Abstract
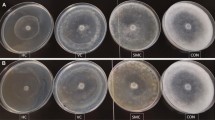
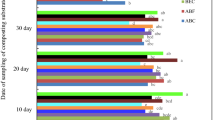
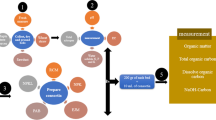
Effectiveness of certain agro wastes in different combinations was ascertained in survival and multiplication of Trichoderma harzianum, in order to improve its shelf life for more than 120 days. Population changes of T. harzianum were followed in five composts prepared from on farm wastes, Farm yard manure and amended soil for a period of 80 days at an interval of 20 days. Maximum population of T. harzianum was estimated in Prosopis juliflora compost. Proportions of P. juliflora compost were then standardized and different amount of P. juliflora compost and FYM were modified by adopting Indore type of composting. In subsequent experiment, most promising combination of P. juliflora compost mixed in talc and various food substrates viz., barley meal, wheat bran, pulse meal and wood saw dust were tested for their effectiveness in improving survival of T. harzianum. Studies revealed that the population of T. harzianum was ten-fold higher in the substrate unsterilized P. juliflora compost, wood saw dust + talc compared to other substrates used. This study showed that a combination of agro wastes (P. juliflora compost + wood saw dust) and inert material promote survival and multiplication of T. harzianum for a period of more than 105 days. P. juliflora is abundantly growing shrub in Indian arid region and its compost also reduces charcoal rot caused by Macrophomina phasolina in crops grown under rain fed conditions.

Similar content being viewed by others
References
Adhikari BK, Barrington S, Martinez J, King S (2008) Characterization of food wastes and bulking agents for composting. Waste Manag 28:795–804
Aghnoom R, Falahati-Rastegar M, Jafarpour B (1999) Comparison of chemical and biological control of cumin wilt (Fusarium oxysporum f. sp. cumini) in laboratory and greenhouse condition. Iran J Agri Sci 30:619–630
Bareja M, Mawar R, Mathur M, Lodha S (2013) On –farm waste-based composts suppressive to Macrophomina phaseolina inciting dry root rot of guar in an arid environment. Aust Plant Path 42:9–16
Bora J, Das BC, Borakoty K, Dutta PK (2010) Studies on the growth and multiplication of Trichoderma harzianum and Trichoderma viride on different compost manures and their population dynamics in vitro and in vivo conditions. J Biol Control 24:253–256
Chaudhari PJ, Shrivastava P, Khadse AC (2011) Substrate evaluation for mass cultivation of Trichoderma viride. Asiatic J Biotech Res 2:441–446
Elad Y, Chet I, Katan J (1980) Trichoderma hamatum: A biological agent effective against Sclerotium rolfsii and Rhizoctonia solani. Phytopathology 70:119–121
Elad Y, Chet I, Henis Y (1981) A selective medium for improving quantitative isolation of Trichoderma species from soil. Phytoparasitica 9:59–67
Howard A, Wad YD (1931) The waste products of agriculture and their utilization as humus. Oxford University, London
Ikram N, Dawar S (2013) Effect of Prosopis juliflora (Sw.) DC in the control of root rot fungi of cowpea (Vigna unguiculata L.) and mung bean {Vigna radiata (L.) Wilczeck}. Pak J Bot 45:649–654
Jayaraj J, Rambadran R (1996) Evaluation of certain organic substrates and adjuvants for the mass multiplication of Trichoderma harzianum Rifai. J Biol Control 10:129–131
Jayarajan R, Ramakrishanan G, Dinkaran D, Sridar R (1994) Development of products of Trichoderma viride and Bacillus subtilis for bio-control of root rot diseases. In: Dwivedi BK (ed) Biotechnology in India. Bioved Research Society, Allahabad, pp 25–36
Kaushal S, Chandel S (2017) Enhancing the shelf life of Trichoderma species by adding antioxidants producing crops to various substrates. J Crop Prot 6(3):307–314
