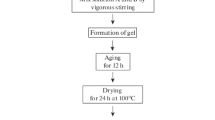
Abstract: Magnesium hydroxide (Mg(OH)2 or brucite) and magnesium oxide (MgO or magnesia) are essential raw materials for many industrial applications. Several studies have explored their production methods and the changes in their properties caused by adjustments in synthesis and calcination conditions. However, the ways the particles' microstructure changes along a full thermal treatment up to sintering remain unclear. This study investigated the effects of thermal treatments (120-1500 °C) on the physical properties and microstructure of clusters of Mg(OH)2 nanoparticles prepared by soluble-Mg-salt precipitation assisted by a surfactant agent.







Similar content being viewed by others
References
Shand, M.A. The Chemistry and technology of magnesia. New Jersey: John Wiley & Sons, Inc. (2006), ISBN-13: 978-0-471-65603-6
Salomão, R., Pandolfelli, V.C.: Magnesia sinter hydration-dehydration behavior in refractory castables. Ceram. Int. 34 (2008) [8] 1829-1834
Papich, M.G.: Magnesium hydroxide. In: Sauder's Handbook of Veterinary Drugs (4th Edition). Amsterdam: Elsevier (2016) 467-468, ISBN: 9780323244855
Shen, S., Chow, P.S., Chen, F., Tan, R.B.H.: Submicron particles of SBA-15 modified with MgO as carriers for controlled drug delivery. Chem. Pharm. Bull. 55 (2007) [7] 985-991
Giorgi, R., Chelazzi, D., Baglioni, P.: Conservation of acid waterlogged shipwrecks: Nanotechnologies for de-acidification. App. Phys. A. 83 (2006) 567-571
Holloway, L.R.: Application of magnesium hydroxide as a flame retardant and smoke suppressant in elastomers. Rubb. Chem. Tech. 61 (1987) [2] 186-193
Norton, R.N., Hornsby, P.R.: Flame retardant effects of magnesium hydroxide. Pol. Deg. Stab. 54 (1996) 383-385
Qiu, L., Xie, R., Ding, P., Qu, B.: Preparation and characterization of Mg[OH]2 nanoparticles and flame-retardant property of its nanocomposites with EVA. Compos. Struc. 62 (2003) 391-395
Gui, H., Zhang, X., Liu, Y., Dong, W., Wang, Q., Gao, J., Song, Z., Lai, J., Qiao, J.: Effect of dispersion of nano-magnesium hydroxide on the flammability of flame retardant ternary composites. Compos. Sci. Tech. 67 (2007) 67 974-980
Salomão, R., Souza, A.D.V., Cardoso, P.H.L.: A comparison of Al(OH)3 and Mg(OH)2 as inorganic porogenic agents for alumina. Interceram: Int. Cer. Rev. 64 (2015) [4] 193-199
Hüttig, G.F.: Die Kinetic der Alterung von aktivem Magnseiumoxyd. Kolloid-Zeitschrift 124 (1951) [3] 160-163
Finch, G.I., Sinha, K.P.: On reaction in the solid state. Proc. Roy. Soc. A. 239 (1957) 145-153
Razouk, R.I., Mikhail, R.S.: The hydration of magnesium oxide from vapor phase. J. Phys. Chem. 62 (1958) 920-925
Glasson, D.R.: Reactivity of lime and related oxides: Hydration of magnesium oxide. J. App. Chem. 13 (1963) [3] 119-123
Layden, G.K., Brindley, G.W.: Kinetics of vapor-phase hydration of magnesium oxide. J. Am. Cer. Soc. 46 (1963) [11] 518-522
Anderson, P.J., Horlock, R.F., Oliver, J.F.: Interaction of water with the magnesium oxide surface. Trans. Farad. Soc. 61 (1965) [516] 2754-2762
Feitknecht, W., Braum, H.: Der Mechanismus der Hydratation von Magnesiumoxid mit Wasserdampf. Helv. Chim. Act. 50 (1967) [7] 2040-2053
Smithson, C.L., Bakhshi, N.N.: The kinetics and mechanism of the hydration of magnesium oxide in a batch reactor. Can. J. Chem. Eng. 47 (1969) [6] 508-513
Birchal, V.S., Rocha, S.D.F., Mansur, M.B., Ciminelli, V.S.T.: A simplified mechanistic analysis of the hydration of magnesia. Can. J. Chem. Eng. 79 (2001) [4] 507-511
Rocha, S.D.F., Mansur, M.B., Ciminelli, V.S.T.: Kinetics and mechanistic analysis of caustic magnesia hydration. J. Chem. Tech. Biotech. 79 (2004) [8] 816-821
Kitamura, A., Onizuka, K., Tanaka, K.: Hydration characteristics of magnesia. Taikabutsu Overs. 16 (1995) [3] 3-11
Salomão, R., Arruda, C.C., Kawamura, M.A.: A systemic investigation on the hydroxylation behavior of caustic magnesia and magnesia sinter. Ceram. Int. 41 (2015) [10] 13998-14007
