses (DOCX 219 kb)
Rights and permissions
About this article
Cite this article
Sharma, B., Deswal, R. “Comparative proteome profiling of seabuckthorn leaves from low altitude ‘Sikkim’ and high altitude ‘Himachal Pradesh’ Himalayan region hints towards differential stress adaptive responses”. J Proteins Proteom 12, 125–141 (2021). https://doi.org/10.1007/s42485-021-00063-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42485-021-00063-0