Khan HA, Lodha V, Ghanim A (1992) Triacontanol from leaves of Prosopis cineraria and Prosopis juliflora. Trans ISDT 17:29–32
Lalithakumari D, Manibhushanrao K (2001) Use of bio-control strategies in plant disease management. In: National Symposium on Eco-friendly approaches for plant disease management held at Chennai, Jan 22–24
Lennox JA, Abriba C, Alabi BN, Akubuenyi FC (2010) Comparative degradation of saw dust by microorganism isolated from it. African J Microbiol Res 4:1804–1807
Lodha S (1995) Soil solarization, summer irrigation and amendments for the control of Fusarium oxysporum f. sp. cumini and Macrophomina phaseolina in arid soils. Crop Prot 14:215–219
Lodha S (1996) Influence or soil moisture conservation techniques on Macrophomina phaseolina population, dry root rot and yield of clusterbean. Indian Phytopath 49:342–349
Lodha S, Mawar R (2010) Efficacy of native bio-control agents on soil microflora, dry root rot incidence and seed yield of rainfed arid crops. Indian Phytopath 63:195–198
Lodha S, Bohra MD, Harsh LN (1999) Evaluation of Prosopis pods as a source of’ carbohydrate for enhancing the growth of soil borne fungi. Indian Phytopath 52:24–27
Mawar R, Lodha S (2002) Brassica amendments and summer irrigation for the control of Macrophomina phaseolina and Fusarium in hot arid region. Phytopathol Mediterr 41:45–54
Mawar R, Lodha SSV (2017) Combining food substrates for improved survival of Aspergillus versicolor, a bioagent. Biopestic Int 13:140–148
Mohiidin FA, Khan MR, Khan SM, Bhat BH (2010) Why Trichoderma is considered super hero (super fungus) against the evil parasites? Plant Pathol J 9(3):92–102
Mustafa A, Khan MA, Pervez MAKSH (2009) Mass multiplication of Trichoderma spp. on organic substrate and their effect in management of seed borne fungi. Pak J Phytopathol 21:108–114
Nemec S, Datnoff LE, Strandberg J (1996) Efficacy of biocontrol agents in planting mixes to colonize plant roots and control root disease of vegetables and citrus. Crop Prot 15:735–742
Oluchukwu AC, Nebechukwu AG, Egbuna SO (2018) Enrichment of nutritional contents of sawdust by composting with other nitrogen rich agr-wastes for bio-fertilizer synthesis. J Chem Tech Metall 53:430–436
Simon S, Anamika (2011) Agro-based waste products as a substrate for mass production of Trichoderma spp. J Agri Sci 3:168–171
Singh A, Srivastava S, Singh HB (2007) Effect of substrates on growth and shelf life of Trichoderma harzianum and its use in biocontrol of diseases. Biores Technol 98:470–473
Smolinska U, Kowalska B, Kowalczyk W, Szczech M (2014) The use of agro-industrial wastes as carriers of Trichoderma fungi in the parsley cultivation. Sci Horti 179:1–8
Snedecor GE, Cochran WJ (1967) Statistical methods. Oxford and IBH, Calcutta
Tawfik AA, Allam ADA (2004) Improving cumin production under soil infestation with fusarium wilt pathogen: I-screening of biocontrol agents. Ass Univ Bull Environ Res 7(2):35–45
Tengerdy RP, Szakacs S (2003) Bioconversion of lignocellulose in solid substrate fermentation. Bio Chem Eng J 13:169–179
Tewari JC, Harris PJC, Harsh LN, Cadoret K, Pasiecznik NM (2000) Managing Prosopis julifora (Vilayati babul)—a technical manual. CAZRI, Jodhpur
Whipps JM (1997) Developments in the biological control of plant pathogens. Adv Bot Res 26:1–13
Acknowledgements
Authors are grateful to SERB–DST, New Delhi, Government of India, for providing financial support under Core Research Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declared that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mawar, R., Mathur, T. Enhancing survival and multiplication of Trichoderma harzianum by Prosopis juliflora based compost in Indian arid region. Indian Phytopathology 75, 797–805 (2022). https://doi.org/10.1007/s42360-022-00495-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42360-022-00495-8