Liu, J.P., Wang, Y.J., Tian, Q., Zhang, S.Z.: Modeling hydration process of magnesia based on nucleation and growth theory: The isothermal calorimetry study. Thermoch. Act. 550 (2012) [20] 27-32
Salomão, R., Arruda, C.C., Souza, A.D.V., Fernandes, L.: Novel insights into MgO hydroxylation: Effects of testing temperature, samples' volume and solid load. Ceram. Int. 40 (2014) [9B] 14809-14815
Chatterji, S.: Mechanism of expansion of conc. due to the presence of dear-burnt CaO and MgO. Cem. Conc. Res. 25 (1995) 51-56
Salomão, R., Bittencourt, L.R.M., Pandolfelli, V.C.: A novel approach for magnesia hydration assessment in refractory castables. Ceram. Int. 33 (2007) [5] 803-810
Khangaonkar, P.R., Othman, R., Ranjitham, M.: Studies on particle breakage during hydration of calcined magnesite. Min. Eng. 3 (1990) [1-2] 227-235
Sutcu, M., Akkurt, S., Okur, S.: A microstructural study of surface hydration on a magnesia refractory. Ceram. Int. 36 (2010) [5] 1731-1735
Kaneyasu, A., Yamamoto, S., Yoshida, A.: Magnesia raw materials with improved hydration resistance. Taikabutsu Overs. 17 (1996) [2] 21-26
Amaral, L.F., Oliveira, I.R., Salomão, R., Frollini, E., Pandolfelli, V.C.: Temperature and common-ion effect on magnesium oxide (MgO) hydration. Ceram. Int. 36 (2010) [3] 1047-1054
Amaral, L.F., Oliveira, I.R., Bonadia, P., Salomão, R., Pandolfelli, V.C.: Chelants to inhibit magnesia [MgO] hydration. Ceram. Int. 37 (2011) [5] 1537-1542
Salomão, R., Pandolfelli, V.C.: Citric acid as anti-hydration additive for magnesia containing refractory castables. Ceram. Int. 37 (2011) [6] 1839-1842
Souza, T.M., Luz, A.P., Pandolfelli, V.C.: Magnesium fluoride role on alumina-magnesia Cem.-bonded castables. Ceram. Int. 40 (2014) [9] 14947-14956
Salomão, R. Bittencourt, L.R.M., Pandolfelli, V.C.: A novel magnesia based binder [MBB] for refractory castables. Interceram: Int. Cer. Rev. Refrac. Man. (2009) 21-24
Souza, T.M., Luz, A.P., Bráulio, M.A.L., Pagliosa, C., Pandolfelli, V.C.: Acetic acid role on magnesia hydration for Cem.-free refractory castables. J. Am. Cer. Soc. 97 (2014) [4] 1233-1241
Henrist, C., Mathieu, J.P., Vogels, C., Rulmont, A., Cloots, R.: Morphological study of magnesium hydroxide nanoparticles precipitated in dilute aqueous solution. J. Cryst. Grow. 249 (2003) 321-330
Lv, J., Qiu, L., Qu, B.: Controlled growth of three morphological structures of magnesium hydroxide nanoparticles by wet precipitation method. J. Cryst. Grow. 267 (2004) 676-684
Pokrovsky, O.S., Schott, J.: Experimental study of brucite dissolution and precipitation in aqueous solutions: Surface specification and chemical affinity control. Geochim. Cosmochim. Act. 68 (2004) [1] 31-45
Hsu, J.P., Nacu, A.: Preparation of submicron-sized Mg[OH]2 particles through precipitation. Coll. Surf. A. 262 (2005) 220-231
Yan, C., Xue, D., Zou, L., Yan, X., Wang, W.: Preparation of magnesium hydroxide nanoflowers. J. Cryst. Grow. 282 (2005) 448-454
Chen, D., Zhu, L., Liu, P., Zhang, H., Xu, K., Chen M.: Rod-like morphological magnesium hydroxide and magnesium oxide via a wet coprecipitation process. J. Por. Mat. 16 (2009) [1] 13-18
Wang, W., Qiao, X., Chen, J., Li, H.: Facile synthesis of magnesium oxide nanoplates via chemical precipitation. Mat. Lett. 61 (2007) 3218-3220
Yoshida, T., Tahaka, T., Yoshida, H., Funabiki, T., Yoshida, S., Murata, T.: Study of dehydration of magnesium hydroxide. J. Phys. Chem. 99 (1995) [27] 10890-10896
Mishakov, I.V., Bedilo, A.F., Richards, R.M., Chesnokov, V.V., Volodin, A.M., Zaikovskii, V.I., Buyanov, R.A., Kladunde, K.J.: Nanocrystalline MgO as a dehydrohalogenation catalyst. J. Cat. 206 (2002) 40-48
Gregg, S.J., Razouk, R.I.: Kinetics of thermal decomposition of magnesium hydroxide. J. Chem. Soc. (1949) S36-44
Brewer, L., Porter, R.F.: A thermodynamic and spectroscopic study of gaseous magnesium oxide. J. Chem. Phys. 22 (1954) 1867-1877
Goodman, J.F.: The decomposition of magnesium hydroxide in an electron microscope. In: Proc. Roy. Soc. Lon. A. 247 (1958) [1250] 346-352
Ball, M.C., Taylor, F.W.: The dehydration of brucite. Min. Mag. and J. Min. Soc. 32 (1961) [253] 754-766
Anderson, P.J., Horlock, R.F.: Thermal decomposition of magnesium hydroxide. Trans. Farad. Soc. 58 (1962) [475] 1993-2004
Turner, R.C., Hoffman, I., Chen, D.: Thermogravimetry of the dehydration of Mg[OH]2. Can. J. Chem. 41 (1963) [2] 243-251
Pampuch, R., Librant, Z., Piekarczyk, J.: Texture and sinterability of MgO powders. Ceram. Inter. 1 (1975) [1] 14-18
Gordon, R.S., Kingery, W.D.: Thermal decomposition of brucite: I, Electron and optical microscope studies. J. Am. Cer. Soc. 49 (1966) [12] 654-660
Brett, N.H., Anderson, P.J.: Mechanism of decomposition of brucite. Trans. Farad. Soc. 63 (1967) 2044-2050
Liu, C., Liu, T., Wang, D.: Non-isothermal kinetics study on the thermal decomposition of brucite by thermogravimetry. J. Therm. Anal. Calor. 134 (2018) 2399-2347
Kim, M.G., Dahmen, U., Searcy, A.W.: Structural transformation in decomposition of Mg[OH]2 and MgCO3. J. Am. Cer. Soc. 70 (1987) [3] 146-154
Phillips, V.A., Opperhauser, H., Kolbe, J.L.: Relations among particle size, shape and surface area of Mg[OH]2 and its calcination product. J. Am. Cer. Soc. 61 (1978) [1] 78-81
Green, J.: Calcination of precipitated Mg[OH]2 to active MgO in production of refractory and chemical grade MgO. J. Mat. Sci. 18 (1983) 637-651
Wang, J.A., Novaro, A.O., Bokhimi, X., López, T., Gómez, R., Navarrete, J., Llanos, M.E., López-Salina, E.: Characterizations of the thermal decomposition of brucite prepared by sol-gel technique for synthesis of nanocrystalline MgO. Mat. Lett. 35 (1998) 317-323
Kabalah-Amitai, L., Mayzel, B., Kauffmann, Y., Fitch, A.N., Bloch, L., Gilbert, P.U.P.A., Pokroy, B.: Vaterite crystals containing two interspersed crystal structures. Science 340 (2013) [6131] 454-457
Wang, C., He, C., Tong, Z., Liu, X., Ren, B., Zeng, F.: Combination of adsorption by porous CaCO3 microparticles and encapsulation by polyelectrolyte multilayer films for sustained drug delivery. Int. J. Pharm. 308 (2006) 160-167
Costa, L.M.M., Sakihama, J., Salomão, R.: Characterization of porous calcium hexaluminate produced from calcined alumina and microspheres of Vaterire (µ-CaCO3). J. Eur. Cer. Soc. 38 (2018) 5208-5218
Fernandes, L., Salomão, R.: Preparation and characterization of mullite-alumina structures formed "in situ" from calcined alumina and different grades of synthetic amorphous silica. Mat. Res. 21 (2018) [3] e20170783
Cimino, A., Porta, P., Valigi, M.: Dependence on the lattice parameter of magnesium oxide on crystallite size. J. Am. Cer. Soc. 49 (1966) [3] 152-156
Guilliat, I.F., Brett, N.H.: X-ray line broadening as a measure of crystallite size in oxide powders. Phil. Mag. 21 (1970) [172] 671-680
Wuensch, B.J., Vasilos, T.: Grain-boundary diffusion in MgO. The American Ceramic Soc. 47 (1964) [2] 63-68
Robertson, W.M.: Kinetics of grain boundary grooving on magnesium oxide. In: Kuczynski, G.C., Hooton, N.A., Gibbon, C.F., eds. Sintering and Related Phenomena. New York. Gordon and Breach, Science Publishers (1965) 215-230. Library of Congress Catalog Card 67-26576. LCCN Permalink: https://lccn.loc.gov/67026576.
Lowrie, R.C., Cutler, I.B.: The effect of porosity on the rate of grain growth of magnesia. In: Kuczynski, G.C., Hooton, N.A., Gibbon, C.F., eds. Sintering and Related Phenomena. New York. Gordon and Breach, Science Publishers (1965) 527-539. Library of Congress Catalog Card 67-26576. LCCN Permalink: https://lccn.loc.gov/67026576.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Salomão, R., Arruda, C. & Antunes, M. Synthesis, Dehydroxylation and Sintering of Porous Mg(OH)2-MgO Clusters: Evolution of Microstructure and Physical Properties. Interceram. - Int. Ceram. Rev. 69, 52–62 (2020). https://doi.org/10.1007/s42411-019-0067-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42411-019-0067-y